Liquids Vapor Pressure Vapor Gas phase of substance

Liquids & Vapor Pressure

Vapor • Gas phase of substance that is normally liquid at room temperature • Some evaporation occurs at all temperatures • The easier a substance evaporates, the weaker the IMF
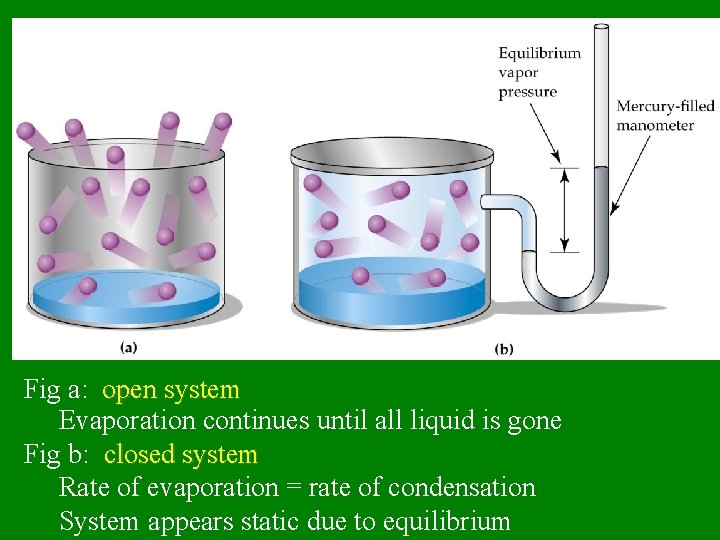
Fig a: open system Evaporation continues until all liquid is gone Fig b: closed system Rate of evaporation = rate of condensation System appears static due to equilibrium
How does evaporation occur?
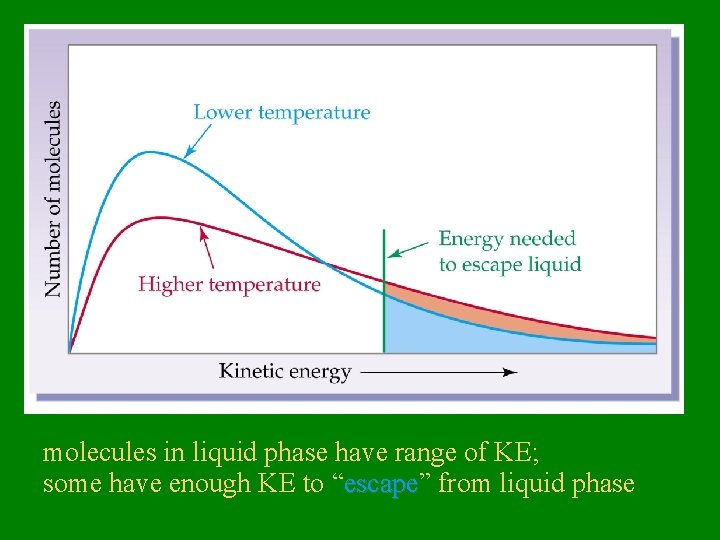
molecules in liquid phase have range of KE; some have enough KE to “escape” escape from liquid phase

Vapor Pressure – Pressure exerted by vapor over its liquid – VP is affected by temperature: • higher the T, higher the VP
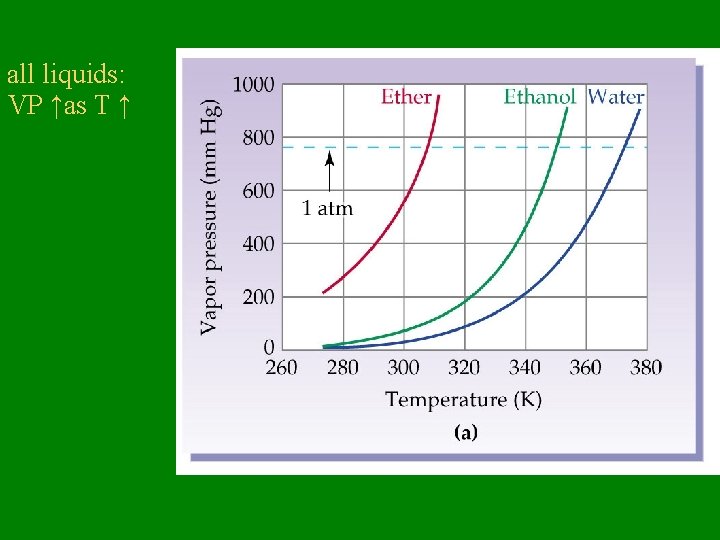
all liquids: VP ↑as T ↑

