Lithoplasty Balloon for a Calcific Aortic Valve Update
Lithoplasty Balloon for a Calcific Aortic Valve Update William A. Gray, MD, FSCAI Chief of Cardiovascular Services Main Line Health System

Disclosures Consultant/Institutional Research Support: Shockwave Medical

Lithotripsy for Structural Heart: Clinical Programs FIH I Study FIH III Study Feasibility Acute, Open Procedure Acute, Direct Aortic Chronic, Trans-femoral N=3 N=4 N ~ 20 Study Completed Planned 2018
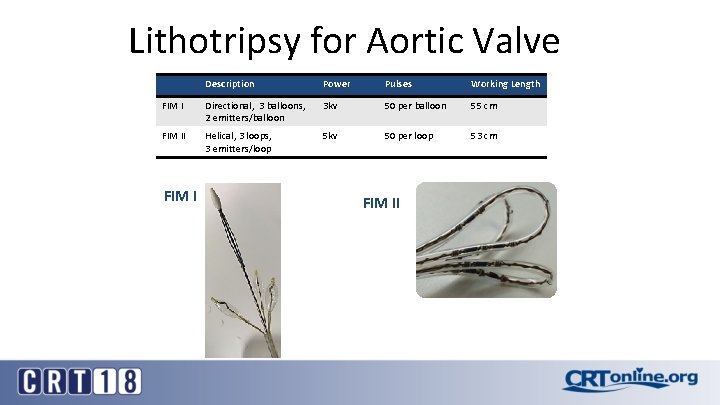
Lithotripsy for Aortic Valve Description Power Pulses Working Length FIM I Directional, 3 balloons, 2 emitters/balloon 3 kv 50 per balloon 55 cm FIM II Helical, 3 loops, 3 emitters/loop 5 kv 50 per loop 53 cm FIM II

Lithotripsy: Loop In Action

Aortic Valve Lithotripsy – FIM II • Prospective, single-arm, single-center, clinical study • Aortic Valve Lithotripsy followed by assessment of: – Impact on safety parameters – Change in pressure gradient – Leaflet mobility • Subjects undergo aortic valve replacement following excision of aortic valve leaflets Adrian Ebner, MD Italian Hospital Asuncion, Paraguay

