Lithium Sulfur Batteries Presented by Timothy Cleary Oral

Lithium Sulfur Batteries Presented by Timothy Cleary Oral English Assessment Exam

A brief two part presentation: 1. Review of LFP batteries 2. Introduction to Li-S batteries

Review of Lithium Iron Phosphate Batteries

Electrospun Nanofiber-Based Anodes, Cathodes, and Separators for Advanced Lithium-Ion Batteries
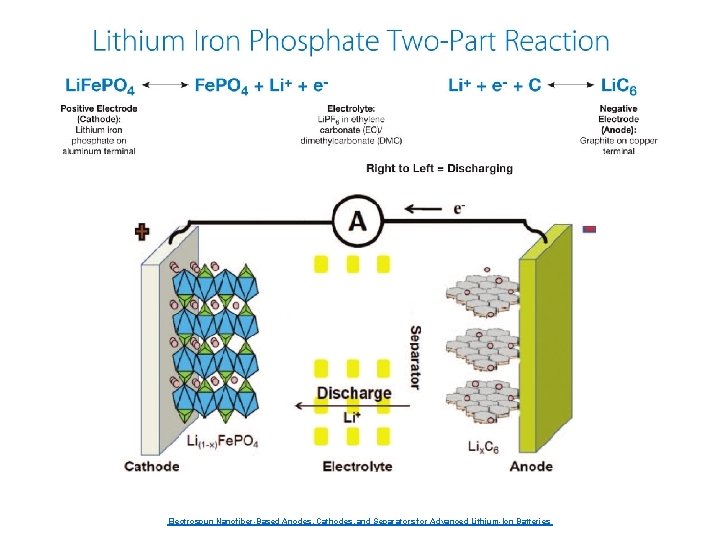
Electrospun Nanofiber-Based Anodes, Cathodes, and Separators for Advanced Lithium-Ion Batteries
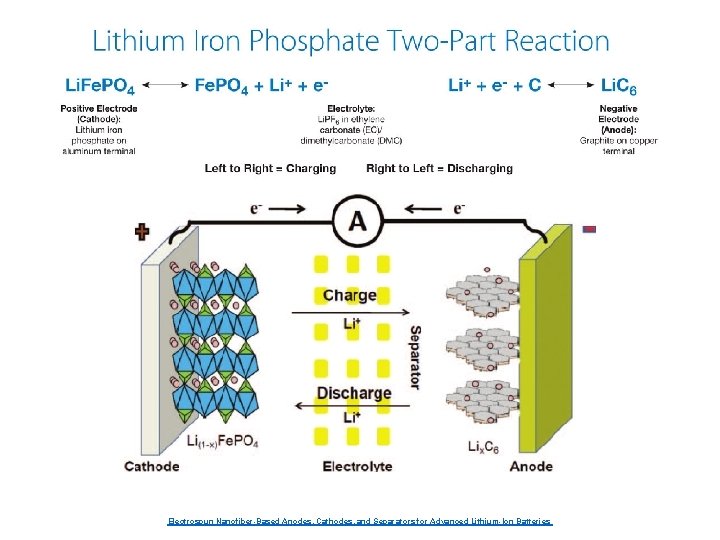
Electrospun Nanofiber-Based Anodes, Cathodes, and Separators for Advanced Lithium-Ion Batteries
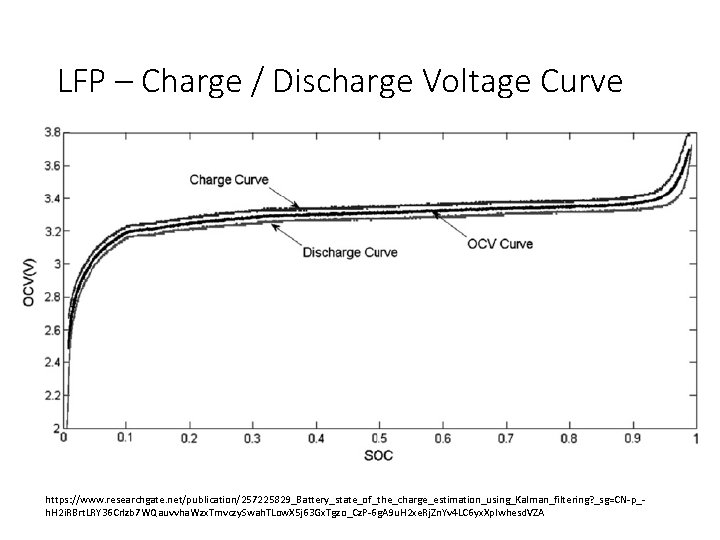
LFP – Charge / Discharge Voltage Curve https: //www. researchgate. net/publication/257225829_Battery_state_of_the_charge_estimation_using_Kalman_filtering? _sg=CN-p_h. H 2 i. RBrt. LRY 36 Crlzb 7 WQauvvha. Wzx. Tmvczy. Swah. TLow. X 5 j 63 Gx. Tgzo_Cz. P-6 g. A 9 u. H 2 xe. Rj. Zn. Yv 4 LC 6 yx. Xp. Iwhesd. VZA

