Living By Chemistry SECOND EDITION Unit 1 ALCHEMY

Living By Chemistry SECOND EDITION Unit 1: ALCHEMY Matter, Atomic Structure, and Bonding

Lesson 6: A New Language Chemical Names and Symbols

Chem. Catalyst Two bottles are on a shelf in a chemistry lab. Both contain a shiny yellow metal. Bottle A is labeled Au(s). Bottle B is labeled Fe. S 2(s). 1. What do you think the symbols on the bottles mean? 2. Do you think both bottles contain gold? Why or why not?

Key Question What do chemical names and symbols tell you about matter?

You will be able to: • • • define the terms element, compound, and aqueous recognize whether a substance is an element or a compound based on its chemical formula or symbol decipher some basic chemical formulas and symbols

Prepare for the Activity Work in groups of eight.

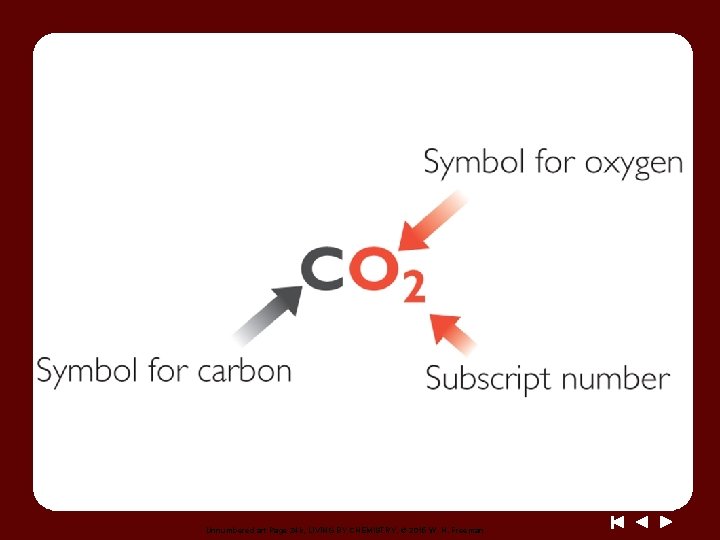
Discussion Notes The Language of Chemistry Symbol for sulfur Symbol for copper Symbol for oxygen Subscript for oxygen

Discussion Notes (cont. ) All matter in the universe either is an element or is made of some combination of elements. Element: A unique substance that cannot be broken down into simpler substances through chemical processes. Elements serve as the building materials of all matter. Compound: A pure substance that is a chemical combination of two or more elements in a fixed ratio.

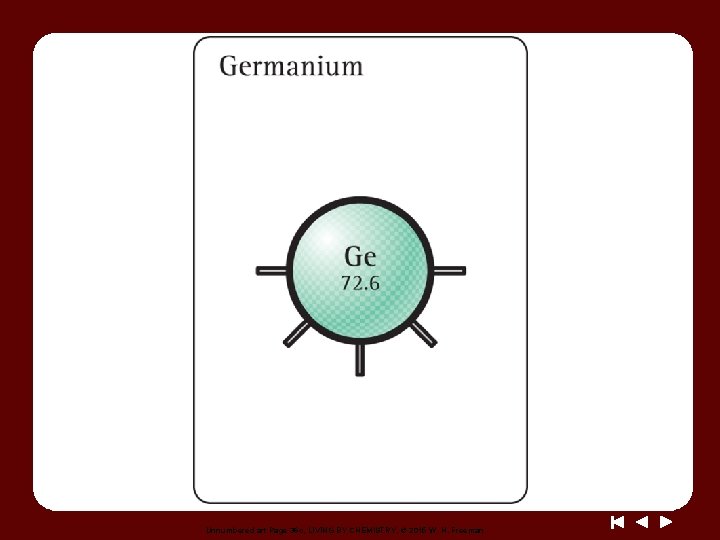
Discussion Notes (cont. ) The first letter of an element’s symbol is always uppercase, and if there is a second letter it is always lowercase. Compounds are represented by chemical formulas. Chemical formula: A combination of symbols and subscripts that indicates the number and types of elements in a compound.

