LIVER IN PREGNANCY By Amr Abd Elmoty THE

LIVER IN PREGNANCY By Amr Abd Elmoty
THE LIVER IN PREGNANCY INTRODUCTION The liver is influenced by the physiologic state of pregnancy and thus abnormalities that may usually signify hepatic dysfunction may not represent actual liver damage and should be interpreted with caution. In addition, the anatomic location of the liver is changed because it is shifted superiorly as pregnancy progresses secondary to the enlarging gravid uterus. Therefore liver that is palpable below the right subcostal margin during pregnancy usually indicates an ongoing pathologic hepatic process.

Liver diseases unique to pregnancy can be classified: 1. Hyperemesis gravidarum (HG)—Usually occurs in the first trimester of pregnancy and is not associated with preeclampsia. 2. The syndrome of hemolysis, elevated liver enzymes, and low platelets (HELLP)—Usually occurs in the third trimester and is associated with preeclampsia. 3. Intrahepatic cholestasis of pregnancy (ICP)—Can occur at any time during pregnancy and is not associated with preeclampsia. 4. Acute fatty liver disease of pregnancy (AFLP)—Usually occurs in the third trimester and is associated with preeclampsia.

Associated with preeclampsia/eclampsia Not associated with preeclampsia/eclampsia Help syndrome Hyperemesis gravidarum Acute fatty liver disease of pregnancy Intrahepatic cholestasis of pregnancy First trimester Hyperemesis gravidarum Second trimester Third trimester Help syndrome Acute fatty liver of pregnancy Intrahepatic cholestasis of pregnancy

Changes in liver anatomy and function during normal pregnancy Liver anatomy and histology Anatomically, the gross appearance of the liver does not change. Histologically, subtle changes that include: 1. Increased variability in hepatocyte size and shape. 2. Enhanced granularity of hepatocyte cytoplasm. 3. Increased numbers of cytoplasmic fat vacuoles in centrilobular hepatocytes. 4. Hypertrophied Kupffer cells. 5. Hepatocytes exhibit proliferation of the smooth and rough endoplasmic reticula; and giant mitochondria.

Hemodynamic and hepatic blood flow • Pregnancy is characterized by an increase in extracellular and plasma volume of 50% to 70%. • Red blood cell mass also increases, but the increase is moderate (20% to 30%) and delayed. • • As a consequence hemodilution occurs. • Cardiac output increases to a similar degree as the blood volume until the second trimester, and then decreases and normalizes near term. • Absolute hepatic blood flow remains unchanged, but the percentage of cardiac output to the liver decreases. This phenomenon of hemodilution should be considered during interpretation of all serum concentrations during pregnancy.

CHANGES IN LIVER FUNCTION Drug Metabolism Various hemodynamic changes during pregnancy, such as the increase in blood volume, cardiac output, and glomerular filtration rate, may contribute to altered drug metabolism, disposition, and clearance. Gastrointestinal absorption or bioavailability of drugs may vary because of changes in gastric secretion and motility. Drug properties such as lipid solubility, protein-binding characteristics, and ionization constant influence the placental passage of drugs. Pregnancy alters the ability of a drug to be distributed within the body, due to reduced concentrations of both albumin and α 1 -acid glycoprotein.

CHANGES IN LIVER FUNCTION Drug Metabolism Moreover, the increase in body weight in late pregnancy results in a decrease in dose per kilogram. Caffeine metabolism is reduced during pregnancy because of decreased activity of CYP 1 A 2. The activity of P-450 2 A 6 is increased and drugs such as nicotine exhibit substantially lower serum concentrations. The activity of CYP 3 A 4 is increased and drugs such as nifedipine, catbamazepine, midazolam, indinavir, lopinavir, and ritonavir, metoprolol, fluoxetine, citalopram, and nortriptyline may exhibit increased clearance.

CHANGES IN LIVER FUNCTION Drug Metabolism The dose of selective serotonin re-uptake inhibitors (SSRIs) must be increased to maintain efficacy in pregnancy. Furthermore, glomerular filtration rate is increased in pregnancy because of an increase in cardiac output; therefore drugs that are eliminated by renal mechanisms have increased clearance rates, such as ampicillin, cefuroxime, ceftazidime, cephradine, cefazolin, piperacillin, atenolol, sotalol, digoxin and lithium. Both estrogen intake and pregnancy impair hepatic activity of glucuronosyltransferase (UGT).
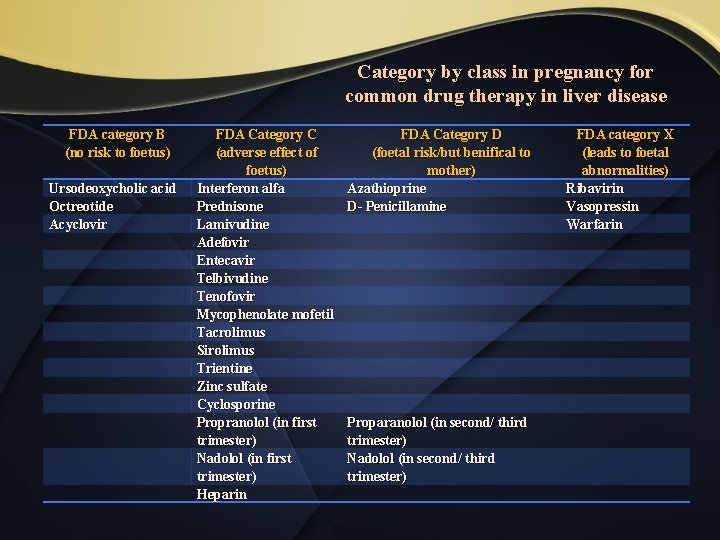
Category by class in pregnancy for common drug therapy in liver disease FDA category B (no risk to foetus) Ursodeoxycholic acid Octreotide Acyclovir FDA Category C (adverse effect of foetus) Interferon alfa Prednisone Lamivudine Adefovir Entecavir Telbivudine Tenofovir Mycophenolate mofetil Tacrolimus Sirolimus Trientine Zinc sulfate Cyclosporine Propranolol (in first trimester) Nadolol (in first trimester) Heparin FDA Category D (foetal risk/but benifical to mother) Azathioprine D- Penicillamine Proparanolol (in second/ third trimester) Nadolol (in second/ third trimester) FDA category X (leads to foetal abnormalities) Ribavirin Vasopressin Warfarin

SERUM PROTEINS AND LIPIDS A. Normally, up to 10 g of albumin is produced and secreted by the liver daily. Serum albumin concentrations decrease during the second trimester reaching concentrations approximately 70% to 80% of normal values at the time of delivery, secondary to hemodilution. By contrast, there is an increase in serum concentration of some proteins such as α 2 -macroglobulin, α 1 antritrypsin, and ceruloplasmin. B. Levels of fibronogen and most coagulation factors (II, VIII, IX and XII) increase, protein S levels decrease, and fibrinolysis is inhibited. These physiologic changes in hemostasis limit bleeding during delivery but are associated with an increased risk of thromboembolism during pregnancy and the postpartum period.