FIM II Study: Key Inclusion / Exclusion Inclusion • Age of subject is >18. • Patient with severe degenerative aortic valve stenosis – AV area (EOA) of <1 cm 2, Mean AV gradient >40 mm. Hg, LVEF > 40% • Patients must be symptomatic from the aortic valve stenosis (dyspnea in NYHAclass II or greater, angina pectoris, or syncope) • Patients eligible for surgical valve replacement Exclusion • Aortic valve insufficiency > than 2+ • Coronary artery co-morbidity requiring revascularization • Previous median sternotomy or cardiac surgery • Myocardial infarction or percutaneous coronary intervention within the last 6 months • Stroke or TIA within last 90 days
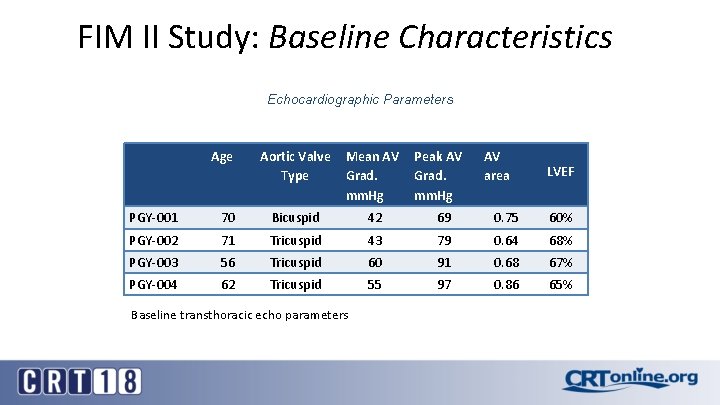
FIM II Study: Baseline Characteristics Echocardiographic Parameters Age Aortic Valve Type Mean AV Grad. mm. Hg Peak AV Grad. mm. Hg AV area LVEF PGY-001 70 Bicuspid 42 69 0. 75 60% PGY-002 71 Tricuspid 43 79 0. 64 68% PGY-003 56 Tricuspid 60 91 0. 68 67% PGY-004 62 Tricuspid 55 97 0. 86 65% Baseline transthoracic echo parameters
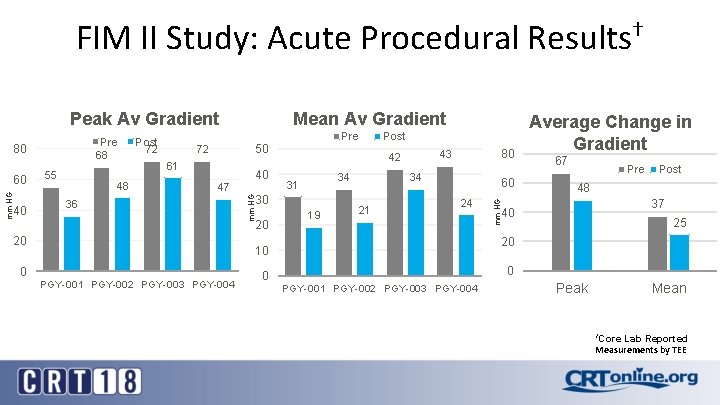
FIM II Study: Acute Procedural Results† mm HG 60 40 55 48 Post 72 Pre 50 72 61 40 47 36 20 20 Post 34 31 19 80 43 42 30 mm HG Pre 68 80 Mean Av Gradient 34 21 60 24 PGY-001 PGY-002 PGY-003 PGY-004 67 Pre Post 48 37 40 25 20 10 0 Average Change in Gradient mm HG Peak Av Gradient 0 0 PGY-001 PGY-002 PGY-003 PGY-004 Peak Mean †Core Lab Reported Measurements by TEE
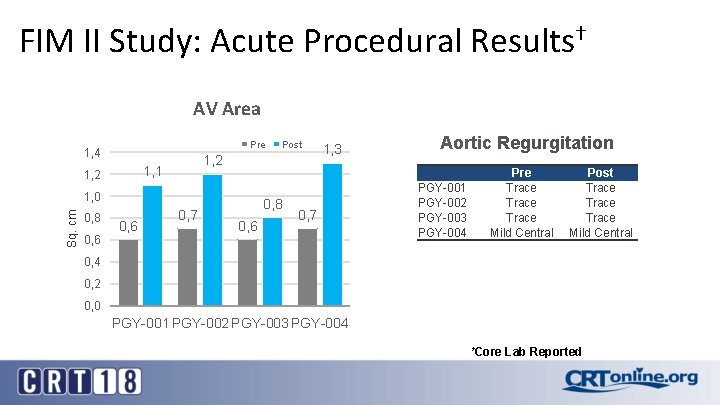
FIM II Study: Acute Procedural Results† AV Area Pre 1, 4 1, 2 1, 1 1, 2 Sq. cm 1, 0 0, 8 0, 6 Post 0, 7 0, 8 0, 6 1, 3 0, 7 Aortic Regurgitation PGY-001 PGY-002 PGY-003 PGY-004 Pre Trace Mild Central Post Trace Mild Central 0, 4 0, 2 0, 0 PGY-001 PGY-002 PGY-003 PGY-004 †Core Lab Reported
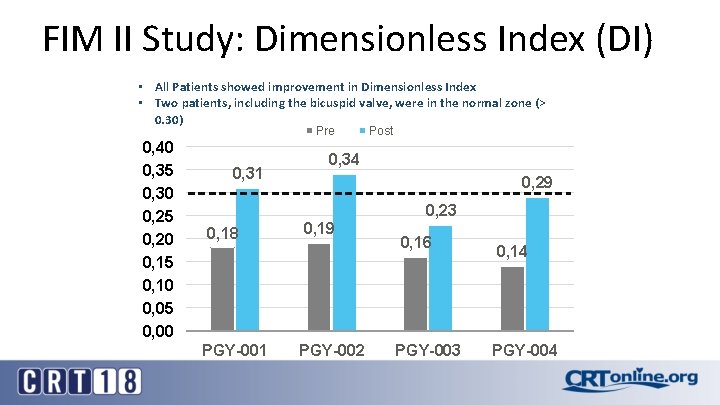
