Liquids Solids and Intermolecular Forces Chapter 11 Geckos














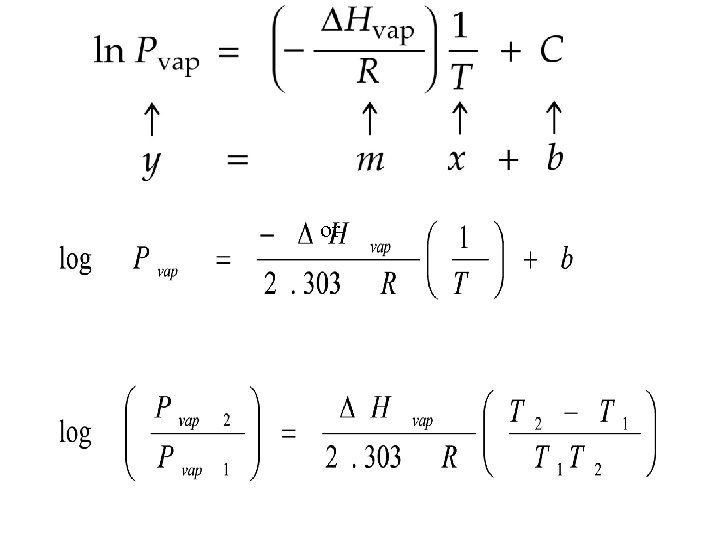














- Slides: 68

Liquids, Solids, and Intermolecular Forces Chapter 11

Gecko’s Hairy Feet • Nanostructures on the soles of gecko feet. Thanks to about one billion hierarchically organized nanohairs, the gecko can go for a walk on walls and ceilings, unlike people. • Image: Max Planck Institute for Metals Research


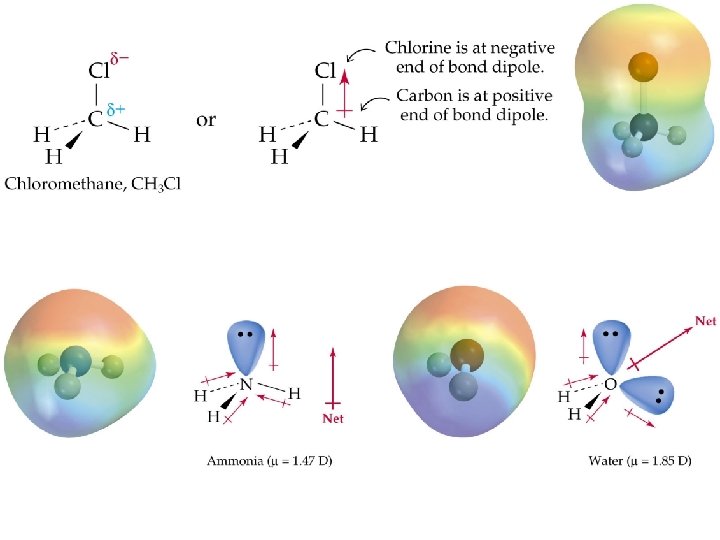
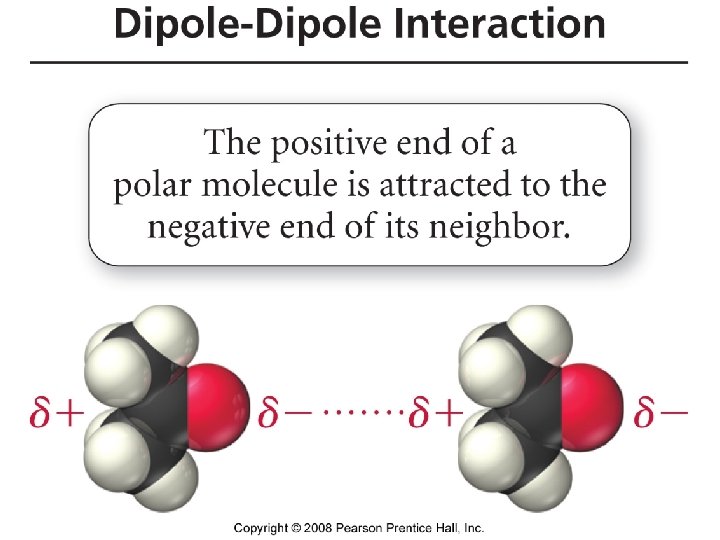
Polar Molecules • Dipole - A molecule such as HF which has a positive and a negative end. This dipolar character is often represented by an arrow pointing towards the negative charge. • Dipole moments – the measure of the net molecular polarity – Measure of separation of charge • Measured in units of Debyes (D) = Qr (charge x separation)