SERUM PROTEINS AND LIPIDS C. Prothrombin time (PT) is generally not affected by pregnancy and any change in the prothrombin time during pregnancy should be considered pathologic and warrants further investigation. D. Serum cholesterol, triglyceride, and phospholipid concentrations increase in late pregnancy by 25% to 50%, whereas concentrations of serum triglycerides increase twice to four times.

Changes in laboratory profile during normal pregnancy Test Change WBCs Increased Hemoglobin Decreased Platelets Albumin Dcreased Aminotransferase Alkaline phosphatase Increased GGT Bilirubin -/ Decreased Prothrombin time Fibrinogen Increased Globulins Increased in α- and β-globulins; decreased in γ-globulin Glucose Creatinine Uric acid Bile acid Cholesterol Increased Triglycerides Increased α-Fetoprotein Increased Ceruloplasmin Increased Ferritin Increased CGT, γ-glutamyltransferase, WBC, white blood cell

In pregnant women the total and free bilirubin concentrations are significantly lower during all three trimesters, due to hemodilution. HELLP syndrome has been associated with postpartum indirect hyperbilirubinemia and should be differentiated from Gilbert syndrome. Patients with Crigler-Najjar type I (CN 1) disorder the fetus is at high risk of being adversely affected by the bilirubin, because unconjugated bilirubin can cross the placenta and may cause kernicterus , a potentially neurotoxic condition. Successful pregnancy in patients with Crigler-Najjar syndrome has been reported with the use of phenobarbital and phototherapy. Dubin-Johnson syndrome (DJS), Pregnancy and use of oral contraceptives in women with Dubin-Johnson syndrome cause a reversible increase in serum conjugated bilirubin level. but pruritus and signs of generalized cholestasis are absent.

BILE ACIDS Pregnancy could be associated with subclinical cholestasis. Organic anion transport, including bilirubin and sulfobromophthalein (BSP), is impaired during pregnancy. This is due to that estrogen/pregnancy-induce decreases in the canalicular organic aniontransporting pump multidrug resistanceassociated protein-2 (MRP 2: ABC C 2). Concentrations of bile salts in blood are within the normal range in most pregnant women, but levels of glycocholate, taurocholate, and chenodeoxycholate may rise progressively until term and exceed levels measured early in pregnancy by two- to three-fold.

Pregnancy- or estrogen-induced decreases bilesalt transport due to Pregnancy could be associated with subclinical cholestasis. Organic anion transport, including bilirubin and sulfobromophthalein (BSP), is impaired during pregnancy. This is due to that estrogen/pregnancy-induce decreases in the canalicular organic aniontransporting pump multidrug resistanceassociated protein-2 (MRP 2: ABC C 2). Concentrations of bile salts in blood are within the normal range in most pregnant women, but levels of glycocholate, taurocholate, and chenodeoxycholate may rise progressively until term and exceed levels measured early in pregnancy by two- to three-fold.

Pregnancy- or estrogen-induced decreases bilesalt transport due to A. Reductions in both sinusoidal (Na+/ taurocholate co-transporting polypeptide [NTCP]. B. Reduction in canalicular (bile-salt export pump [BSEP]. C. Reduction in ATP-binding cassette [ABC]. In clinical practice, pruritus during pregnancy, measurement of serum bile acid concentration may be useful for the diagnosis of cholestasis, especially when routine liver function tests are still within normal limits.

CHANGES IN LIVER FUNCTION TEST VALUES A. Serum ALT and AST: activity levels remain within the normal limits established in nonpregnant women. Thus it should be emphasized that serum ALT and AST activity values above the upper limit of normal values before labor should be considered pathologic and should lead to further investigations. B. Serum alkaline phosphates activity levels increase in late pregnancy, mainly during the third trimester. This increase during pregnancy is not due to an increase in the hepatic isoenzyme but rather Largely attributable to the production of the placental and bone isoezymes. Thus measurement of serum alkaline phosphates activity is not a suitable test for the diagnosis of cholestasis during late pregnancy.

LIVER-RELATED SYMPTOMS AND PHYSICAL EXAMINATION IN PREGNANCY Nausea and vomiting are common symptoms of early pregnancy and occur in more than half of all pregnant women. Hypermesis gravidarum defined by severe vomiting beginning in early pregnancy and often requiring hospitalization, is much less frequent. Nausea or vomiting occurring during the second or third trimester should be considered pathologic and prompt investigation, including measurement of serum aminotrasferases activity. Vascular spiders and plamar erythema are commonly associated with chronic liver disease and pregnancy.

