Liver Function Testing Liver Multitude of processes Metabolism

Liver Function Testing

Liver • Multitude of processes: • Metabolism of carbohydrates- converts glucose to glycogen (glycogenesis). Uses enzymes to convert glycogen back to glucose for energy (glycogenolysis) • Storage- stores glycogen; temporarily small amounts of protein and lipid, Vitamins(A, D, B 12), Iron • Excretory- produces and excretes bile; bile then stored in gallbladder • Protective- destroys foreign objects and detoxifying poisons; contains phagocytic cells know as Kupffer cells that remove foreign materials, such as bacteria, from the blood; takes toxic chemicals and converts them into less toxic forms that are water-soluble and can be excreted by the kidneys (ie. Ammonia conversion) • Circulatory-serves as blood storage area and helps to regulate blood volume • Blood coagulation- synthesizes protein-clotting factors

Liver function panel • ALB • TBILI • DBILI • AST • ALP • Additional enzyme testing: GGT, LDH

Specimen • No hemolysis-falsely decrease • Light sensitive-conjugated and unconjugated bilirubin

Normal ranges • ALT: 7 -45 U/L • AST: 5 -35 U/L • ALP • Males/females: 4 -15 yrs: 54 -369 U/L • Males 20 -50: 53 -128 U/L, >60: 56 -119 U/L • Females 20 -50 y: 42 -98 U/L, > 60: 53 -141 U/L • • • ALB: 3. 5 -5. 0 g/d. L TBILI: 0. 2 -1. 0 mg/d. L DBILI: <0. 8 mg/d. L IBILI: <0. 2 mg/d. L GGT: male: 6 -55 U/L • Female: 5 -38 U/L • LDH: 125 -220 U/L

Bilirubin analysis • Based on Diazo reaction- Diazotized sulfanilic acid (diazo reagent) reacts with bilirubin to form a reddish-purple color at neural p. H and blue color at high or low p. H ranges. 1916 -discovered the addition of alcohol would speed up the reaction of diazo reagent and unconjugated bilirurubin. DBIL fraction-reaction that took place without alcohol. Addition of alcohol achieved a total bilirubin result. Subtract direct from total and would get indirect fraction

Direct Bilirubin analysis • Jendrassik-Grof method • • Measures conjugated and total bilirubin Direct bilirubin reacts with diazo reagent Incubated Add ascorbic acid Alkaline tartrate and caffeine added Formation of blue-green azobilirubin Measured at 600 nm

Total bilirubin analysis • Pretreat specimen with caffeine-sodium benzoate • HCL and diazo reagent added • Ascorbic acid added • Alkaline tartrate added=blue-green azobilirubin

Indirect bilirubin • Calculated: TBILI – DBILI = IBILI

Aspartate aminotranserase- pointe analysis • L-Aspartate + a-Ketogluterate ---AST---- > Oxalacetate + L-glutamate Oxalacetate + NADH + H+ -----MDH---- > L-Malate + NAD + H 2 O AST catalyzes the transfer of the amino group from L-aspartate to aketoglutarate to yield oxalacetate and L-glutamate. The oxaloacetate undergoes reduction with simultaneous oxidation of NADH to NAD in the MDH catalyzed indicator reaction. The resulting rate of decrease in absorbance at 340 nm is directly proportional to the AST activity

Alanine aminotransferase- pointe analysis • L-alanine + a-ketoglutarate -----ALT------ > pyruvate + L-glutamate Pyruvate + NADH + H -----LDH---- > L-Lactate + NAD + H 2 O ALT catalyzes the transfer of the amino group from L-alanine to aketoglutarate resulting in the formation of pyruvate and L-glutamate. Lactate dehydrogenase catalyzes the reduction of pyruvate and the simultaneous oxidation of NADH to NAD. The resulting rate of decrease in absorbance is directly proportional to ALT activity.

Alkaline phosphatase-pointe analysis p-NPP + H 2 O ----Alk Phos------ > p-Nitrophenol + H 3 PO 4 p-Nitrophenyl phosphate is hydrolyzed to p-Nitrophenol and inorganic phosphate. The rate at which the p-NPP is hydrolyzed, measured at 405 nm, is directly proportional to the alkaline phosphatase activity.