Discussion Notes (cont. ) Some common groupings of elements have their own specific names. Substances come in different physical forms, called phases. Phase: The physical form a substance is in, such as solid, liquid, or gas. Phase is greatly influenced by temperature. Aqueous: A substance is aqueous when it is dissolved in water.

Wrap Up What do chemical names and symbols tell you about matter? • Chemical symbols represent the elements. Each element has either a one- or two-letter symbol. The first letter is always uppercase; the second letter is always lowercase. • The chemical formula of a substance indicates what elements are in it as well as the relative amounts of each element in that substance. • The symbols (s), (l), (g), and (aq) indicate the physical form or phase of a substance and whether it is dissolved in water.

Check-In Imagine that you find a vial labeled Na 2 SO 4(aq). What does the label tell you about what is in this vial?

Unnumbered art Page 23 b, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
Unnumbered art Page 24 k, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
Unnumbered art Page 27, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
Unnumbered art Page 32 a, LIVING BY CHEMISTRY, © 2015 W. H. Freeman

Unnumbered art Page 32 b, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
Unnumbered art Page 33 b, LIVING BY CHEMISTRY, © 2015 W. H. Freeman

Unnumbered art Page 35, LIVING BY CHEMISTRY, © 2015 W. H. Freeman

Lesson 8

Living By Chemistry SECOND EDITION Unit 1: ALCHEMY Matter, Atomic Structure, and Bonding

Lesson 8: What Goes Around Comes Around Conservation of Matter

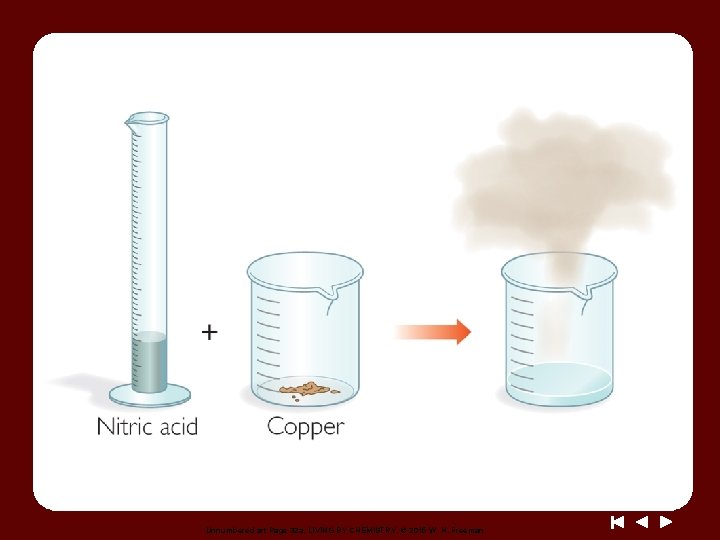
Chem. Catalyst What do you think happened to the copper powder in the copper cycle experiment when it was mixed with the nitric acid?

Key Question What happens to elements in a chemical change?

You will be able to: • explain that the product of chemical reactions depends on what was present at the time of reaction • explain that matter cannot be created or destroyed in a chemical reaction