VIRAL HEPATITIS Viral hepatitis_ caused by hepatitides A, B, C, D, E , herpes simplex, cytomegalovirus, and Epstein – Barr virus. The clinical and serologic course of acute hepatitis in the western world is generally the same as that observed in the nonpregnant patient. Hepatitis E, which in the third trimester of pregnancy may lead to fulminant liver failure and may carry a high mortality (up to 31. 1%). Herpes simplex hepatitis is rare. The death rate is about 40%. Patients with herpes hepatitis present with severe or fulminant ”anicateric” hepatitis in the third trimester.
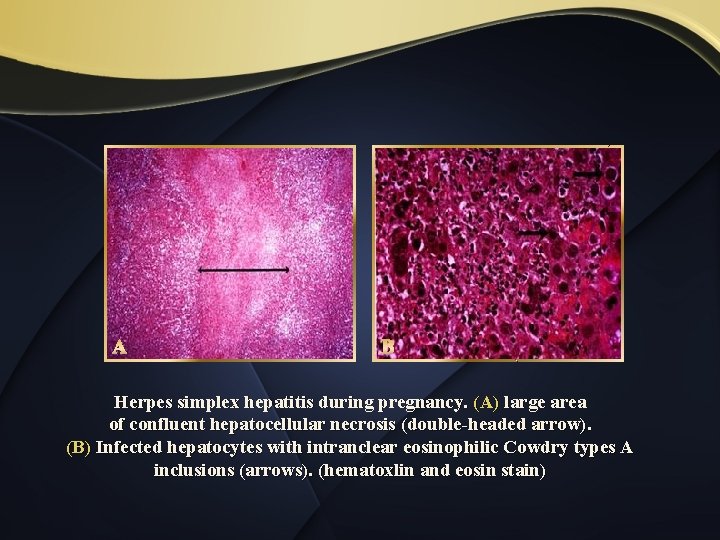
A B Herpes simplex hepatitis during pregnancy. (A) large area of confluent hepatocellular necrosis (double-headed arrow). (B) Infected hepatocytes with intranclear eosinophilic Cowdry types A inclusions (arrows). (hematoxlin and eosin stain)
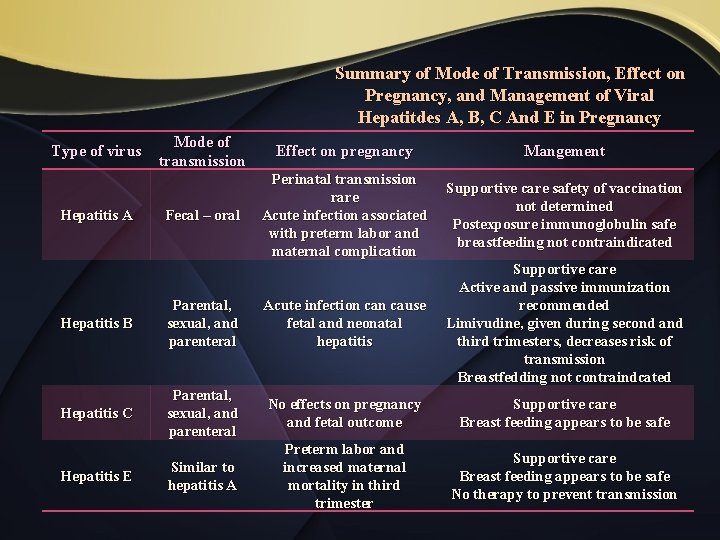
Summary of Mode of Transmission, Effect on Pregnancy, and Management of Viral Hepatitdes A, B, C And E in Pregnancy Type of virus Mode of transmission Effect on pregnancy Mangement Perinatal transmission Supportive care safety of vaccination rare not determined Acute infection associated Postexposure immunoglobulin safe with preterm labor and breastfeeding not contraindicated maternal complication Supportive care Active and passive immunization Acute infection cause recommended fetal and neonatal Limivudine, given during second and hepatitis third trimesters, decreases risk of transmission Breastfedding not contraindcated Hepatitis A Fecal – oral Hepatitis B Parental, sexual, and parenteral Hepatitis C Parental, sexual, and parenteral No effects on pregnancy and fetal outcome Supportive care Breast feeding appears to be safe Similar to hepatitis A Preterm labor and increased maternal mortality in third trimester Supportive care Breast feeding appears to be safe No therapy to prevent transmission Hepatitis E

HEPATITIS A The overall case/fatality ratio among reported cases is less than 1% and does not lead to chronic infection, although 10% to 15% of symptomatic individuals can have a prolonged or relapsing disease lasting up to months. If a pregnant woman becomes infected with hepatitis A, generally the baby is not affected. Intrauterine transmission of hepatitis A virus is very rare; however, prenatal transmission could occur. The management of acute HAV in pregnancy does not differ from that used in nonprenant patients. The safety of hepatitis A vaccination during pregnancy has not been determined. Mothers infected with hepatitis A virus are encouraged to breastfeed and HAV infection is not a contraindication for breastfeeding.

HEPATITIS B Hepatitis B virus has a high rate of vertical transmission causing fetal and neonatal hepatitis. Because it is highly pathogenic and infectious, perinatal transmission of HBV infection represents the single largest cause of chronically infected individuals worldwide. However, if the mother is seropositive for both HBs. Ag and hepatitis B eantigen (HBe. Ag), the frequency of vertical transmission increases to approximately 90% without neonatal prophylaxis. Therefore, the American Congress of obstetricians and gynecologists and the centers for disease control and prevention recommend universal screening for HBs. Ag in all pregnant women during the third trimester.

HEPATITIS B Pregnant women who are directly exposed to hepatitis B virus should receive a hepatitis B immunoglobulin (HBIG) injection ideally within 72 hours of exposure and then a hepatitis B vaccine within 7 days of exposure. HBV vaccines contain noninfectious HBs Ag and should cause no potential risk to the fetus. Hepatitis B vaccines administration to pregnant women is relatively safe and its benefits outweigh its risks. Hepatitis B vaccination can be delayed more than 24 hours after the baby's birth but should be given within the first week of delivery.

HEPATITIS B The combination of passive and active immunization is very effective the reducing the frequency of perinatal transmission of hepatitis B virus (85% to 95% efficacy). Interferon and oral nucleoside analogue that are classified as an FDA Category C drug in terms of safety, is given to mothers with high HBV DNA during the second and third trimesters to reduce the risk of transmission at the time of delivery.

HEPATITIS C All pregnant women should be screened for hepatitis C so that risk stratification can be performed and measures taken to both reduce perinatal transmission. Individuals who are HCV positive should have a PCR test fot HCV RNA as the risk of perinatal transmission is dependant on the presence of HCV RNA . liver function should be performed at the time of checking HCV RNA status as HIV co-infection increases the risk of transmission Risk of vertical transmission is increased with high viral load prolonged rupture of membranes and invasive procedures where possible fetal scalp electrodes and fetal scalp sampling should be avoided in women with HCV. Caesarean section is not recommended as a means of reducing perinatal transmission of hepatitis C.

HEPATITIS C As for blood borne infections it is recommended to bath the baby to remove any maternal body secretions and blood prior to IM injections. HCV infection is not a contraindication to breastfeeding except in the presence of craked or bleeding nipples. In this instance expression and discarding of the milk is adviced whilist waiting for healing of the cracked nipples. All infants of HCV positive mothers should be screened following delivery to determine they have been infected care should be taken to ensure the appropriate interval passed for the neoborn to become pcr +- antibody positive.

HEPATITIS C Given that antiviral curative treatment for hepatitis c is now readily avilable consideration should be given to screeining all women prior to pregnancy so that they are able to make an informed choice regarding treatment prior to embarking on pregnancy , exiting treatments for hcv are not recommended during pregnancy of breastfeeding in particular ribavrin is teratogenic. For all women and male parterns recieving ribavrin reliable contraceptoion must be used during treatment and for 6 months after completion of treatment.

