List of Contents Artificial blood Cell salvage Immunomodulation


List of Contents • Artificial blood • Cell salvage • Immunomodulation & Leukoreduction

Artificial Blood

Reasons for Developing • Storage requirement for human RBC limit the availability of blood at disaster or battlefield - sterilization - cross-matching • Donor blood shortage • Blood-transmitted disease

Ideal Artificial Blood • • • Transport oxygen Require no cross-matching or compatibility test Suitable for long-term storage (at room temp. ) Survive in the circulation for several weeks Free of side effects Effectively deliver oxygen to tissues

Types of Artificial Blood • Hemoglobin-based oxygen carriers (HBOCs) - Cross-linked Hb - Polymerized Hb - Conjugated Hb • Perfluorocarbons

HBOCs • Source of Hb - human; shortage - bovine; bovine spongiform encephalopathy - recombinant; expensive • Colloid oncotic pressure; plasma expander • Vasopressor effect; partly d/t scavenging NO advantageous for septic shock

Perfluorocarbons • Acceptable by certain group of people for religious reasons • Not miscible to acqueous solution; emulsions • Contribution to O 2 content; linear to Pa. O 2 requires a high Pa. O 2 • Short intravascular dwell time (T 1/2; 12 – 18 h) --- suitable for augmenting ANH
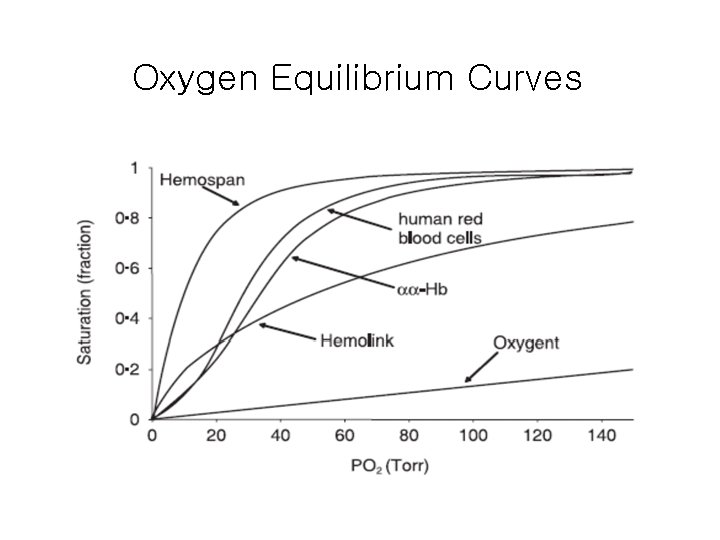
Oxygen Equilibrium Curves
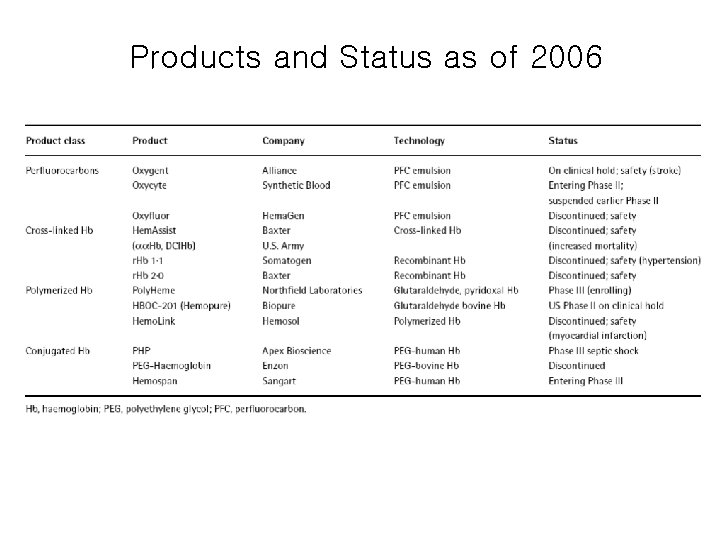
Products and Status as of 2006

Summary • Much improved safety of human blood products (AIDS; 1/800, 000, HCV; 1/600, 000) • Still there is a shortage of donor blood for transfusion • Long way to go before artificial blood can replace real blood in routine transfusions

Cell Salvage

Contraindications to CS • Hemolytic agents; water, alcohol, H 2 O 2 • Pharmacologic agents - clotting agents - irrigating solutions (betadine etc. ) - methymethacrylate • Contaminants - urine, bone chips, fat - bowel contents, infection, amniotic fluid • Malignancy

Bacterial Contamination - I • Bowel surgery, penetrating abdominal trauma or infected wound surgery; bacteremia or sepsis • Bacterial contamination of CS blood; routine - CS blood in cardiac surgery; 30% - liver transplant; 9% - usually skin origin with no clinical sequela • Contamination with frank stool; no ↑in sepsis