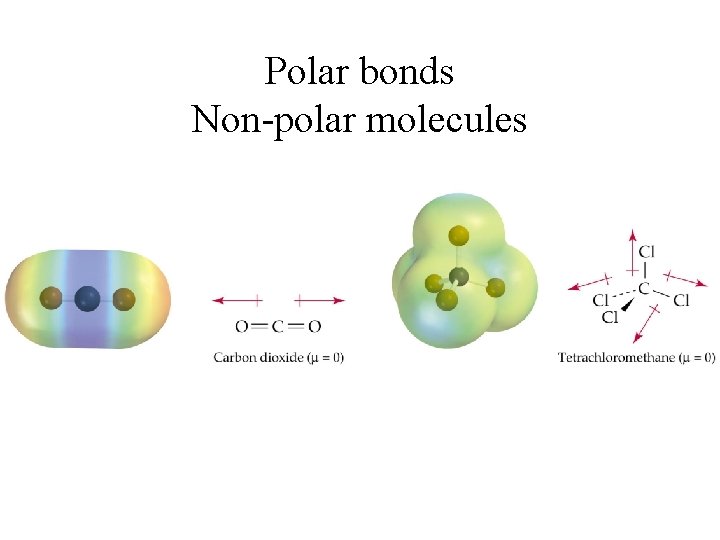
Polar bonds Non-polar molecules

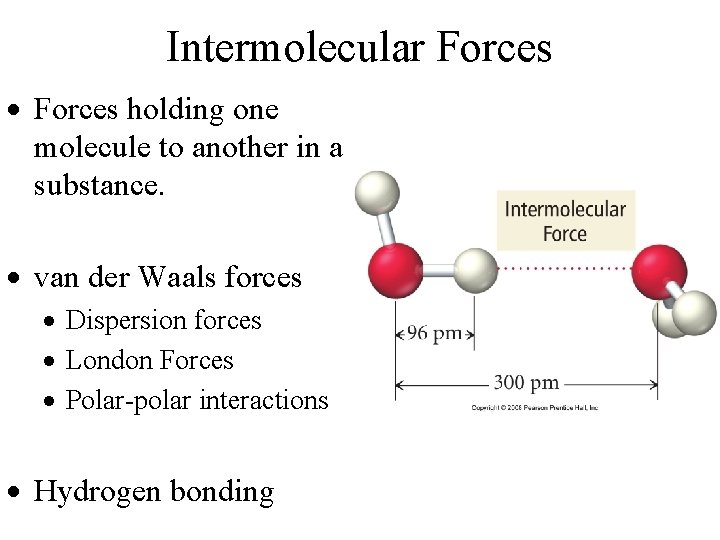
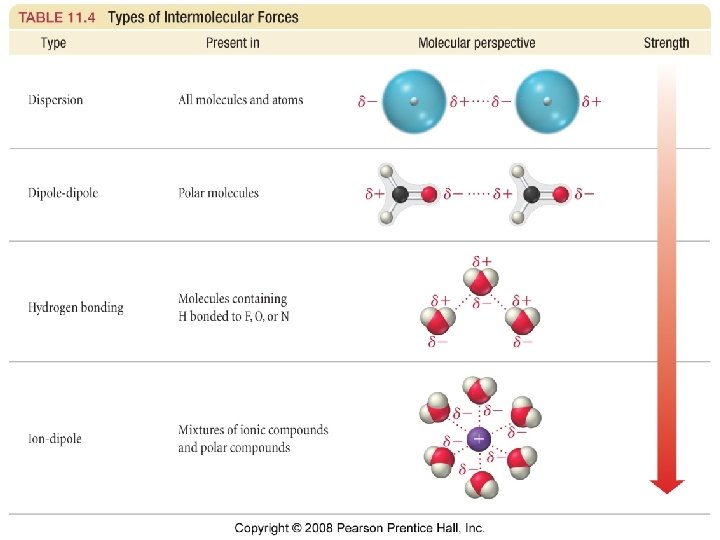
Intermolecular Forces · Forces holding one molecule to another in a substance. · van der Waals forces · Dispersion forces · London Forces · Polar-polar interactions · Hydrogen bonding