Albumin- pointe analysis • Albumin is bound by the bromocresal green dye producing an increase in the blue-green color measured at 630 nm. The color increase is proportional to the concentration of albumin present

Total protein-Pointe analysis • Biuret color reaction: color reaction of protein molecules with cupric ions. • Pointe analysis uses modifications of Biuret principle. Protein + Cu++ -----Alkali---- > Colored complex Protein in serum forms a violet colored complex when reacted with cupric ions in an alkaline solution. The intensity of the violet color is proportional to the amount of protein present when compared to a solution with known protein concentration.

CLS 242: Clinical Chemistry II Liver Function
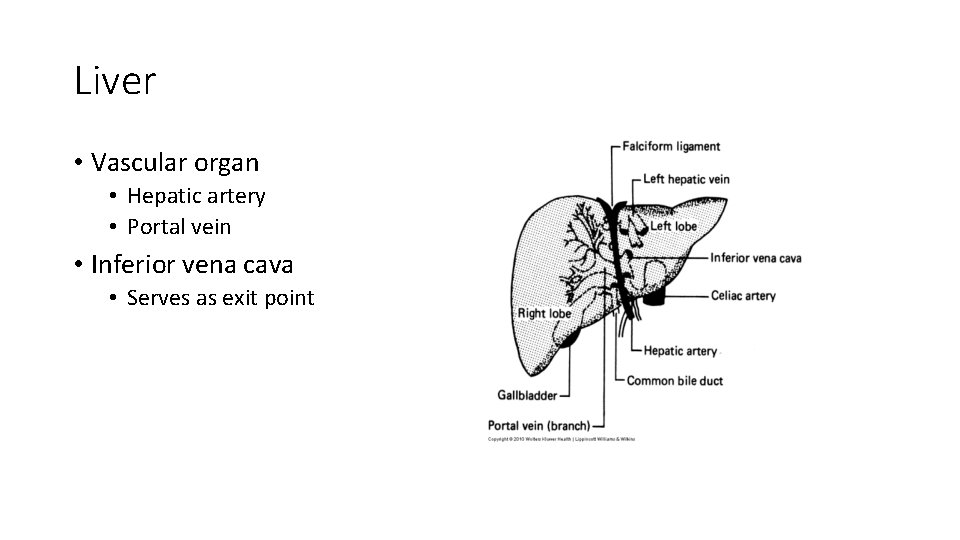
Liver • Vascular organ • Hepatic artery • Portal vein • Inferior vena cava • Serves as exit point
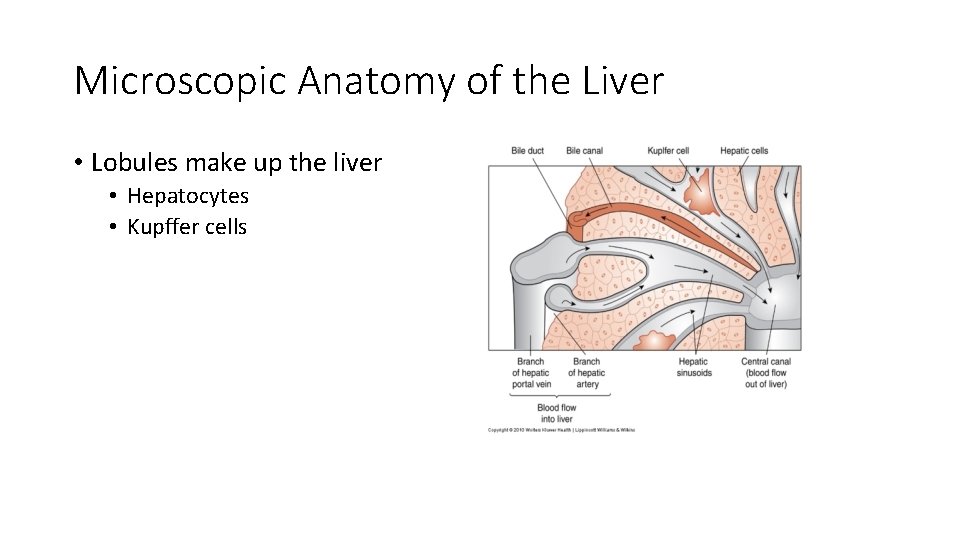
Microscopic Anatomy of the Liver • Lobules make up the liver • Hepatocytes • Kupffer cells