Bacterial Contamination - II • ↓bacteria to 5 – 23% after washing • ↓bacteria to 1% with cell washing & WBC filter • Differentiating between gross contamination and possible or unobserved contamination • Bacterial contamination of allogeneic blood --- Top 3 cause of death (espiecially platelet) • Broad-spectrum antibiotics • Bacterial contamination of CS blood; theoretical risk

Obstetrics • Amniotic fluid embolism; rare (1/8, 000 -30, 000) • Exact pathogenesis; unknown (Dx of exclusion) • Washing removes every component of amniotic fluid effectively except fetal squamous cells which can be removed by WBC depletion filter • Fetal Hemoglobin cause isoimmunization --- prevented by anti-D immune globulin • 390 cases; CS in amniotic fluid contaminted blood without filtration

Malignancy • Tumor cells in the patient’s circulation is not uncommon preoperatively and intraoperatively • Percentage of circulating tumor cells capable of forming metastatic lesion; 0. 01 – 0. 000001% • Prospective study in radical prostatectomy --- comparable outcome btw CS and PAD • Method of removal of tumor cells in CS blood - WBC depletion filter - irradiaion; 50 Gy

Immunomodulation & Leucoreduction

Immunologic Responses to Allogeneic Transfusion • Alloimmunization • Downregulation of immune responses → “Transfusion-related immunomodulation” TRIM

Clinical Effects associated with TRIM • Improved renal allograft survival • Impaired cancer prognosis • Increased risk of postop bacterial infections
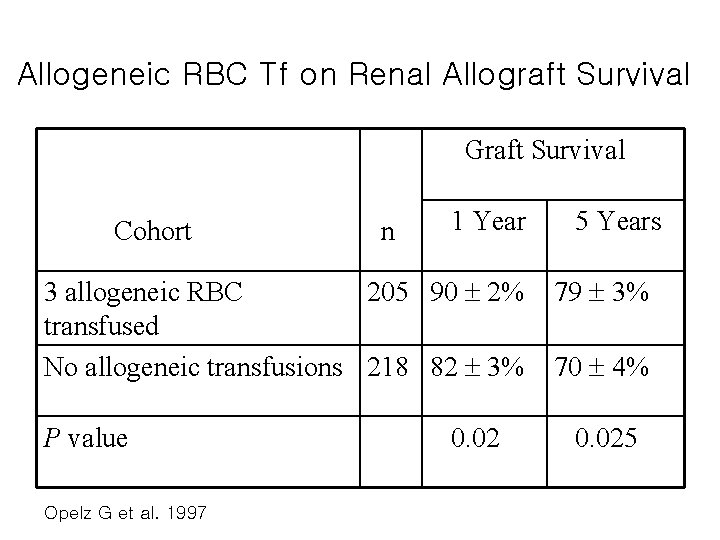
Allogeneic RBC Tf on Renal Allograft Survival Graft Survival Cohort n 1 Year 3 allogeneic RBC 205 90 2% transfused No allogeneic transfusions 218 82 3% P value Opelz G et al. 1997 0. 02 5 Years 79 3% 70 4% 0. 025
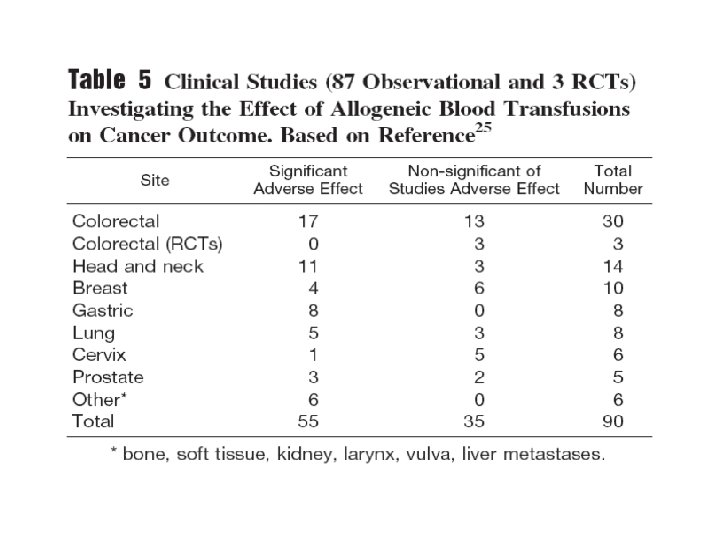

Allogeneic Transfusion & Tumor Growth - human studies (90 studies) - • No convincing data • Differences in prognostic features btw groups - clinical stage of the malignancy - size, histological grade and type of the tumor - patient age - preoperative hemoglobin - duration and extent of surgery - amount of perioperative blood loss - frequency of chronic systemic illness

Allogeneic Transfusion & Tumor Growth - animal studies (25 studies) - • 4 studies; no demonstrable effect • 3 studies; inhibition of tumor growth • The majority (18 of 25); ↑tumor growth --- This effect can be ameliorated by prestorage leukoreduction of the allogeneic blood