Prepare for the Activity Work in groups of four.
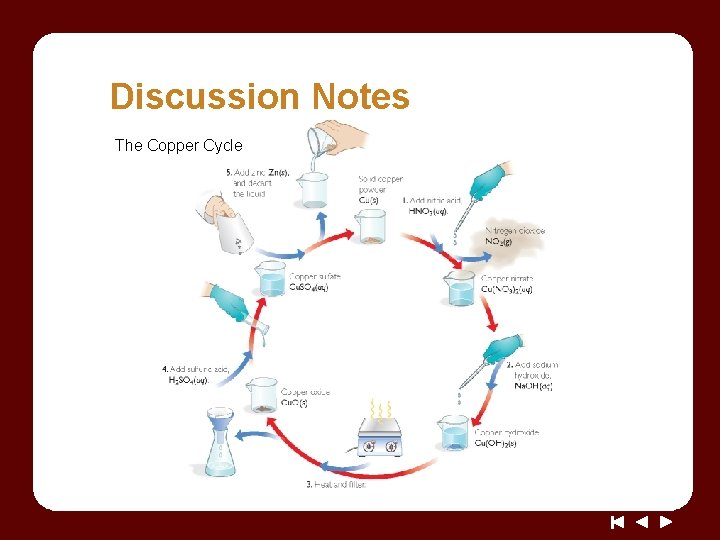
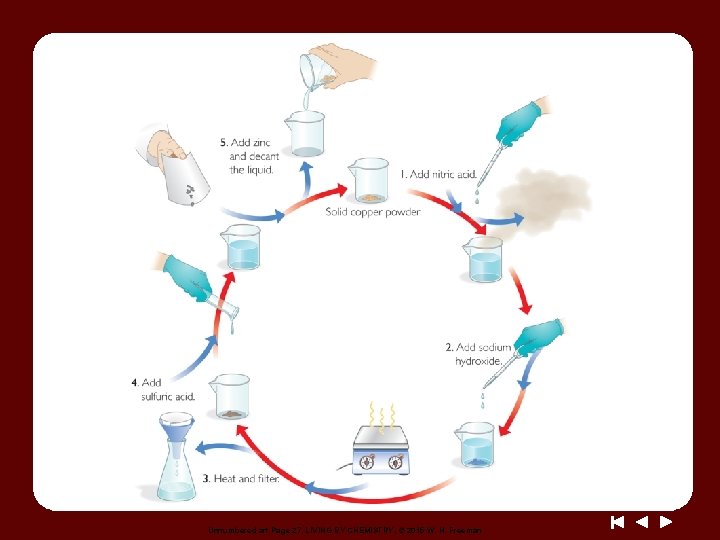
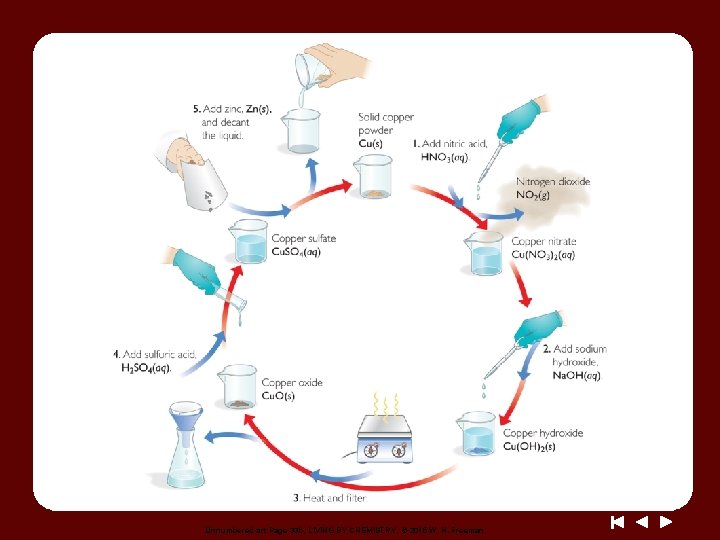
Discussion Notes The Copper Cycle

Discussion Notes (cont. ) You could prove that copper was not destroyed by measuring the mass of the copper powder at the beginning and at the end of the experiment. Law of conservation of mass: The law states that mass cannot be gained or lost in a chemical reaction–matter cannot be created or destroyed.

Discussion Notes (cont. ) No matter what was done to the copper, it was not broken down any further. The observations we have made in the copper cycle lab provide further evidence that the golden penny is not gold.