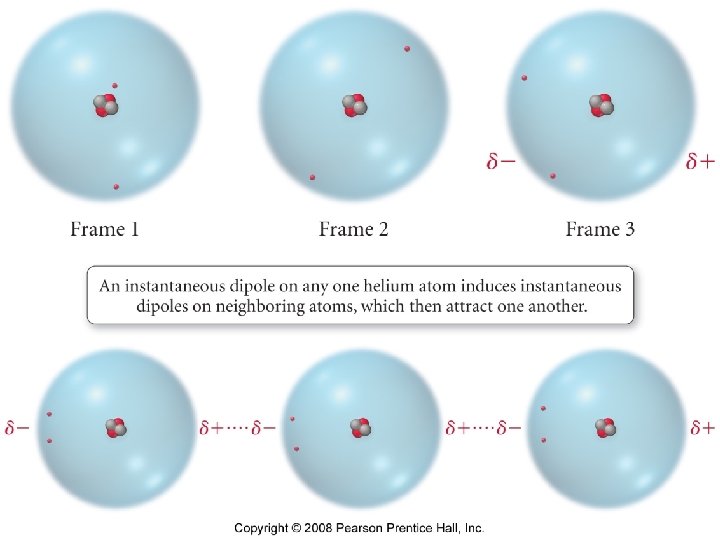
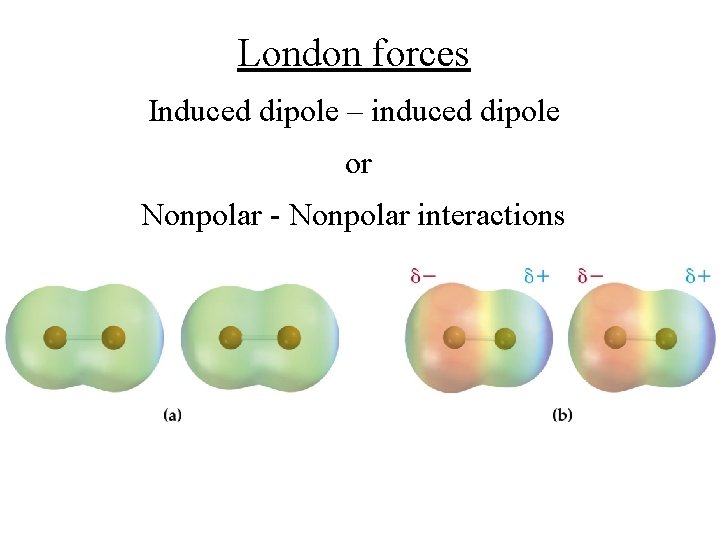
London forces Induced dipole – induced dipole or Nonpolar - Nonpolar interactions