Functions of the Liver • Metabolism • Excretion • Detoxification • Storage • Immunologic functions
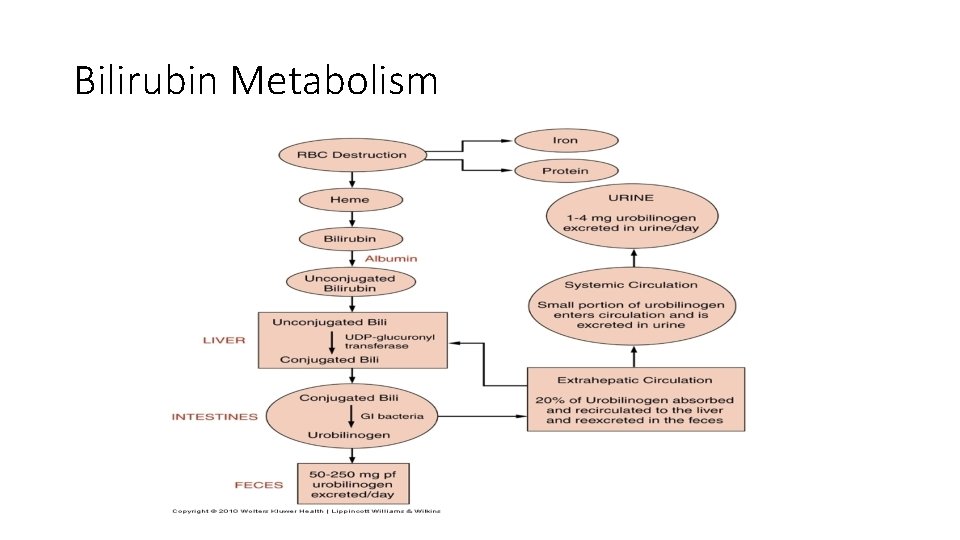
Bilirubin Metabolism

Jaudice/Icterus • Yellow discoloration of the skin, eyes and mucous membranes • Due to the presence of bilirubin • Kernicterus

Prehepatic Jaundice • Abnormality is outside the liver • Liver function is normal • Cause: too much bilirubin presented to liver

Hepatic Jaundice • Problem within the liver • Disorders of bilirubin metabolism and transport defects • Inherited • Acquired • Injury to hepatocytes
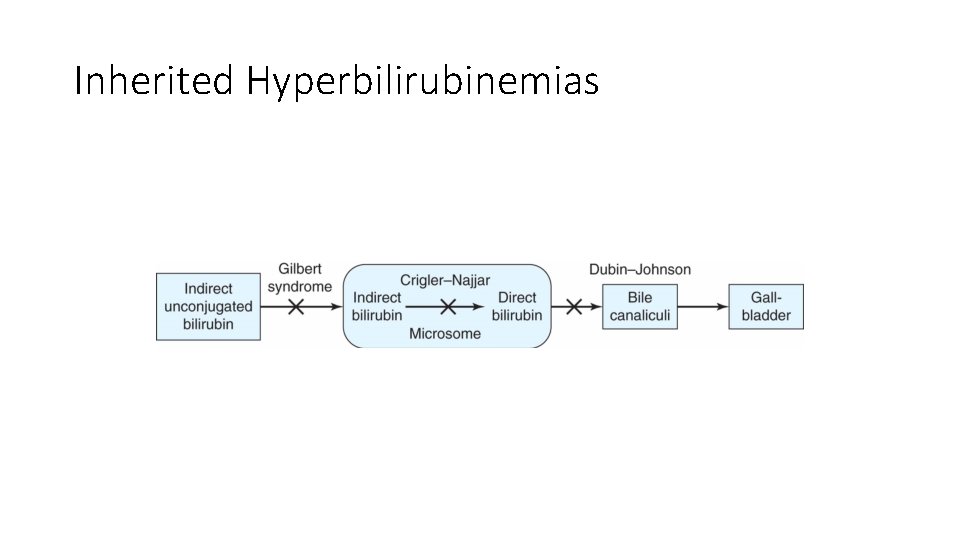
Inherited Hyperbilirubinemias