Wrap Up What happens to elements in a chemical change? • Elemental copper can be transformed through chemical reactions and then recovered. • We can represent elements with symbols and keep track of them during chemical reactions. • Elements combine and recombine but are not created or destroyed in chemical reactions.

Check-In Sodium chloride, Na. Cl (aq), is added to silver nitrate, Ag. NO 3 (aq), resulting in Na. NO 3 (aq) and a white solid. Identify the white solid from the list below. Explain your choice. A. Ag. Cl (s) B. Ag. Cl (aq) C. Ag. NO 3(s) D. Na. Cl (s)

Unnumbered art Page 36 a, LIVING BY CHEMISTRY, © 2015 W. H. Freeman

Unnumbered art Page 36 b, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
Unnumbered art Page 36 c, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
Unnumbered art Page 36 d, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
Unnumbered art Page 39, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
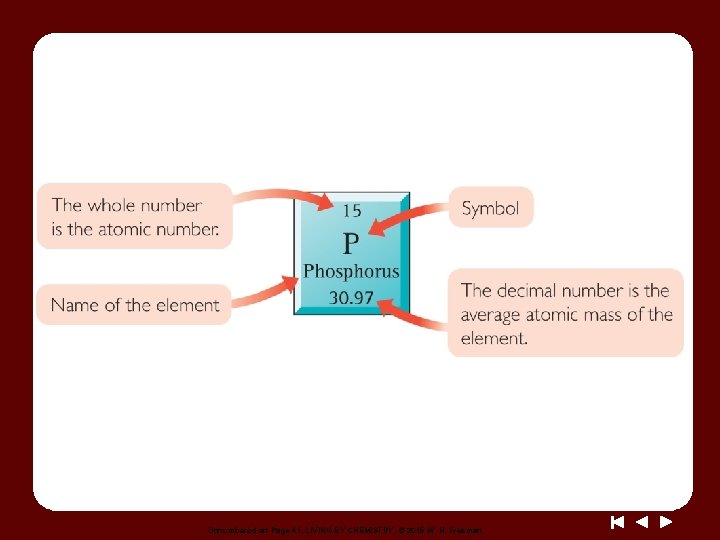
Unnumbered art Page 41, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
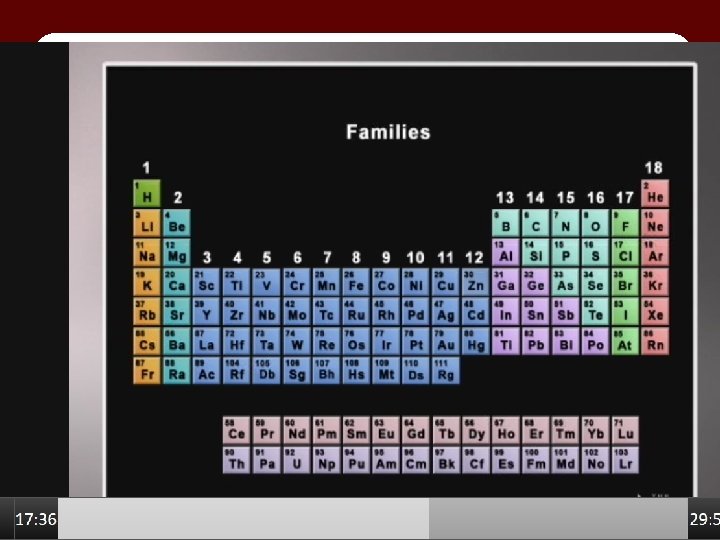