FIM II Study: Dimensionless Index (DI) • All Patients showed improvement in Dimensionless Index • Two patients, including the bicuspid valve, were in the normal zone (> 0. 30) Pre 0, 40 0, 35 0, 30 0, 25 0, 20 0, 15 0, 10 0, 05 0, 00 0, 31 Post 0, 34 0, 29 0, 23 0, 18 PGY-001 0, 19 PGY-002 0, 16 PGY-003 0, 14 PGY-004
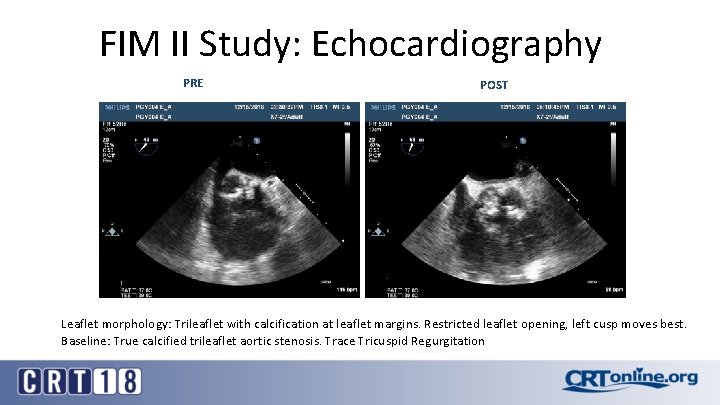
FIM II Study: Echocardiography PRE POST Leaflet morphology: Trileaflet with calcification at leaflet margins. Restricted leaflet opening, left cusp moves best. Baseline: True calcified trileaflet aortic stenosis. Trace Tricuspid Regurgitation
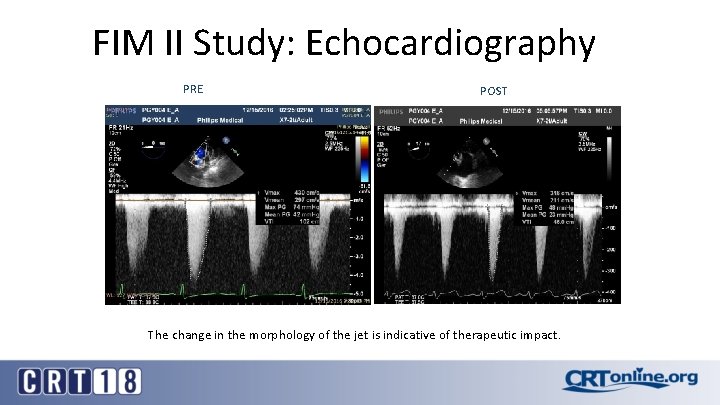
FIM II Study: Echocardiography PRE POST The change in the morphology of the jet is indicative of therapeutic impact.
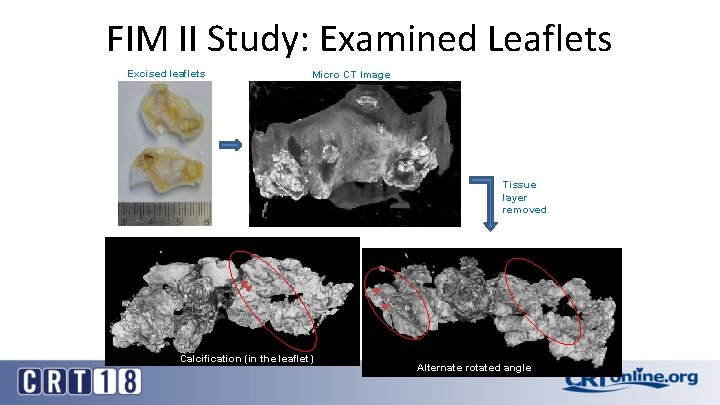
FIM II Study: Examined Leaflets Excised leaflets Micro CT Image Tissue layer removed Calcification (in the leaflet) Alternate rotated angle
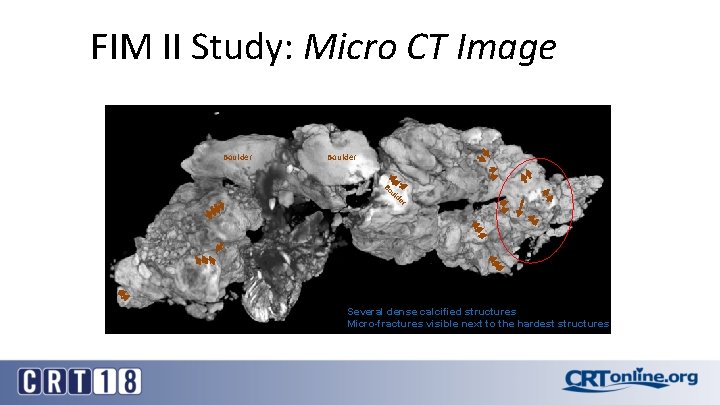
FIM II Study: Micro CT Image Boulder Bo ul d er Several dense calcified structures Micro-fractures visible next to the hardest structures

Design Updates for FIM III Description Power Pulses Working Length FIM I Directional, 3 balloons, 2 emitters/balloon 3 kv 50 per balloon 55 cm FIM II Helical, 3 loops, 3 emitters/loop 5 kv 50 per loop 53 cm FIM III Helical, 3 loops, 6 emitters/loop 5 kv 50 per loop 110 cm FIH III

Summary • FIM II Study demonstrated successful treatment of four patients with severe aortic stenosis. • Independent core lab adjudicated that all patients had improvement in AVA with reduction in peak/mean gradients (Average 19 & 12 mm Hg) • Reduction in mean gradient of at least 8 mm Hg in all patients • No significant change in aortic regurgitation in all four patients. • This FIM study demonstrates the early feasibility of a new non-implant based treatment for patients with aortic stenosis. • FIM III will begin in 2018 Device is not available for sale
- Slides: 17