Acquried hyperbilirubinemias • Neonatal jaundice • UDP-transferase not fully developed • Liver becomes fully functional around day 3

Posthepatic Jaundice • • Abnormality is outside the liver Liver function is normal Biliary obstruction due to gallstones, tumors, edema Stool turns clay-colored due to lack of bile

Cirrhosis • Scar tissue replaces normal healthy liver tissue • Symptoms: • • Abdominal pain Nose bleeds Pale colored stools Edema in legs and abdomen

Reye Syndrome • Associated with viruses and aspirin • Present with • Vomiting • Mental impairment • Symptoms involve the liver

Drug and Alcohol Disorders • Accounts for 1/3 to ½ of acute liver failure • immune mediated injury to the hepatocytes • Ethanol is the most significant • Acetaminophen

Hepatitis • Types of hepatitis • Subtypes on next slide • Clinical Symptoms • Jaundice, dark urine, fatigue, nausea, abdominal pain

Hepatitis Viruses Virus Incubation Period Mode of transmission Vaccine Chronic Infection Hepatitis A 2 -6 weeks Fecal-oral Yes NO Hepatitis B 8 -26 weeks Parenteral, sexual Yes Hepatitis C 2 -15 weeks Parenteral, sexual No Yes Parenteral, sexual Yes Fecal-oral No ? Hepatitis D Hepatitis E 3 -6 weeks

Liver Function Panel • Albumin • Total bilirubin • Direct bilirubin • AST • ALP

Direct Bilirubin Analysis • Jendrassik-Grof • Measures Total and Conjugated bilirubin • Principle • • • Direct bilirubin reacts with diazo reagent Incubated Add ascorbic acid Alkaline tartrate and caffeine added Formation of blue-green azobilirubin Measured at 600 nm

Total Bilirubin Anaylsis • Pretreat specimen with caffeine-sodium benzoate • HCl and diazo added • Ascorbic acid added • Alkaline tartrate added = blue-green azobilirubin

Indirect Bilirubin • Calculated • Total bilirubin – direct bilirubin

Specimen Collection and Storage • No hemolysis • Light sensitive

Reference Ranges • Total Bilirubin: 0. 2 -1. 0 mg/d. L • Direct: < 0. 8 mg/d. L • Indirect: < 0. 2 mg/d. L

Enzymes • GGT • LDH

Urobilinogen • End product of bilirubin metabolism • Urine • Fecal

Prothrombin Time • Differentiates between cholestasis and severe hepatocellular disease