HEPATITIS E HEV infection is known to cause severe hepatitis, fulminant liver failure, preterm labor, and increased mortality in pregnant women, especially in their third trimester with reported maternal death rates as high as 20% to 31. 1%. The mechanisms of fulminant hepatitis E in pregnancy is not clear but is thought to be attributable to induction of type 2(TH 2) cytokines. At the present time, it appears safe to continue breastfeeding during epidemics of HEV in underdeveloped and endemic areas to prevent a greater risk of infant mortality from other infectious diseases.

AUTOIMMUNE HEPATITIS Pregnant women with AIH have a reduced fertility rate secondary to amenorrhea and anovulation, which might be related to hypothalamicpituitary dysfunction. However, AIH has been reported to occur de novo during pregnancy or the postpartum period. Because of improved management of AIH patients and reduced hepatocellular injury from use of immunosuppression therapy, menstruation may ensue and more women with AIH are able to conceive. In a retrospective study the rate of serious maternal complications was 9% and a high rate (52%) of postpartum exacerbation was noted. The rate of adverse pregnancy outcome was 26%, which was highly associated with the presence of antibodies to SLA/LP and Ro/ SSA.

AUTOIMMUNE HEPATITIS These data and other illustrate that pregnancy can ameliorate autoimmune hepatitis, whereas delivery can exacerbate it. Immunosuppressive therapy with azathioprine (AZA) and corticosteroid therapy induces clinical, laboratory, and histologic improvements in 80% of patients with autoimmune hepatitis, but most women with AIH require maintenance immunosuppression therapy. At this time successful completion of pregnancy is a realistic expectation for patients with well-controlled AIH. Corticosteroids and azathioprine are generally safe during pregnancy but birth defects have been described. Pregnant women with AIH need careful monitoring during pregnancy and for several months postpartum.

WILSON DISEASE Wilson disease is a rare autosomal recessive disorder of hepatic copper transport leading to inhibition of biliary copper excretion. This causes an increase in copper deposition in vital organs such as liver, kidney, brain, and eyes. Two copper transporting ATPases, Menkes (AT 7 A; MNK) and Wilson (ATP 7 B; WND), are expressed in the placenta and both are involved in placental copper transport. Pregnancy does not seem to have an adverse effect on the clinical course of Wilson disease although recurrent abortions are common in untreated patients, which can be seen in 26% of cases.

WILSON DISEASE Indeed, untreated symptomatic women with Wilson disease tend to suffer amenorrhea, oligomenorrhea, irregular menses, and multiple miscarriages, However, pregnancy in women with Wilson disease is safe and successful when treatment with a chelating durg is continued uninterrupted. With the current available copper chelators such as D-penicillamine, trientine, and Zinc, fertile women are able to conceive. Pencillamine and trientine have teratogenic effects in animals, and penicillamine has known teratogenic effects in humans as well. D-Penicillamine probably inhibits thyroperoxidase activity in utero.

WILSON DISEASE Infants born to mothers with Wilson disease may develop transient goitrous hypothyroidism. Zinc intake at a dose of 25 to 50 mg three times daily in pregnancy appears to be safe with very minimal teratogenicity. It is important that treatment of Wilson disease with anticopper agent continues during pregnancy without interruption. Both mother and baby should be monitored while on chelation therapy.

PORTAL VEIN THROMBOSIS Portal vein thrombosis (PVT) is a rare occurrence during pregnancy. PVT is caused by a combination of local and systemic risk factors. Causes: Malignant tumors, cirrhosis, intrabdominal inflammation (e. g. , pancreatitis, appendicitis, cholecystitis, duodenal ulcer, inflammatory bowel disease) Additionally, systemic prothrombotic risk factors such as myeloproliferative disorders; antiphosholipid syndrome; protein C, protein S, and antihrombin deficiency; and factor V Leiden, factor II, and methylenetrahydrofolate reductase gene mutations are other potential risk factor for PVT and are responsible for 60% to 70% of cases.

CLINICAL MANIFESTATIONS Acute PVT usually presents with abdominal or lumbar pain that either is sudden in onset or progresses over a few days. Partial thrombosis of the portal vien is associated with fewer symptoms. Rapid and complete obstruction of the portal or mesenteric venous arches, induces intestinal congestion that is manifested by severe and continuous colicky abdominal pain with occasional nonbloody diarrhea. Acute septic PVT referred to as acute pyleophlebitis is characterized by infected thrombus. The presentation is spiking fever and chills, a tender liver, and occasionally shock, persistence of severe pain beyond 5 to 7 days, bloody diarrhea, and ascites along with features of acidosis and renal or respiratory dysfunction are suggestive of intestinal infarction.

LABORATORY AND IMAGING FEATURES Liver function is persevered in both acute and chronic PVT because increased hepatic arterial blood flow compensates for the decreased portal inflow. Abdominal US with Doppler studies of the portal veins reveals absent or sluggish flow. Persistent pain, presence of ascites, or development of multiorgan failure indicates that intestinal infarction is likely and surgical exploration should be considered. A tumor-like cavernoma, a rare form of chronic PVT, is characterized by tiny collateral channels forming a mass that encases the main bile duct and can be confused with carcinoma of the main bile duct

TREATMENT ACUTE (PVT) 1. In the presence of pyelophlebitis, antibiotics should be administered. 2. Anticoagulation medication should be administered for at least 3 month and permanent anticoagulation therapy should be considered for patients with prothormbotic conditions. 3. Surgical thrombectomy, systemic or in situ thrombolysis, and transjugular intrahepatic portosystemic shunt (TIPS) are extremely limited. Chronic (PVT)

TREATMENT ACUTE (PVT) Pregnant patients with chronic PVT should be offered screening for gastroesophageal varices. Anticoagulation therapy should not be initiated until after adequate prophylaxis for varical bleeding. Additionally, long-term anticoagulation therapy should be considered only in patients with chronic PVT, without cirrhosis, and with a permanent risk factor for venous thrombosis.

BUDD-CHIARI SYNDROME Clinical and Laboratory features Presentation can range from complete absence of symptoms to fulminant hepatic failure. This syndrome usually present in the last trimester or the puerperium. The characteristic clinical triad of acute BCS is right upper quadrant pain, hepatomegaly, and ascites.