↑Risk of Infection ass. with TRIM • 8 randomized controlled trials - 4 studies; ↑ association - possible explanation for lack of consistency --- TRIM effect; quite small (< 10%) an RCT enrolling 10, 000 to 20, 000 subjects would be required

Methods of Leukoreduction Method WBC removal (%) Buffy coat removal 80 % Washed RBC 80% Leukodepletion filter > 99% Irradiation > 99%

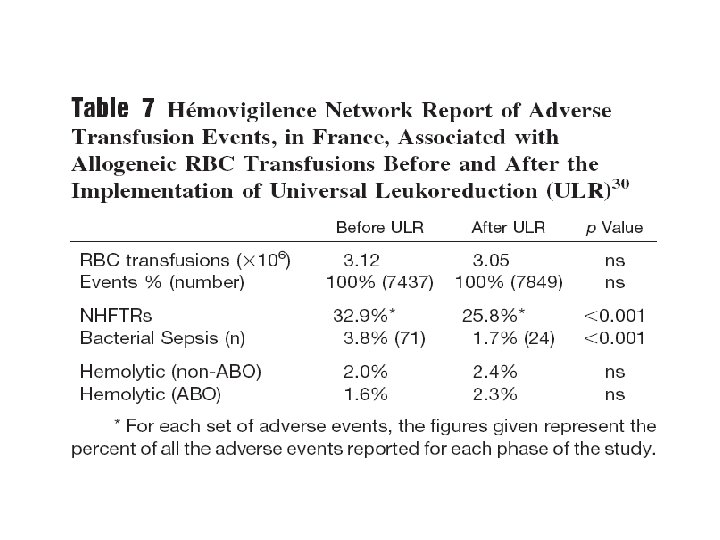
↓Frequency & Severity of NHFTRs • Causes of NHFTR - not WBC - cytokines in the transfused blood products • Leukoreduction (prestorage) is associated with a significant reduction in the NHFTRs
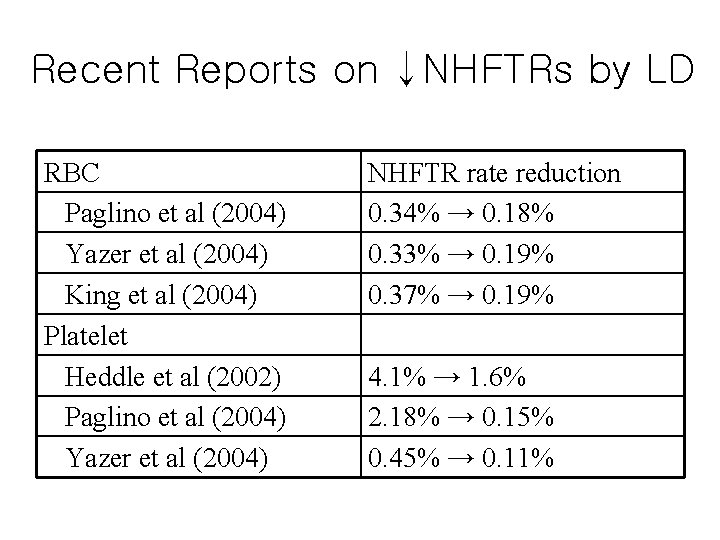
Recent Reports on ↓NHFTRs by LD RBC Paglino et al (2004) Yazer et al (2004) King et al (2004) Platelet Heddle et al (2002) Paglino et al (2004) Yazer et al (2004) NHFTR rate reduction 0. 34% → 0. 18% 0. 33% → 0. 19% 0. 37% → 0. 19% 4. 1% → 1. 6% 2. 18% → 0. 15% 0. 45% → 0. 11%

↓Post-transfusion CMV • High risk group for post-transfusion CMV - low birth weight infants - some oncology patients - allogeneic bone marrow transplant patients • Prevalence of post-transfusion CMV infection - without any prevention technology; 30% - leukoreduced RBC or platelet; 1. 3 – 2. 5%

Alloimmunization & Plt refractoriness • Can be reduced considerably by leukoreduction • Relative risk reduction of HLA alloimmunization - Sintnicolas et al. (1995) 0. 32 (95% CI; 0. 18 – 0. 59) - TRAP (1997) 0. 30 (95% CI; 0. 20 – 0. 46)

Variant CJD Transmission • Universal leukoreduction over the past 6 yrs in Western European countries • ↓risk of TSE infectivity in a hamster model --- 48. 1% → 31. 5%

Clinical Benefits of Leukoreduction • Proven relevant clinically - ↓frequency and severity of NHFTRs - ↓risk of CMV transmission - ↓risk of HLA-alloimmunization & plt refractoriness • Likely clinically relevant - ↓infection risk associated with TRIM - ↓organ dysfunction and mortality - ↓direct risk of transfusion-transmitted bacterial infection • Unproven clinically - Avoidence of v. CJD transmission, HTLV, EBV etc. - ↓risk of GVHD, TRALI
- Slides: 34