Introduction to Lithium Sulfur Batteries

Interesting Voltage Curve Tight voltage range (2. 5 to 1. 4 V) Different charge/discharge profiles Discharge - S 8 to Li 2 S (2. 3 V) High plateau reduction of long chain lithium polysulfides (Li 2 Sx, 4 <= x) (2. 1 V) Low plateau reduction of short chain lithium polysulfides (Li 2 Sx, x <= 4) Charge - Li 2 S to S 8 (Copyright 2013, Royal Society of Chemistry)
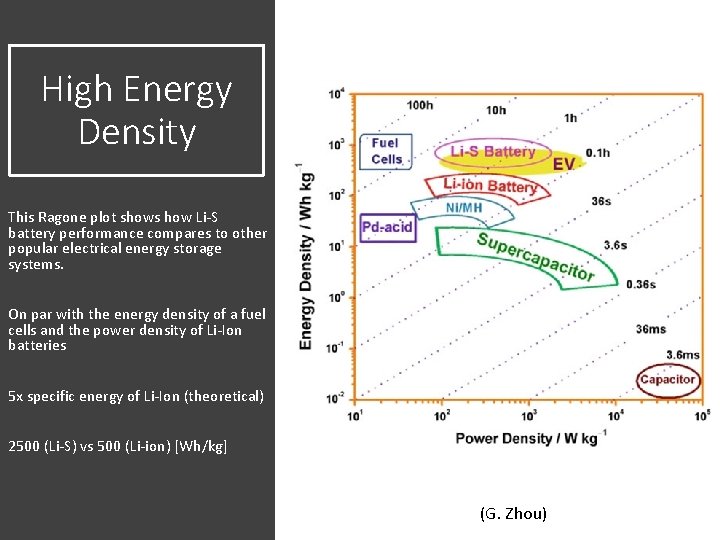
High Energy Density This Ragone plot shows how Li-S battery performance compares to other popular electrical energy storage systems. On par with the energy density of a fuel cells and the power density of Li-Ion batteries 5 x specific energy of Li-Ion (theoretical) 2500 (Li-S) vs 500 (Li-ion) [Wh/kg] (G. Zhou)

Sulfur • Inexpensive • Non toxic • 10 th most abundant element

Safety Performance Impressive nail puncture performance, no smoke and almost no energy release

What cause this cell to fail? Shuttle effect / Sulfur insulation • The soluble polysulfide intermediates (Li 2 Sx, 3 ≤ x ≤ 8) in the organic liquid electrolyte during the cycle process bring about the polysulfide “shuttle effect, ” which leads to irreversible capacity loss and corrosion on the lithium–metal anode [Yang, Wang]. • Insulating characteristic of sulfur and its discharge products (Li 2 S), leading to a low utilization of active material [Wang]; Structural instability • Large volumetric expansion/shrinkage (80%) during discharge/charge due to the different densities of sulfur (2. 03 g cm− 3) and lithium sulfides (1. 67 g cm− 3), resulting in an instability of the electrode structure; As a result, todays realistic life of a Li. S cell is about 500 to 1000 cycles

Research Goals In preparation for when Li-S cells become commercially available • Modeling & characterization • Application of observers/estimators for SOC prediction • Life cycle optimization

Companies Making Li-S Cells • OXIS Energy – Achieving ~ 500 cycle and bosting 400 Wh/kg • SAFT – Li SO 2 (primary battery) • Sony – Announces plans for commercialization of lithiumsulfur (Li-S) / magnesium sulfur (Mg-S) by 2020

References • 2017_Book_Design. Fabrication. And. Electroche © Springer Nature Singapore Pte Ltd. 2017 G. Zhou, Design, Fabrication and Electrochemical Performance of Nanostructured Carbon Based Materials for High-Energy Lithium–Sulfur Batteries, Springer Theses, DOI 10. 1007/978 -981 -10 -3406 -0_1 • OXIS Energy - https: //oxisenergy. com/technology/ • Wang D-W et al (2013) Carbon-sulfur composites for Li–S batteries: status and prospects. J Mater Chem A 1(33): 9382– 9394 • Wang D-W et al (2012) A microporous-mesoporous carbon with graphitic structure for a high-rate stable sulfur cathode in carbonate solvent-based Li–S batteries. Phys Chem Phys 14(24): 8703– 8710 • Yang Y, Zheng G, Cui Y (2013) Nanostructured sulfur cathodes. Chem Soc Rev 42(7): 3018– 3032
- Slides: 16