BUDD-CHIARI SYNDROME Clinical and Laboratory features Chronic thrombosis typically evolves slowly with dull abdominal pain, lower extremity edema, gastrointestinal bleeding, and hepatic encephalopathy. Jaundice is relatively uncommon. Marked dilation of subcutaneous veins on the trunk has a high specificity but a low sensitivity for IVC obstruction. Levels of serum aminotransferases and alkaline phosphatase can be normal or increased. Levels of serum albumin, serum bilirubin, and prothombin can be normal or abnormal, and in some patients are markedly abnormal. The protein level in ascetic fluid varies from patient to patient. Ascites protein content is greater than 3. 0 gl/dl, and serum ascites albumin gradient (SAAG) greater than 1. 1 is generally suggestive of portal hypertension.

IMAGING FEATURES Diagnostic modalities including right upper quadrant abdominal US with Doppler studies of the hepatic veins have the advantage of being noninvasive or minimally invasive.

TREATMENT 1. Women with history of BCS who wishes to become pregnant should be screened for portal hypertension by upper endoscopy and, when indicated, prophylaxis of varices. 2. Non pregnant should recieve long-term warfarin therapy. 3. Pregnant patients should recieve low-molecular-weight heparin (LMWH) as soon as possible, preferably before conception. It should be given to twice-daily dosing of LMWH. Delivery should be scheduled 24 hours after the last therapeutic dose and restarted 12 hours after vaginal delivery or 24 hours after cesarean section either with LMWH or with warfarin. 4. BCS diagnosed during pregnancy, Doppler US should be used as the first modality.

TREATMENT Warfarin (FDA category X) is contraindicated. LMWH at a curative dose should be started as soon as the diagnosis is established. Pharmacologic and endoscopic therapy for portal hypertension can be applied. Angioplasty and TIPS insertion have been reported for refractory varicel bleeding.

HEREDIATRY HEMORRHAGIC TELANGIECTASIA Characaterized by widespread cutaneous, mucosal, and visceral arteriovenous malformations that can involve the lund, brain, and/or liver. Liver vascular malformations include both microscopic and macroscopic malformation of variable size, ranging from tiny telangicetasis to discrete arteriovenous malformation. The majority of cases are asymptomatic.

The three most common initial clinical presentations 1. High-output heart failure. (shortness of breath, dyspnea on exertion, ascites, and/or edema) 2. Portal hypertension. 3. Biliary ischemia. The diagnosis of HHT by Doppler US Treatment In patients with symptomatic HHT, treatment is directed toward the clinical manifestation of heart failure and portal hypertension.

CIRRHOSIS AND PORTAL HYPERTENSION During pregnancy a hypervolemic state develops, leading to an increase in portal flow and elevation of portal pressure transmitted to the collateral veins with increased risk of variceal bleeding. PHT occurs during the last stages of the second trimester of pregnancy and is associated with increased risk of varical bleeding in the later stages of pregnancy. Esophageal variceal bleeding has been reported in 18% to 32% of pregnant women with cirrhosis and in up to 50% of those with known portal hypertension. Of those with preexisting varices, 78% will have gastrointestinal bleeding during pregnancy, with a mortality of 18% to 50%.

CIRRHOSIS AND PORTAL HYPERTENSION Pregnant patients with cirrhosis face unique risks that include spontaneous abortion, prematurity, pulmonary hypertension, splenic artery aneurysm rupture, and postpartum hemorrhage, and a potential for life-threatening variceal hemorrhage and heptic decompensation. Endoscopic surveillance and banding of esophageal varices is recommended during pregnancy. On the basis of the endoscopic finding, primary prophylaxis with nonselective β-blockers such as propranolol and/or nadolol (designated by FDA as Pregnancy Category C) is recommended. The risks of nonselective β-blockers include fetal bradycardia, hypotension, hypoglycemia, and intrauterine growth retardation.

CIRRHOSIS AND PORTAL HYPERTENSION Endoscopic banding ligation seems to be a safe procedure in pregnancy. When bleeding is not arrested endoscopically, an emergency TIPS procedure should be considered. Ascites rarely occurs in pregnant women with cirrhosis and the mainstay of treatment is sodium restriction and the use of diuretics. Hepatic encephalopathy In the setting of cirrhosis, secondary to medications, Sepsis, hypoxia, gastrointestinal bleeding, and hypotension. Postpartum uterine hemorrhage occurs in 7% to 10% of pregnancies in patients with cirrhosis and represents a potential source of maternal morbidity and mortality.

CIRRHOSIS AND PORTAL HYPERTENSION Vaginal delivery is usually safe and early assistance with forceps delivery or vacuum extraction should be considered to prevent further rise in portal pressure secondary to prolonged straining during labor. Termination of pregnancy is warranted in the presence of progressive hepatic decompensation.

GALLSTONES AND BILIARY TRACT DISEASE There are several changes that occur during the course of pregnancy that lead to the formation of biliary sludge and gallstone. 1. Decline in the contractility of the gallbladder. This is most likely caused by hormonal change during pregnancy, with progesterone having the greatest effect. 2. Alteration in the content of bile. Compared with bile acids and phospholipids, cholesterol secretion increase in the second and third trimesters, leading to lithogenic. Despite their prevalence in 5% to 12% of pregnant women, symptomatic gallstones occur in only 0. 1% to 0. 3% of pregnancies.

TREATMENT 1. Cholecystectomy during pregnancy is not associated with a high rate of fetal demise. Laparoscopic cholecystectomy is probably a safe option. during the second trimester of pregnancy. 2. Endoscopic management of biliary tract disease using endoscopic retrograde cholangiopancreatography (ERCP) is also a viable option.

LIVER DISEASE UNIQUE TO PREGNANCY Four unique disorders of liver dysfunction have been recognized during pregnancy. These include 1. Hepatic involvement in HG. 2. ICP. 3. HELLP syndrome. 4. AFLP.

HEPATIC INVOLVEMENT IN HEPEREMESIS GRAVIDARUM HG is a severe and persistent form of NVP that predominantly affects pregnant women in their first trimester and is characterized by intractable vomiting that leads to loss of 5% or more of pre-pregnant body weight and dehydration. HG can be associated with dehydration, imbalanced electrolyte levels, ketonuria, abnormal levels of liver enzymes, low birth weight, avitaminosis, and Wernicke's encephalopathy; rarely, it can be fatal.