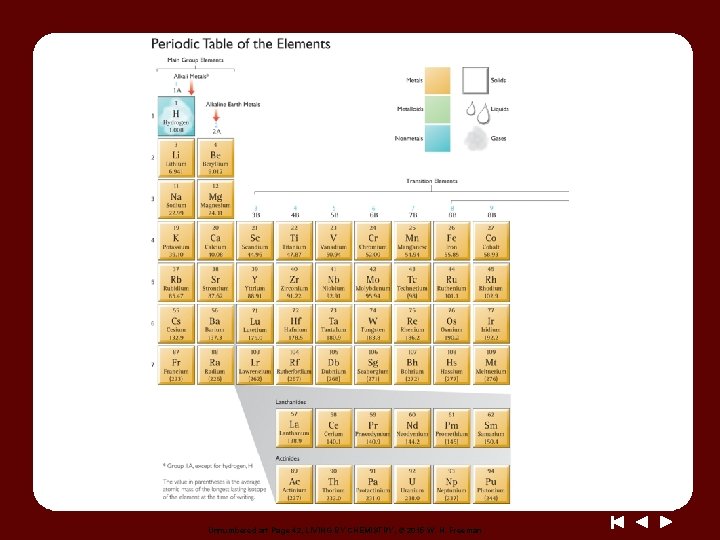
Unnumbered art Page 42, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
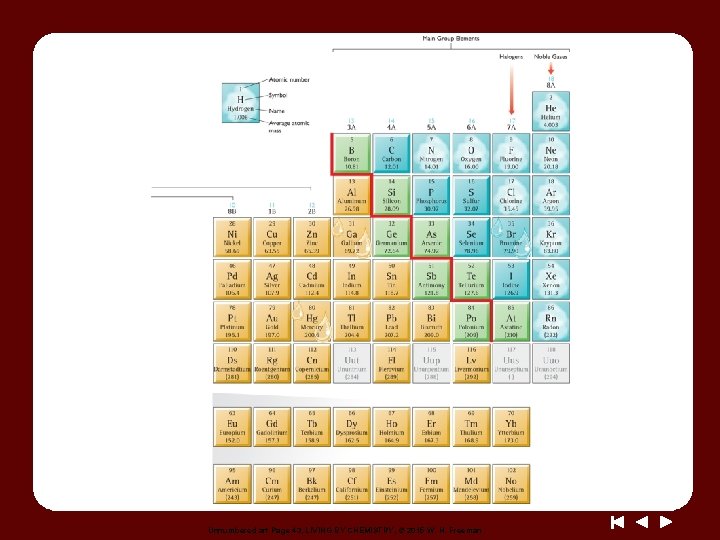
Unnumbered art Page 43, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
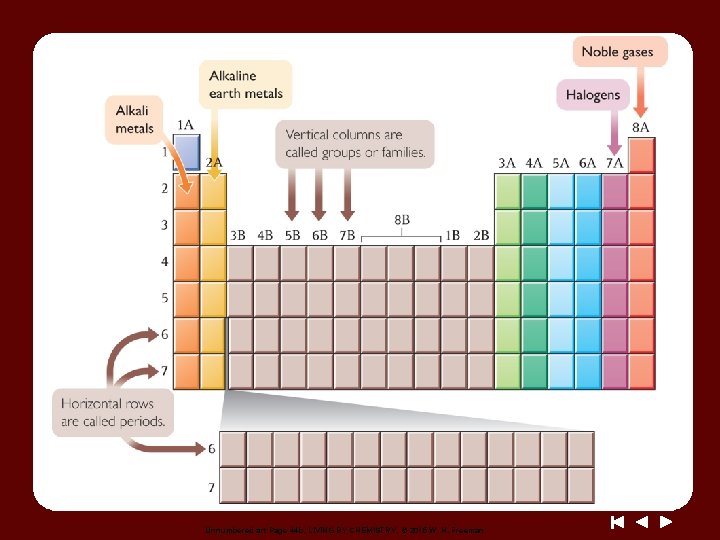
Unnumbered art Page 44 b, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
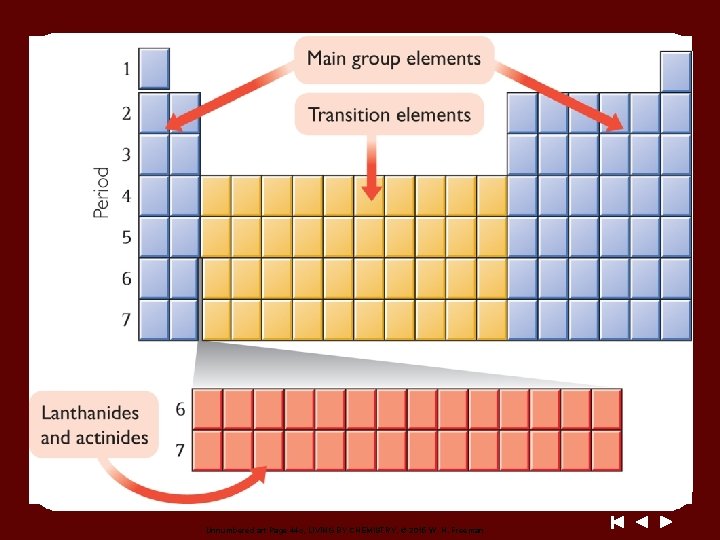
Unnumbered art Page 44 c, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
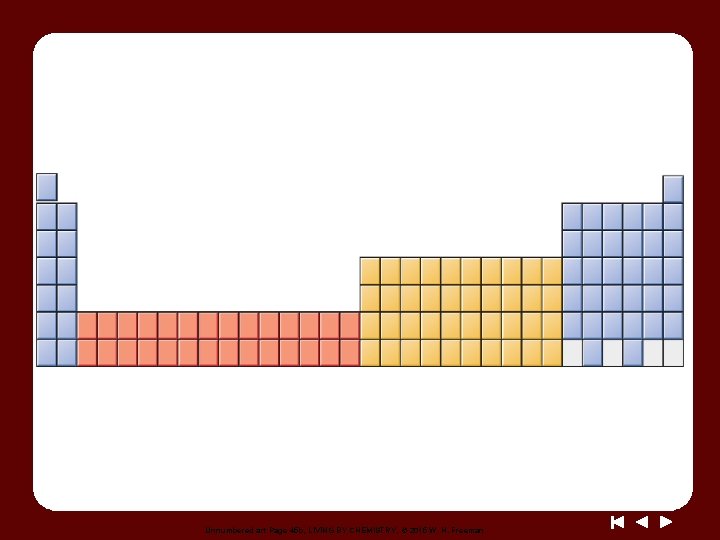
