Localized electrons to Molecular orbitals Hybridization The s

Localized electrons to Molecular orbitals

Hybridization The s, p, d, and f orbitals work when defining electron configurations in single atoms; however, when atoms bond the s, p, d, f arrangement no longer applies. Hydridization is the combination of s and p orbitals to make bonding orbitals.

Types of Hybridization Electron density Shapes areas/Bonding areas sp 3 4 Tetrahedral Trigonal pyramidal Bent 105° sp 2 3 Trigonal planar Bent 120° sp 2 Linear
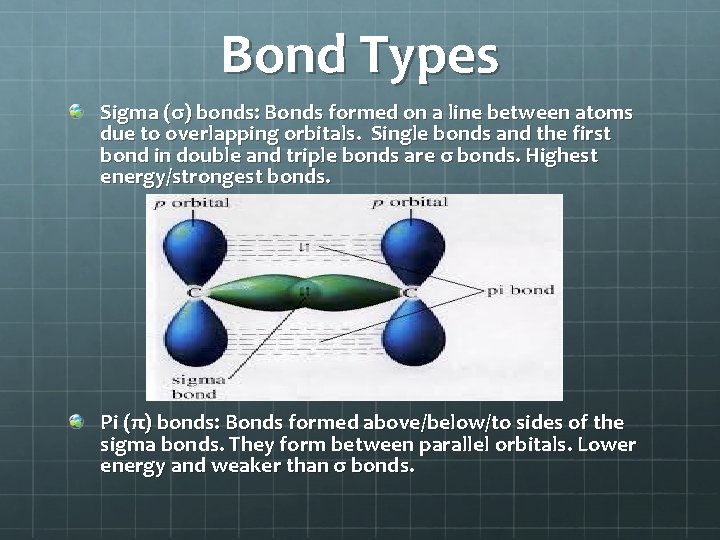
Bond Types Sigma (σ) bonds: Bonds formed on a line between atoms due to overlapping orbitals. Single bonds and the first bond in double and triple bonds are σ bonds. Highest energy/strongest bonds. Pi (π) bonds: Bonds formed above/below/to sides of the sigma bonds. They form between parallel orbitals. Lower energy and weaker than σ bonds.
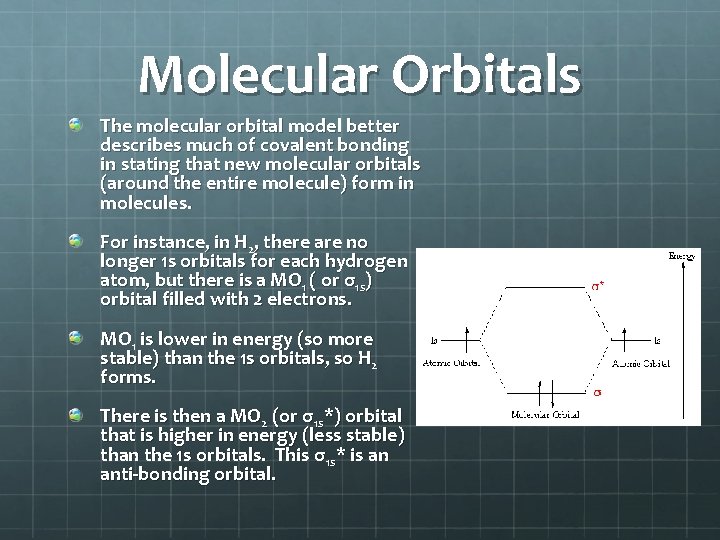
Molecular Orbitals The molecular orbital model better describes much of covalent bonding in stating that new molecular orbitals (around the entire molecule) form in molecules. For instance, in H 2, there are no longer 1 s orbitals for each hydrogen atom, but there is a MO 1 ( or σ1 s) orbital filled with 2 electrons. MO 1 is lower in energy (so more stable) than the 1 s orbitals, so H 2 forms. There is then a MO 2 (or σ1 s*) orbital that is higher in energy (less stable) than the 1 s orbitals. This σ1 s* is an anti-bonding orbital.