Etiology and pathophysiology: The underlying mechanism of maternal liver disease associated with hyperemesis gravidarum is not clear. Risk Factors: Women with previous history of hyperemesis gravidarum, previous molar pregnancy, preexisting diabetes, gastrointestinal disorders, asthma, singleton female pregnancies, pregnancies with multiple male fetuses, multiple gestations, hyperthyroid disorders, psychiatric illness, and low prepregnancy body weight are at increased risk. The relationship between infection with Helicobacter pylori and HG is not clear. Pathogenesis of liver injury in HG is not clear. Intense vomiting leads to dehydration, ketonuria, starvation, and malnutrition, may result in depletion of glycogen stores and an increase in mitochondrial injury caused by oxidative stress

DIAGNOSIS 1. Intense nausea and vomiting in the first trimester associated with a 5% or greater weight loss, dehydration, ketonuria, electrolyte imbalance, and metabolic alkalosis is highly suggestive of hyperemesis gravidarum. 2. Clinically; fever, abdominal pain, headache, elevation in white blood cell count, anemia, thrombocytopenia, and coagulopathy should elude to nausea and vomiting of other causes. 3. Mild elevation of levels of liver transaminases (usually <300 U/L), total bilirubin level is generally less than 4 mg/dl.

MANAGEMENT Nutritional support, hydration, and control of emesis are the mainstay of therapy. Ginger has been shown to be effective in reducing nausea and vomiting. Metoclopramide and vitamin B 6 appear to be safe and effective. Thiamine supplementation for women with prolonged vomiting is recommended to prevent Wernicke-Korsakoff syndrome. Nerve stimulation therapy is effective in reducing nausea and vomiting and in promoting weight gain in symptomatic women in the first trimester of pregnancy. Low –dose prednisolone has been shown to have a similar effect as promethazine in reducing nausea and vomiting with fewer side effects.

INTRAHEPATIC CHOLESTASIS OF PREGNANCY Characterized by; Pruritus, elevated levels of serum bile acids. Although most affected pregnant women have a mild form of the disease, the disease can be associated with meconium staining in 45%, spontaneous preterm labor in 44%, and intrapartum fetal distress in 22% of pregnancies, as well as low birth weight. Etiology • The etiology of ICP is still unknown.

Possible mechanisms A. Genetic: • Occurrence within familial clustering and endemic ethnic groups. • Heterozygous nonsense mutation of the multidrug resistanceassociated protein-3 (MDR 3) gene. B. Hormonal: High levels of maternal sex hormones in a susceptible patient may lead to ICP. Hypersensitivity to estrogen and estrogen-sulfated metabolities is considered to be the main precipitant.

Pathophysiology The exact gene defect in bile acid secretion has not been defined. Low serum selenium concentration and lower levels of serum placental protein 10 (PP 10) have also been noted in ICP patients. Diagnosis A)Clinically: Generalized intractable pruritus in the second or third trimester of pregnancy with elevated levels of maternal serum bile acids in the absence of radiologic evidence of biliary obstruction. Pruitus is severe and tends to follow an ascending pattern of distribution. Initially it involves the palms of the hands and the soles of the feet and subsequently the arms, legs, and trunk are involved. Pruritus tends to abate after resolution of gestation but to occure in subsequent pregnancies.

B) Lab: Elevated levels of maternal serum bile acids are the hallmark of cholestasis and are usually 10µmol/L or greater. A cutoff level of serum bile acids of 40µmol/L or greater is associated with impaired fetal outcome. Serum aminotransferase levels are usually elevated but less than 1000 U/L. Levels of cholestatic enzymes, such as alkaline phosphatase, are also elevated; however, use of alkaline phosphatase level as a single indicator of the disease is not recommended because it is produced by both placenta and bone. Serum γ-glutamyltranspeptidase (GGT) level is normal or slightly elevated.

Intrahepatic cholestasis of pregnancy showing canaliuclar bile plugs (arrows) with well preserved hepatocytes containing yellow (hematoxyli and eosin (H& E) stain)

MANAGEMENT Strict felal monitoring must be carried out, especially during the last few weeks of pregnancy when rates of fetal morbidity and mortality are increased. Ursodeoxycholic acid (UDCA). The recommended dose is 15 mg/kg/day.

SYNDROME OF HEMOLYSIS, ELEVATED LIVER ENZMES, AND LOW PLATELET COUNT Precclampsia is defined as the presence of edema, proteinuria, and hypertension with a blood pressure greater than 140/90 mm Hg in a previously normotensive patient. The exact cause remains unknown and the definitive treatment is still delivery. The liver involvement in preeclampsia may manifest as HELLP syndrome. The incidence of HELLP syndrome is 3. 1 % to 12% of patients with preeclampasia. HELLP syndrome can be life-threatening to both the mother and fetus. The associated liver disease may progress to the point that liver transplantation may become necessary. HELLP syndrome occurs in the antepartum period in 70% to 92 % of cases and during the postpartum period in 8% to 30% of patients.

PATHOGENESIS A. One theory emphasizes structural and functional changes in the systemic vasculature as a major role in the development of HELLP syndrome. B. Another theory is that placenta-derived proteins mediate apoptosis of liver cells. In animal models interaction of placenta-derived CD 95 (APO 1, Fas) with its ligand, CD 95 L (Fas. L), induce apoptosis in hepatic cells, A new therapeutic agent (LY 498919) that blocks CD 95 -induced apoptosis is under investigation.

CLINICAL FINDINGS A. Patients are typically seen in the early third trimester with nonspecific symptoms including right upper quadrant pain, malaise, nausea, and vomiting. B. Concomitant signs and symptoms of preeclampsia (hypertension, proteinuria, and edema) may or may not be present. C. Jaundice, bleeding attributable to thrombocytopenia are a very uncommon mode of presentation. D. Severe right upper quadrant abdominal pain that is radiated to the neck or shoulder may herald impending hepatic rupture or the presence of hepatic hematoma.

DIAGNOSIS Two major diagnostic classification systems are currently used for the classification of HELLP syndrome.