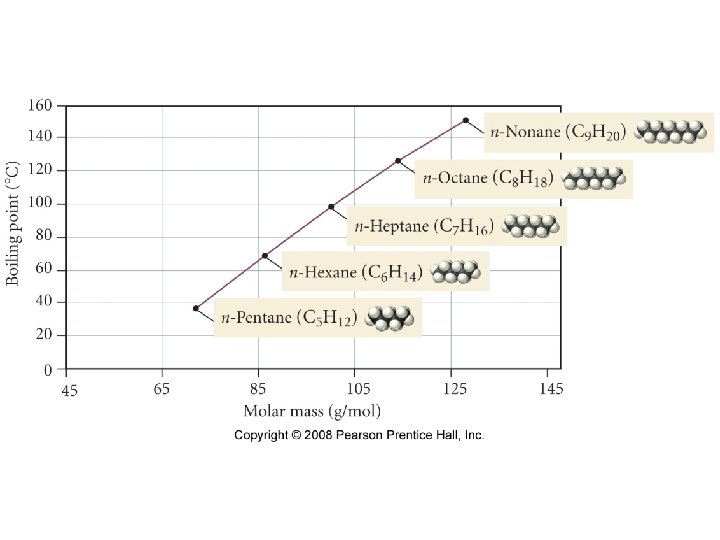
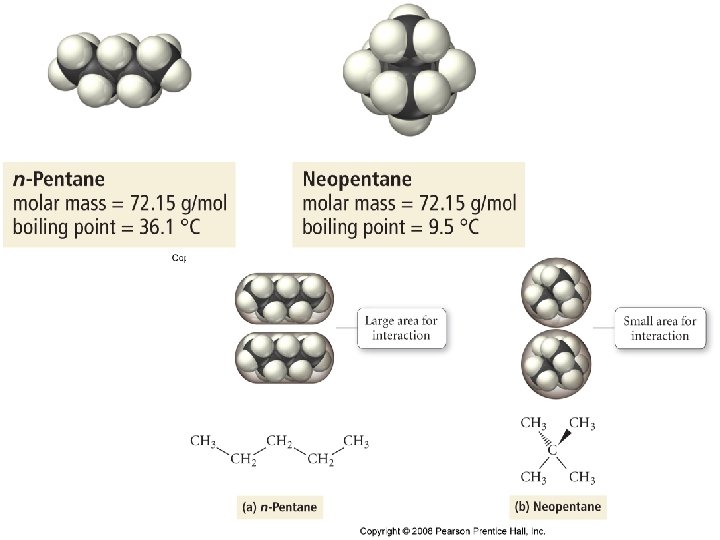
Polarizability • The ease with which a molecule/atoms electron cloud can be distorted, thereby inducing a dipole moment. • Increasing the number of electrons increases the polarizability of an atom or molecule.

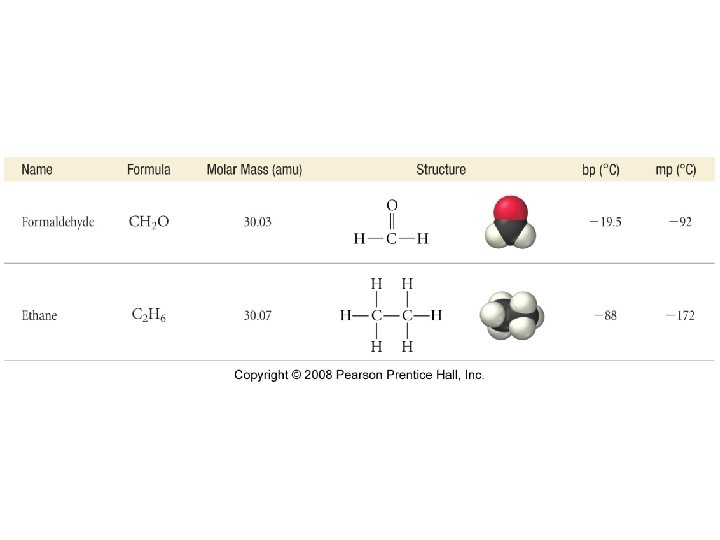
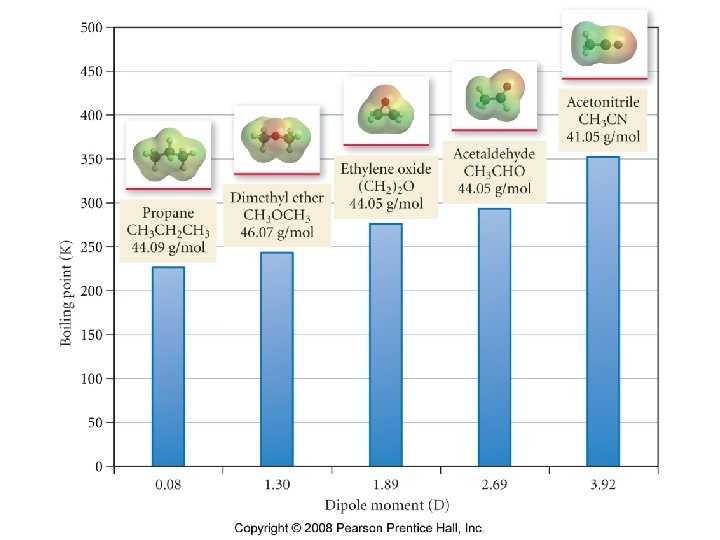
How does polarity affect molecular properties?