Unnumbered art Page 45 b, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
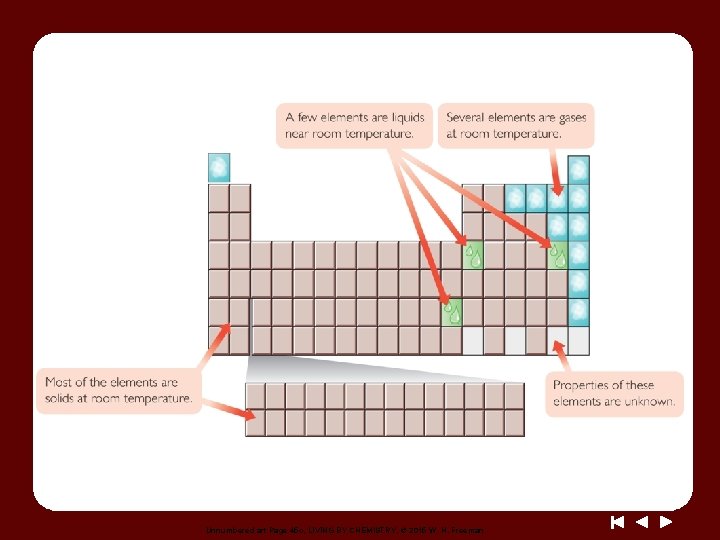
Unnumbered art Page 45 c, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
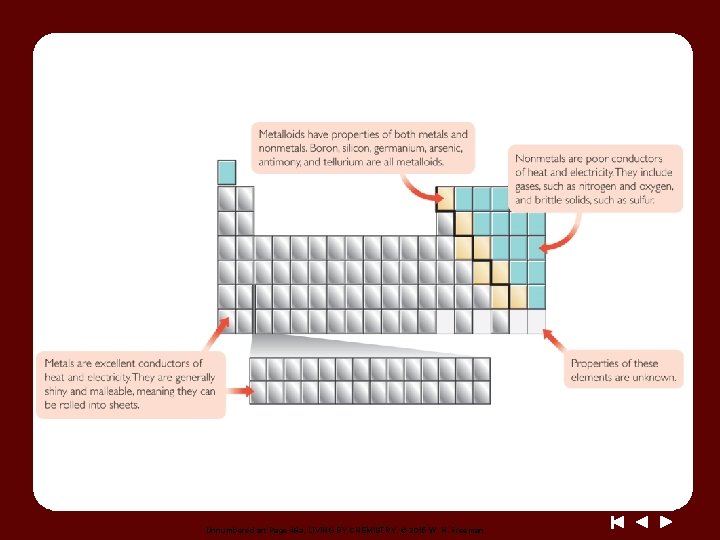
Unnumbered art Page 46 a, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
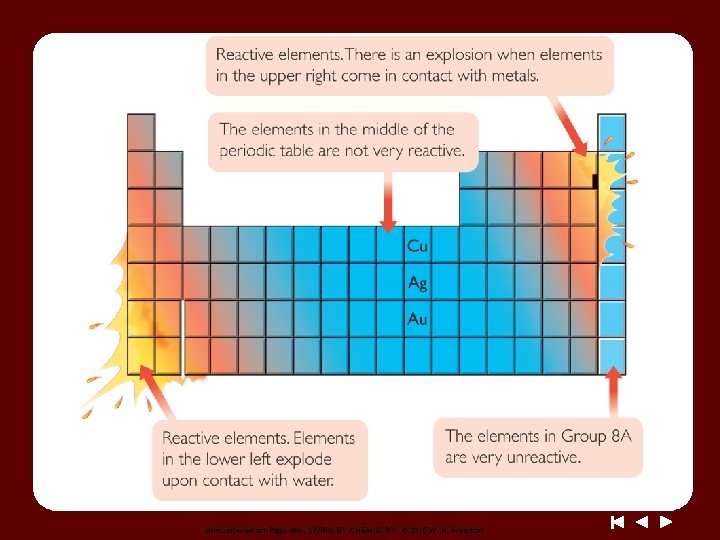
Unnumbered art Page 46 b, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
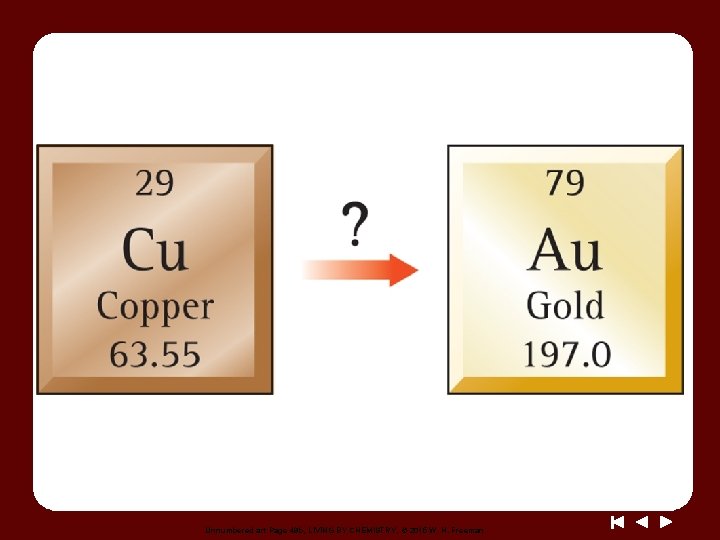
Unnumbered art Page 49 b, LIVING BY CHEMISTRY, © 2015 W. H. Freeman
- Slides: 47