MAIN DIAGNOSTIC CRITERIA OF HELLP SYNDROME Tennessee classification Complete syndrome: Platelets ≤ 100 × 109/L AST ≥ 70 units/L LDH ≥ 600 units/L Mississippi classification Class 1: Platelets ≤ 50 × 109/L AST or ALT ≥ 70 units/L LDH ≥ 600 units/L Class 2: Platelets ≤ 100 × 109/L ≥ 50 × 109/ AST or ALT ≥ 70 units/L Incomplete syndrome any one or LDH ≥ 600 units/L Class 3: Platelets ≤ 150 × 109/L two of above ≥ 100 × 109/ AST or ALT ≥ 40 units/L LDH ≥ 600 units/L
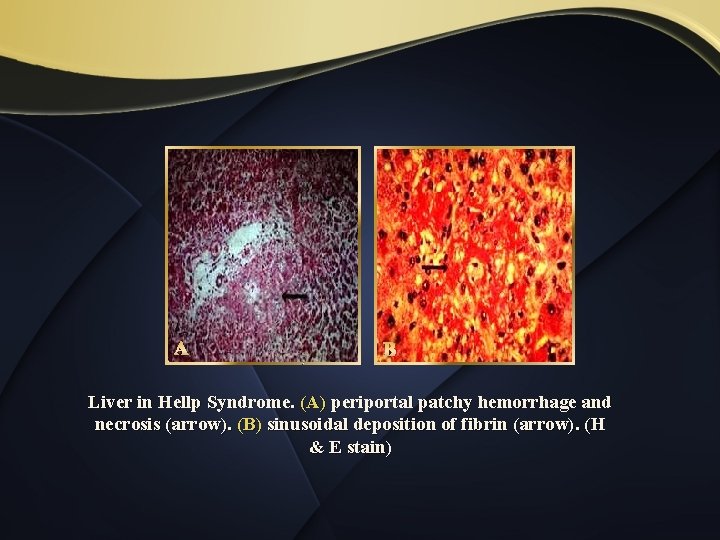
A B Liver in Hellp Syndrome. (A) periportal patchy hemorrhage and necrosis (arrow). (B) sinusoidal deposition of fibrin (arrow). (H & E stain)

ASSESSMENT OF THE SEVERITY OF THE PATHOLOGIC PROCESS Class 1 HELLP syndrome having a worse prognosis and longer hospital stay than either class 2 or class 3. Both HELLP syndrome and acute fatty liver of pregnancy occur in the third trimester and have similar presentations, but liver dysfunction is usually more pronounced in the latter and is more frequently associated with Coagulopathy, hypoglycemia, and renal failure. The coagulopathy of AFLP is due to liver failure, whereas in HELLP syndrome coagulopathy develops as a part of disseminated intravascular coagulation (DIC) syndrome.

MANAGEMENT Because of significant morbidity and mortality, HELLP syndrome is considered an indication for immediate delivery. A)Prompt Delivery: • Gestation is greater than 34 weeks. • Fetal testing results are nonreassuring. • Any of the following conditions are present: • Maternal multiorgan dysfunction, DIC, liver infarction or hematoma, renal failure, or abruption placentae.

MANAGEMENT B) Seizure prophylaxis with magnesium sulfate and blood pressure control is also required C)Corticosteroids D)Platelet transfusion is recommended in the case of bleeding or severe thrombocytopenia (platelet count <20 × 109/L) Prognosis: After delivery, initial worsening of laboratory values with subsequent spontaneous recovery is observed.

COMPLICATION OF HELLP SYNDROME A. Serious maternal complications are common, including DIX, abrupt placentae, acute renal failure, eclampsia, pulmonary edema, acute respiratory distress syndrome, ascites, subcapsular hematoma, hepatic failure, and wound hematomas. B. Indications to proceed with liver transplantation include persistent bleeding form a hematoma or hepatic rupture or liver failure form extensive necrosis. C. Liver rupture is a rare, life-threatening complication of HELLP syndrome that carries a very high maternal mortality. It is usually preceded by an intraparenchymal hemorrhage progressing to a contained subcapsular hematoma in the right hepatic lobe in patients with severe thrombocytopenia.
COMPLICATION OF HELLP SYNDROME Abdominal CT scan may be the most sensitive and specific way to detect hepatic hemorrhage and/or rupture. MANAGEMENT OF LIVER RUPTURE Immediate laparotomy and evacuation of the hematoma with pressure packing and drainage, followed by consideration of hepatic artery embolization or ligation, partial hepatectomy, or oversewing of the laceration. In the rare hemodynamicaly stable patient, angiographic embolization may be considered.

RECURRENCE OF HELLP SYNDROME HELLP syndrome can recur in subsequent pregnancies. Subsequent pregnancies in patients with HELLP syndrome carry a high risk of complications such as preeclampsia, recurrent HELLP, prematurity, intrauterine growth retardation, abruption placentae, and perinatal mortality
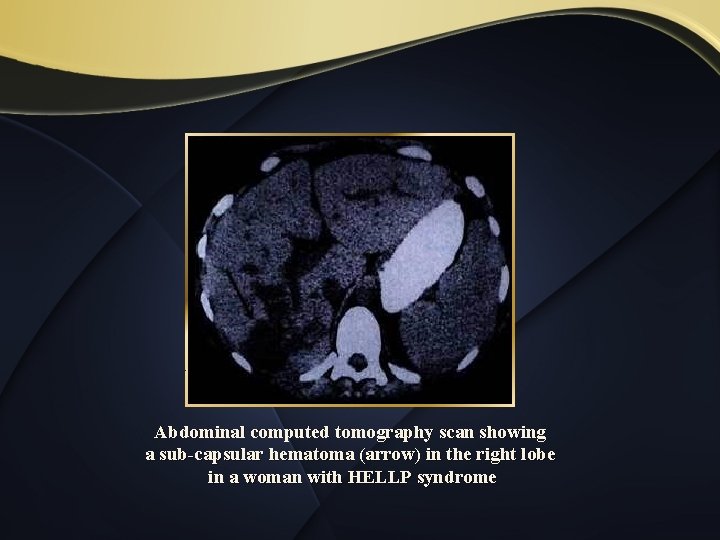
Abdominal computed tomography scan showing a sub-capsular hematoma (arrow) in the right lobe in a woman with HELLP syndrome

ACUTE FATTY LIVER OF PREGNANCY Acute fatty liver of pregnancy is a clinical entity unique to pregnancy that occurs during the third trimester. Although it is a rare condition, it carries significant perinatal and maternal mortality. AFLP is more common in primiparous women and generally occurs between 30 and 38 weeks of gestation. Multiple gestations and gestations with male fetuses results in generally higher prevalence of AFLP when compared with the average population. The disease tends to recur in future pregnancies.

PATHOGENESIS Unknown, Recent molecular advances suggest that AFLP may result from mitochondrial dysfunction. Several reports have documented a strong association between AFLP and a deficiency of the enzyme long-chain 3 -hydroxyacyl-Co. A dehydrogenase (LCHAD) in the fetus, a disorder of mitochondrial fatty acid β-oxidation.