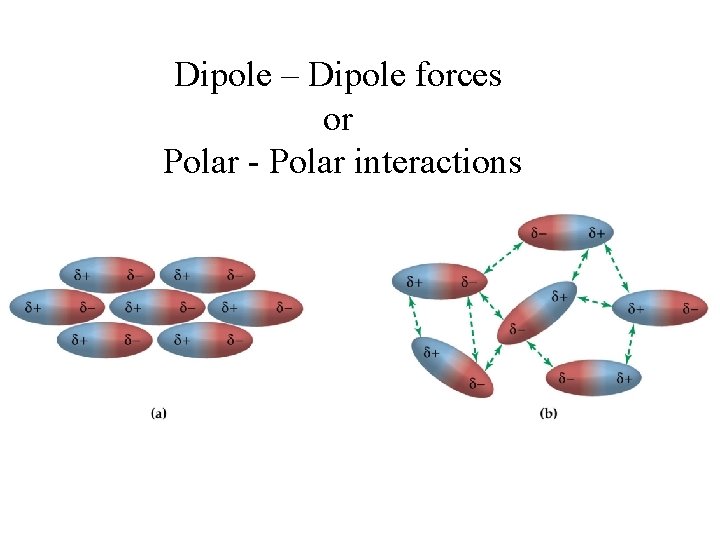
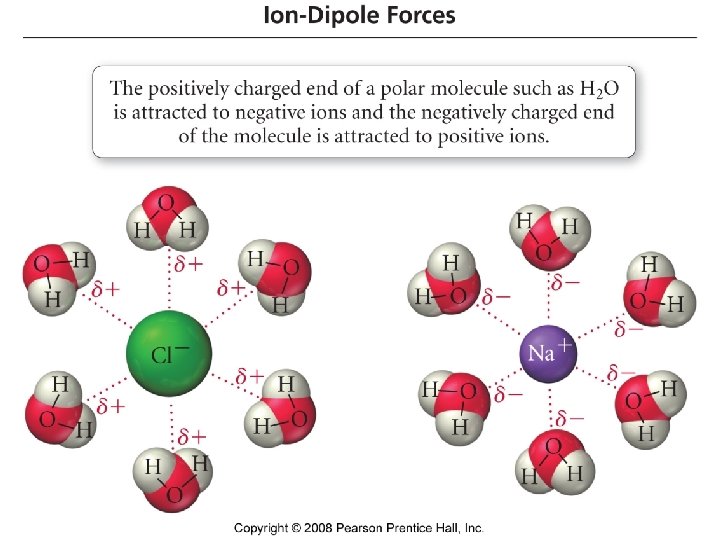
Dipole – Dipole forces or Polar - Polar interactions