Ammonia • Neurotoxic • Elevated: advanced liver disease and renal failure

Enzymes

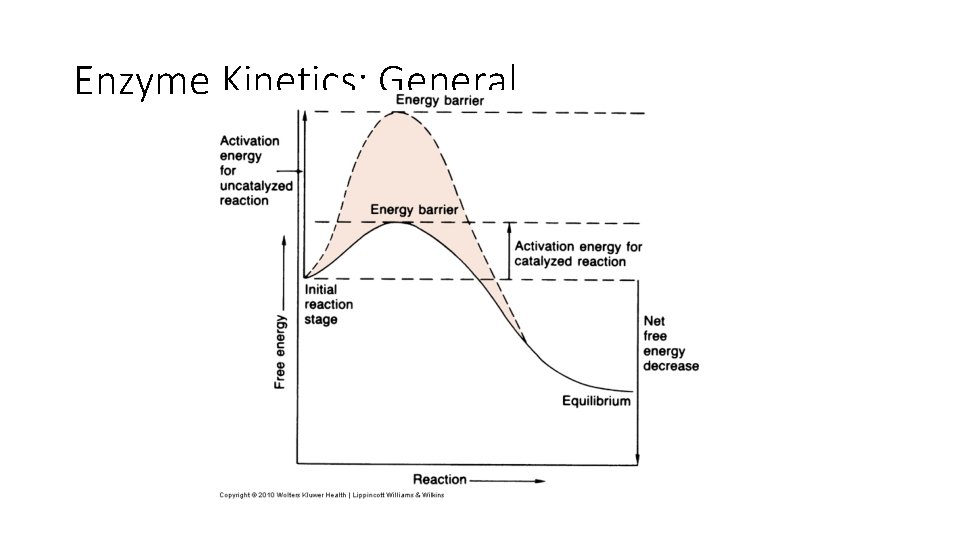
Enzymes • Functional proteins that catalyse biological reactions • Involved in all essential body reactions • Found in all body tissue • Used to diagnose and monitor disease
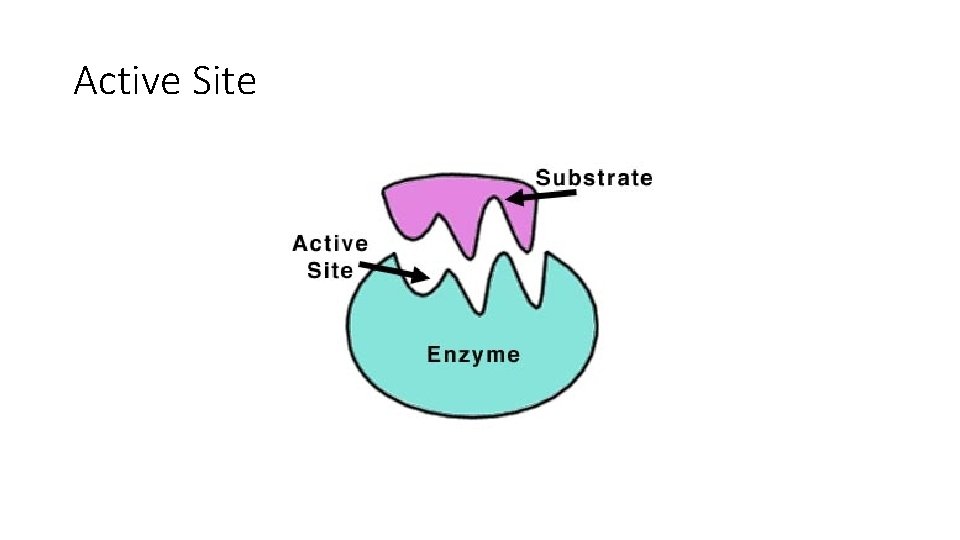
Active Site

Isoenzymes • Structurally different enzymes that catalyze the same reaction • Differing biochemical or immunological characteristics • Different detection methods

Co-factors • Non-protein substances required for normal enzyme activity • Types • • Activator Co-enzymes Holoenzyme Proenzyme/ zymogen

Plasma specific enzymes • Plasma is the normal site of action • Most often liver synthesized • Examples: plasmin, thrombin

Non-plasma specific enzymes • no known physiological function in the plasma • Some are secreted in the plasma • Increased number of this type seen with cell disruption or death
Enzyme Kinetics: General
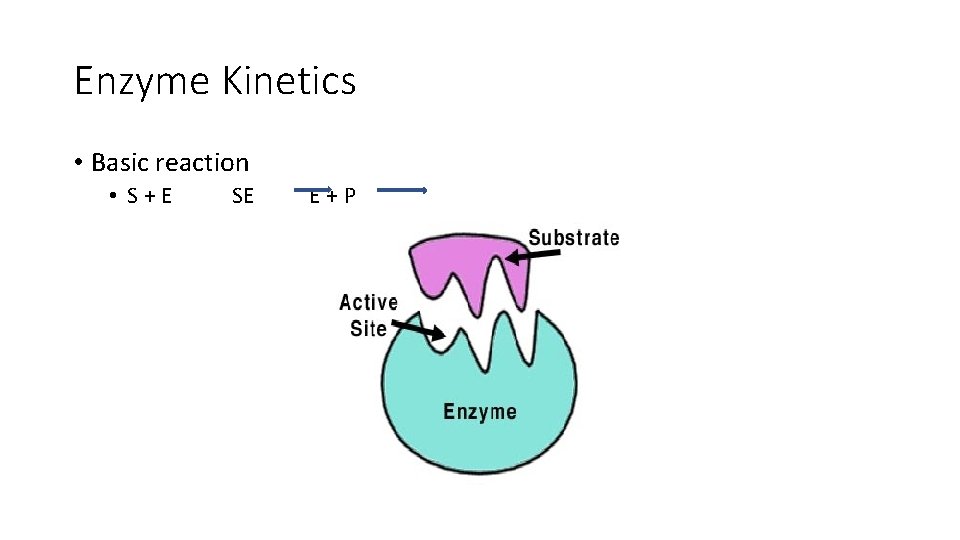
Enzyme Kinetics • Basic reaction • S+E SE E+P

