Living By Chemistry SECOND EDITION Unit 2 SMELLS

Living By Chemistry SECOND EDITION Unit 2: SMELLS Molecular Structure and Properties

Lesson 48: Protein Origami Amino Acids and Proteins
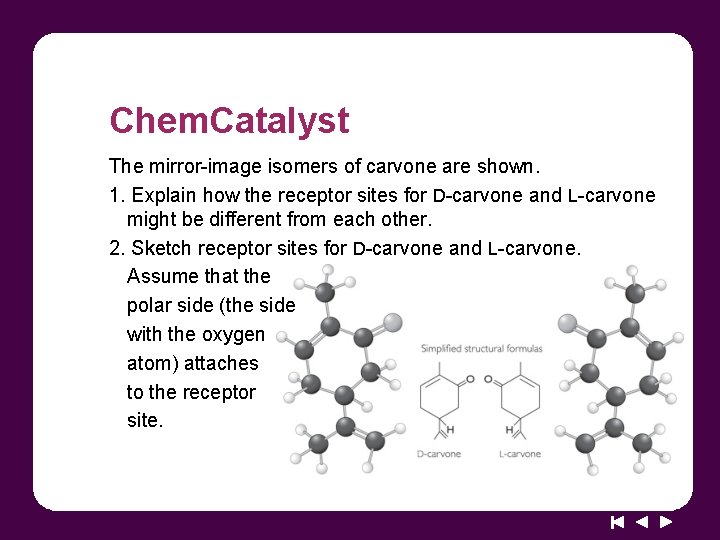
Chem. Catalyst The mirror-image isomers of carvone are shown. 1. Explain how the receptor sites for D-carvone and L-carvone might be different from each other. 2. Sketch receptor sites for D-carvone and L-carvone. Assume that the polar side (the side with the oxygen atom) attaches to the receptor site.

Key Question What is a receptor site made of?

You will be able to: • explain that protein molecules are chains of amino acid molecules • understand that the smell receptor sites are protein chains folded to form a receptor of a specific shape • explain the “handedness” of a smell receptor site

Prepare for the Activity Work in groups of four. Amino acid: A molecule with a carboxyl functional group and an amine functional group. Protein: A large molecule consisting of amino acids bonded together.

Discussion Notes Amino acids are molecules with a carboxyl group, COOH, and an amine group, NH 2. Amino acids can link together to form long chains called proteins. Protein molecules fold to form pockets because of attractions between the R groups on different amino acids in the chain.

Wrap Up What is a receptor site made of? • Amino acids are molecules with a carboxyl group, COOH, and an amine group, NH 2. • Amino acids all have mirror-image isomers. Only left-handed amino acid molecules function in the human body. • The carboxyl group from one amino acid can link with the amine group of another amino acid forming a peptide bond.

Wrap Up (cont. ) • Proteins are long chains of amino acids linked together. • Protein molecules can fold to form smell receptor sites.

Check-In Name two concepts from the Smells Unit that were used today to help you understand proteins.
- Slides: 10