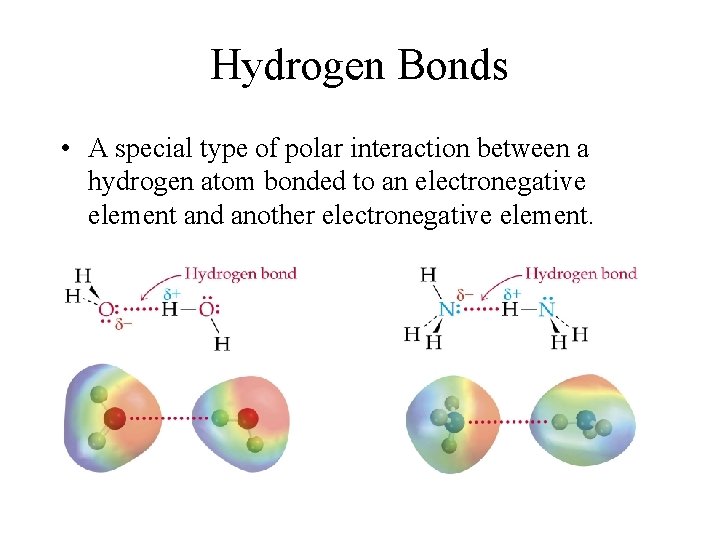
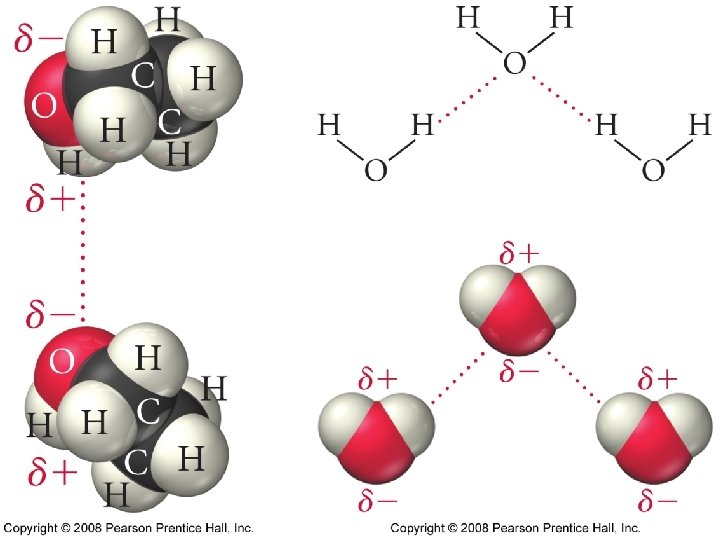
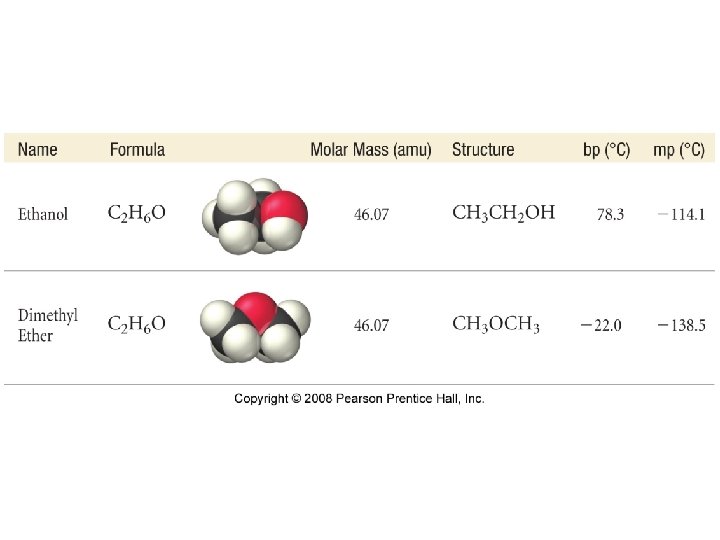
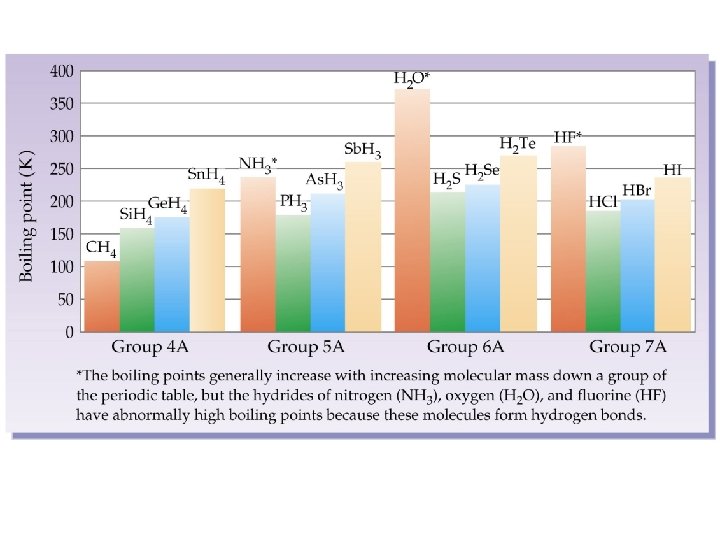
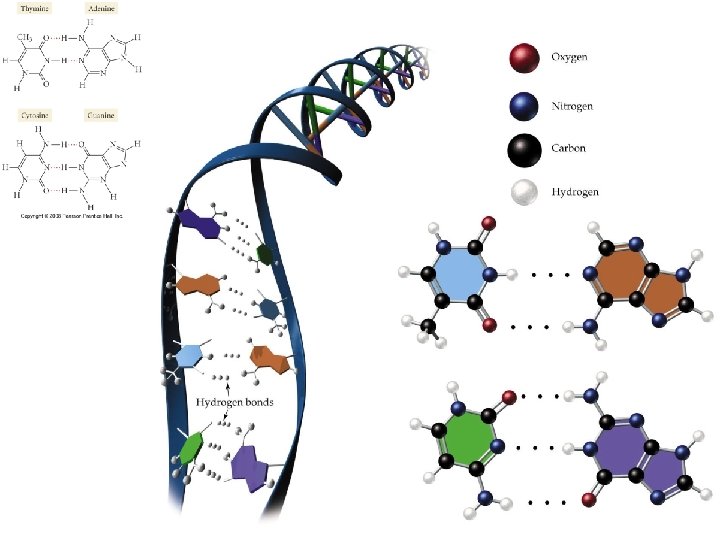