Enzyme Kinetics • Enzymes differ in their ability to react with different substrates • • Absolute specificity Group specificity Bond specificity Stereoisomerism

Influencing Factors on Enzymatic Reactions • Substrate Concentration • Enzyme Concentration • p. H • Temperature • Cofactors • Inhibitors
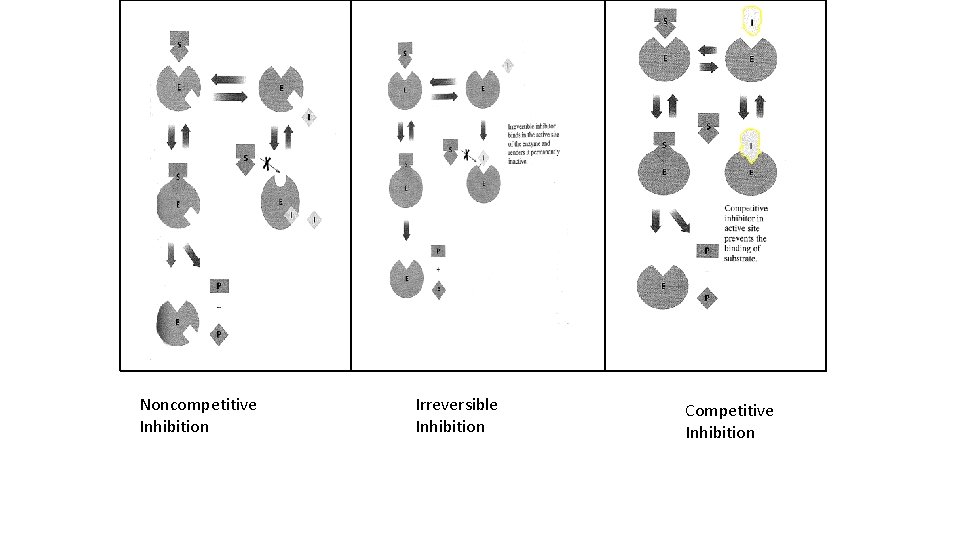
Noncompetitive Inhibition Irreversible Inhibition Competitive Inhibition

Measuring Enzyme Activity • Substrate depletion • Product production • Increase/decrease in cofactor/coenzyme

Measurement of Enzyme Activity Endpoint Kinetic • Measurement of the amount of substrate utilized over a fixed amount of time or by a fixed amount of serum • Measure the change in absorbance at specific time intervals over a specific amount of time

Calculation of Enzyme Activity • Common unit is International unit or IU/L • Conditions to be maintained • p. H • Temperature • Substrate • Using molar absorptivity • Each chemical is unique in absoptivity • Can determine concentration/activity

Lactate Dehydrogenase (LDH/LD) • Action • Catalyzes a reversible reaction between pyruvate and lactate with NAD as a coenzyme • Reaction:

LDH • Highest concentrations • • • Liver Heart Skeletal muscles Kidney RBCs
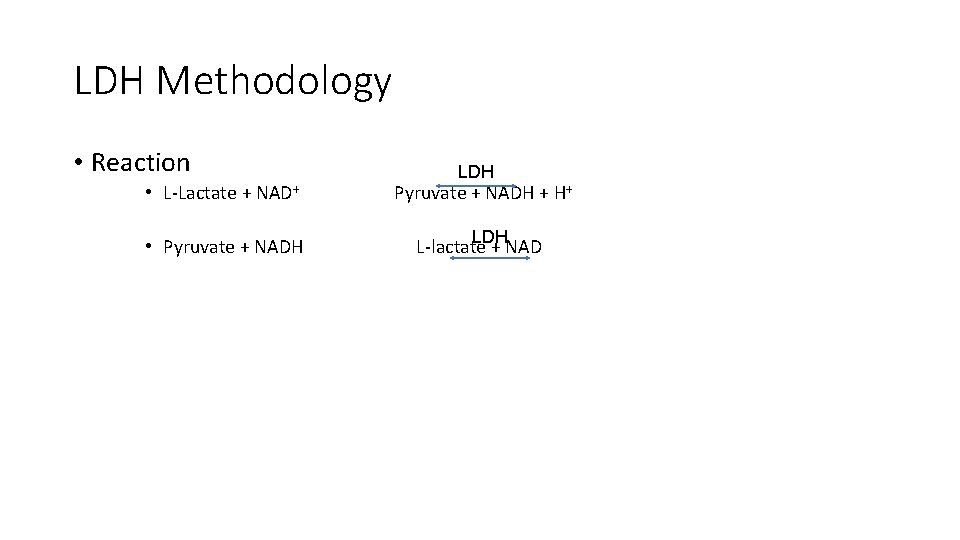
LDH Methodology • Reaction LDH • L-Lactate + NAD+ Pyruvate + NADH + H+ • Pyruvate + NADH L-lactate + NAD

Creatine Kinase (CK) • Action • Associated with the regeneration and storage of ATP • Catalyses the following reaction:

Creatine kinase (CK) • Highest concetrations • Skeletal • Heart • Brain

CK: Testing Methodology • Colorimetric • Absorbance change at 340 nm • Coupled enzyme: three step • Creatine + ATP • ATP + glucose • G 6 P + NADP Creatine phosphate +ATP CK Mg 2+ Glucose-6 - phosphate +ADP Hexokinase 6 -phosphogluconate + NADPH G 6 PD

Aspartate aminotransferase

AST • Highest concentration • • Heart Liver Skeletal muscle kidney

AST Testing Methodology • Coupled reaction • Measured at 340 nm • L-aspartate + alpha- oxoglutarate • Oxaloacetate + NADH + H+ AST oxaloacetate + glutamate Malate dehydrogenase malate +NAD+

Alanine aminotransferase

ALT • Highest concentrations • Liver • Lesser amounts in kidneys and RBCs
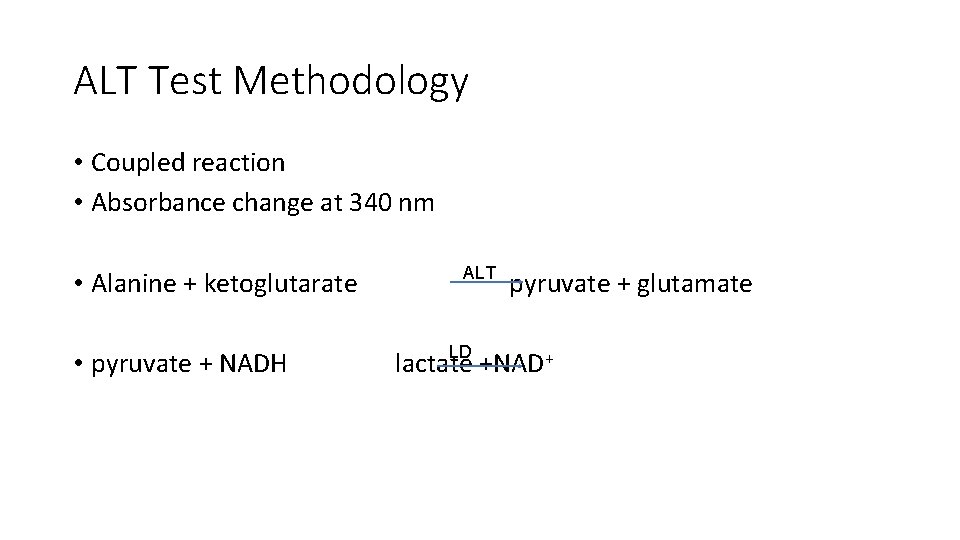
ALT Test Methodology • Coupled reaction • Absorbance change at 340 nm • Alanine + ketoglutarate • pyruvate + NADH ALT LD pyruvate + glutamate lactate +NAD+
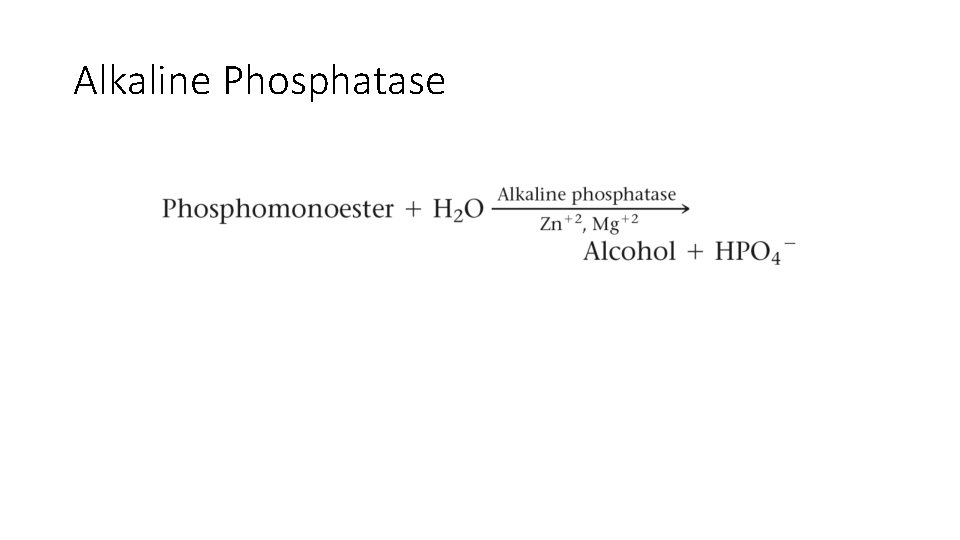
Alkaline Phosphatase