Some simple Molecular orbital diagrams H 2 - He 2 Li 2 Be 2

An interruption Before we look at some more advanced molecular orbital diagrams we need to talk about magnatism Paramagnatism causes a substance to be attracted to a magnetic field. Paramagnetism is associated with unpaired electrons in molecular orbitals. Diamagnetism causes a substance to be repelled from a magnetic field. Diamagnetism is associated with paired electrons in molecular orbitals.

So when you get to boron… B 2 expected But B 2 is paramagnetic So they figured that orbitals mix and it goes like this.

Bond Order Bond order determines the strength of bonds. A positive bond order indicates bonds should form, and the greater the number the stronger the bond. A bond order that is zero indicates that a bond should not form. Bond order = (Number of bonding electrons-Number of anti-bonding electrons)/2 H 2 He 2 N 2 O 2

Molecular Orbitals and Bond Order You can use these molecular orbital diagrams and bond order for heteronuclear molecules. NO NO+ CNAnd to even explain bond polarity. HF
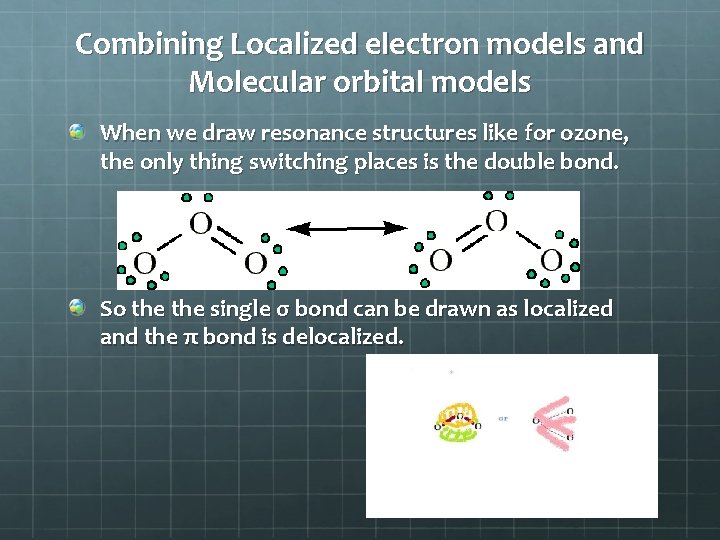
Combining Localized electron models and Molecular orbital models When we draw resonance structures like for ozone, the only thing switching places is the double bond. So the single σ bond can be drawn as localized and the π bond is delocalized.
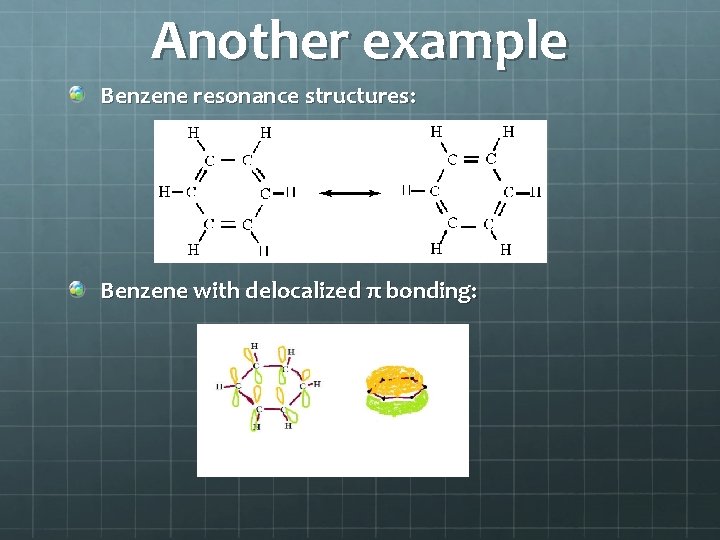
Another example Benzene resonance structures: Benzene with delocalized π bonding:
- Slides: 12