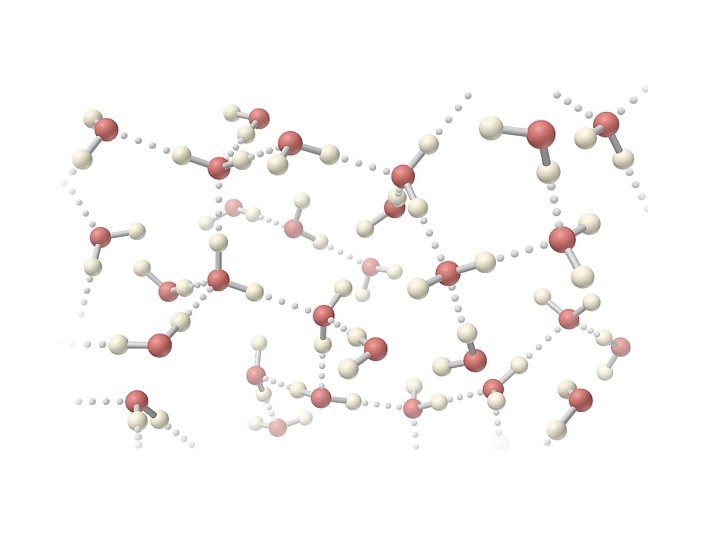
Hydrogen Bonds • A special type of polar interaction between a hydrogen atom bonded to an electronegative element and another electronegative element.