Alkaline Phosphatase (ALP) • Sources • • • Bone Liver Kidney Placenta Intestines

ALP: Clinical Significance Increased Decreased • Hepatobilary disease • Hypophosphatasia • Due to obstruction • Bone disorders • Paget disease • Hapatocellular conditions • Growing children • Pregnancy • Insufficent bone calcification

ALP: Testing Methodology • 4 -Nitrophenyphosphate • p. H: 10. 3 ALP 4 -Nitrophenoxide Mg 2+

Acid Phosphatase (ACP) • Highest concentration • • • Prostate gland Bone Liver Spleen RBCs Platelets

ACP: Clinical Significance • Increased • • • Prostate cancer Bone disease Paget’s disease Breast cancer with bone metastases Platelet damage

ACP: Methodology ACP • P-Nitrophenolphosphate P-Nitrophenol + Phosphate Ion p. H=5. 0

Gamma-Glutamyltransferase (GGT) • Source • • Liver Kidneys Pancreas intestine

GGT: Clinical Significance • Sensitive indicator of liver damage • Increased in patients taking enzyme-inducing drugs such as warfarin, phenobarbital and phenytoin • Indicator of alcoholism • Elevated in acute pancreatitis, diabetes mellitus and MI

GGT: Test Methodology • Colorimetric • P-nitroanilide is product and is chromogenic at 405 nm δ- glutamyl-p-nitroanilide + glycylglycine (Colorless) δ- glutamyl-glycylglycine + pnitroanilide (Yellow)

Amylase • Digestive enzyme • Hydrolzes/catalyzes the breakdown of starch and glycogen to carbohydrates

Amylase (AMS) • Source • • Pancreas Salivary glands Small intestine Fallopian tubes

AMS: Clinical Significance • Increased • • Acute pancreatitis Obstructive liver disease Acute alcoholism Mumps Peptic ulcer Intestinal obstruction Ectopic pregnancy Acute appendictis

AMS: Test Methodology • Amyloclastic • Saccharogenic • Chromogenic • Enzymatic
Lipase • Hydrolyzes triglycerides to produce alcohols and fatty acids

Lipase (LPS) • Found in • • Pancreas Gastric mucosa Intestinal mucosa Adipose tissue

LPS: Clinical Significance • Increased • • • Acute pancreatitis Peptic ulcer Duodenal ulcers Intestinal obstruction Cholecystitis • Inflammation of gallbladder

LPS: Testing Methodology • Titrametric • Measurement of liberated fatty acids • Turbidimetric • Simpler and faster • Activity based on how fast fats are hydrolzed by LPS • Enzymatic
- Slides: 85