CLINICAL FINDINGS Acute fatty liver of pregnancy usually occurs most commonly in the third trimester with a few case reports in the second trimester. Nonspecific with malaise, nausea, vomiting, and headache and can be easily misdiagnosed. Right upper quadrant pain and epigastric abdominal pain. Fever, headache, diarrhea, back pain suggestive of acute pancreatitis. Rarely patients may present with frank liver failure and bleeding attributable to liver failure-induced coagulopathy, Preeclampsia co-exists in >50% of patients with AFLP.

CLINICAL FINDINGS Occasionally, the patient may present with signs and symptoms of eclampsia (agitation, increased thirst, premature labor, seizures). Hypertension is mild or may absent because of a decrease in peripheral vascular resistance associated with hepatic failure. Rarely AFLP may present as asymptomatic elevation of transaminase levels. Jaundice is seen in initial presentation in severe cases.

PHYSICAL FINDINGS Right upper quadrant tenderness may be the only abnormality found. The liver is usually non-palpable. As the disease progresses, jaundice, altered mental status, ascites, and edema arise

COMPLICATIONS Acute renal failure. Acute pancreatitis. Hypoglycemia, and infection. Hepatic encephalopathy. Any combination of these complications may lead to significant maternal and fetal mortality. Delivery is often complicated with severe postpartum bleeding. Diabetes insipidus may also complicate AFLP.

DIAGNOSIS OF AFLP A moderate elevation in the levels of transaminases is usually seen, ranging from just slightly above normal to a value more than 1000 U/L above normal. Normocytic anemia, leukocytosis and thrombocytopenia is common when DIC is present. Elevation of blood urea nitrogen (BUN) and creatinine. As liver function progressively worsens, encephalopathy, hypoglycemia, and elevated ammonia concentration.

Liver Biopsy show cytoplasmic vacuolization predominantly in the perivenular and midzonal regions with microvesicular fatty infiltration. In severe cases lobular disarray is seen with cell dropout and fatty change may be diffuse and involve all zones. Portal inflammatory changes can be seen as well, suggestive of cholangitis. Imaging studies are of little value in the diagnosis of AFLP. They are mainly useful for excluding other pathologic processes in the liver (e. g. , hepatic ischemia, hepatic infarct, Budd-Chiari syndrome, or hepatic hematoma/rupture). Liver US and CT were inconsistent in detecting fatty infiltration of the liver in patients with AFLP; the role of MRI and spectroscopy in particular requires further investigation.

DIFFERENTIAL DIAGNOSIS 1)AFLP AND HELLP SYNDROME Both conditions present late in pregnancy and both can be associated or complicated with preeclampsia. Hypoglycemia and/or prolongation of prothrombin time is suggestive of AFLP. Histopathology shows extensive hepatocellular necrosis, in HELLP syndrome, which is absent in AFLP, and microvesicular fatty infiltration, the predominant feature of AFLP, is generally absent in HELLP syndrome

DIFFERENTIAL DIAGNOSIS 2)AFLP AND ACUTE HEPATITIS A history of exposure and viral serologic testing are the key in making the diagnosis. The degree of elevation of liver transaminase levels is much higher in patients with acute viral hepatitis than in AFLP

MANAGEMENT Patients are usually very ill and require hospitalization in the intensive care unit. Timely delivery after initial patient stabilization will cure the patient

A B C Acute fatty liver of pregnancy. (A) Fat accumulation is greater in pericentral hepatocytes (arrow) compared with periportal hepatocytes (arrowhead). (H & E stain. ) (B) The small (microvesicular fat droplets surround but do not displace hepatocyte nuc; ei (arrows). (H & E stain. ) (C) Fat is readily appreciated with oilred O stain on frozen liver tissue
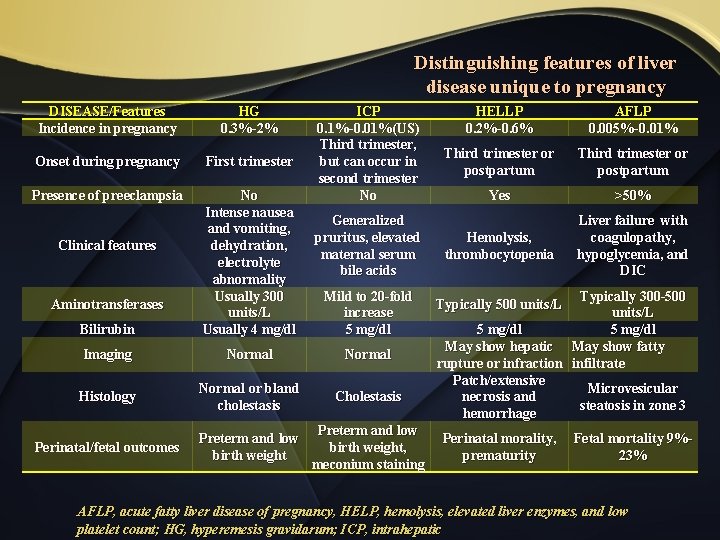
Distinguishing features of liver disease unique to pregnancy DISEASE/Features Incidence in pregnancy HG 0. 3%-2% Onset during pregnancy First trimester Presence of preeclampsia Bilirubin No Intense nausea and vomiting, dehydration, electrolyte abnormality Usually 300 units/L Usually 4 mg/dl Imaging Normal Histology Normal or bland cholestasis Cholestasis Perinatal/fetal outcomes Preterm and low birth weight, meconium staining Clinical features Aminotransferases ICP 0. 1%-0. 01%(US) Third trimester, but can occur in second trimester No Generalized pruritus, elevated maternal serum bile acids Mild to 20 -fold increase 5 mg/dl HELLP 0. 2%-0. 6% AFLP 0. 005%-0. 01% Third trimester or postpartum Yes >50% Hemolysis, thrombocytopenia Liver failure with coagulopathy, hypoglycemia, and DIC Typically 300 -500 units/L 5 mg/dl May show hepatic May show fatty rupture or infraction infiltrate Patch/extensive Microvesicular necrosis and steatosis in zone 3 hemorrhage Typically 500 units/L Perinatal morality, Fetal mortality 9%prematurity 23% AFLP, acute fatty liver disease of pregnancy, HELP, hemolysis, elevated liver enzymes, and low platelet count; HG, hyperemesis gravidarum; ICP, intrahepatic

- Slides: 91