Vapor Pressure • VP does NOT depend on how much liquid is present • VP depends only on temperature

IMF in Liquids WEAK FORCES • • High VP High rate evaporation Low bp Small Hv STRONG FORCES • • Low VP Low rate evaporation High bp Large Hv

Phase Changes • Melting • Boiling – Vaporization – occurs throughout liquid, T constant) • Evaporation (occurs only at surface; T ↓) • Sublimation • Freezing • Deposition • Condensation

Phase Changes are accompanied by energy changes • Endothermic Process: Energy is absorbed • Exothermic Process: Energy is released

• Which phase changes absorb energy (endothermic)? Melting, Boiling (vaporization) • Which phase changes release energy (exothermic)? Freezing , Condensation

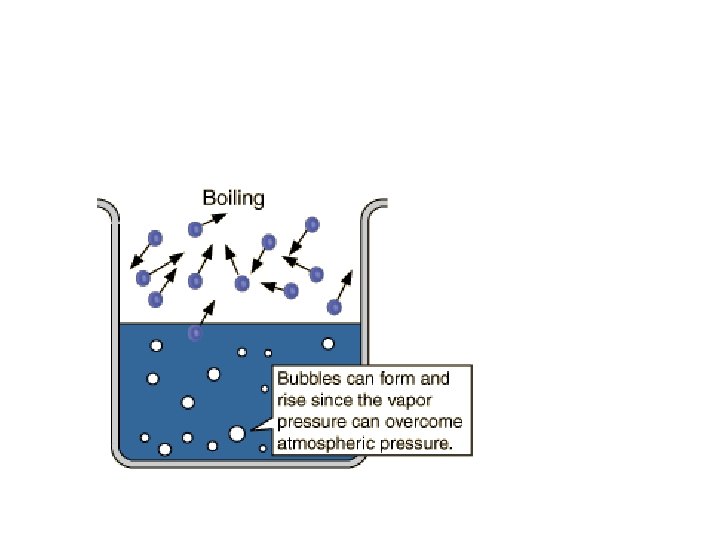
Boiling Point • Temperature at which: VP liquid = external atmospheric P • Normal Boiling Point: Point temp at which VP liquid = 1 atm • Substances with high bp’s have strong IMF’s

Melting Point • = temp at which liquid & solid phases of substance coexist at equilibrium • higher the mp, the stronger the IMF

Freezing Point • Temp at which liquid is converted to crystalline solid • How does fp compare to mp? They’re the same: for H 2 O = 0˚C

Strong or Weak attractive forces? • • High vapor pressure Large Hv High boiling point Low vapor pressure Small Hv Evaporates rapidly Evaporates slowly • • Weak Strong Weak Strong

Boiling and Pressure • If increase external pressure (ex: camping in > than 100 o. C Death Valley), boiling point is ____ • If decrease external pressure (ex: eating Raman noodles at top of Mt. Whitney), the boiling point is ____ < than 100 o. C

Boiling vs. Evaporation BOILING • occurs throughout liquid • occurs at definite T • occurs at constant T • only spontaneous when VP = external pressure • • EVAPORATION occurs at surface of liquid (or solid) occurs at all T liquid cools off as evaporates spontaneous all the time
- Slides: 19