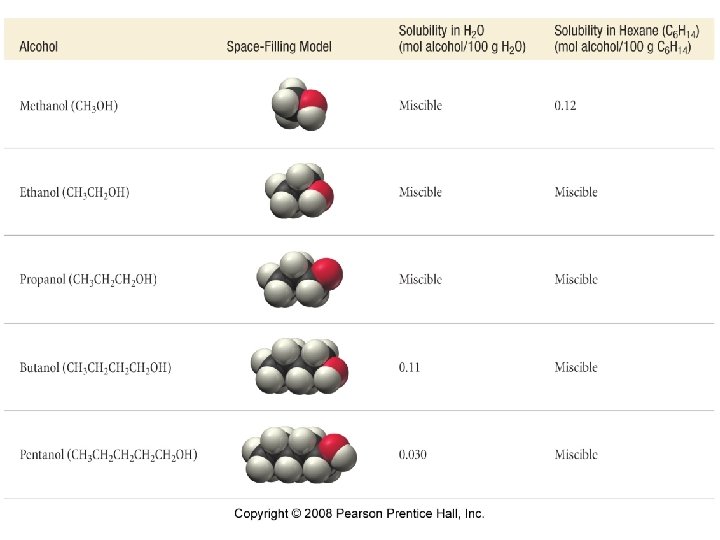
Solubility Like dissolves Like

Solubility • Polar solvents dissolve polar molecules • Nonpolar solvents dissolve nonpolar molecules • Molecules with polar and nonpolar ends are frequently soluble in both polar and nonpolar solvents. • Polar solvents are good for solubilizing salts.


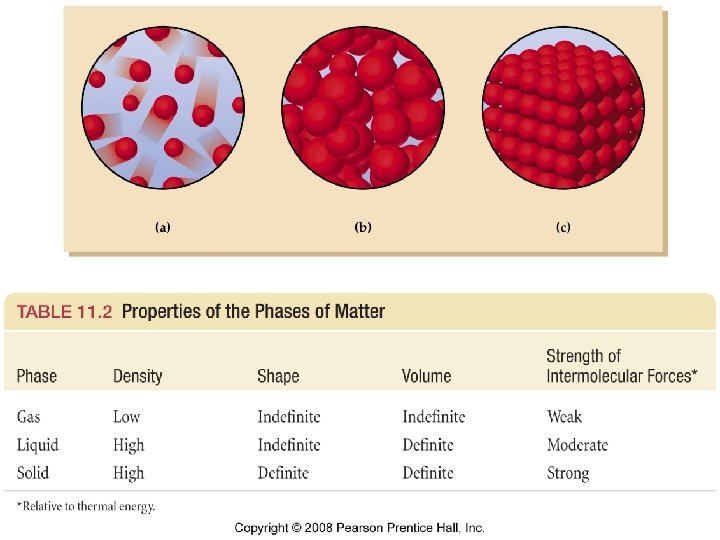
Liquids

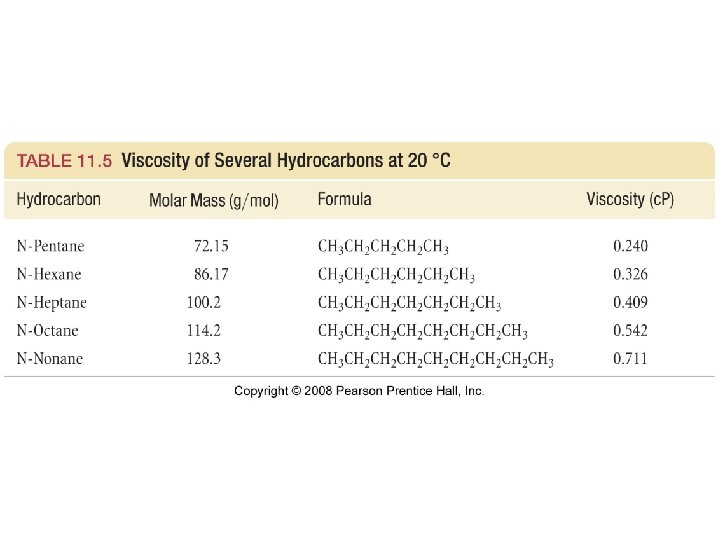
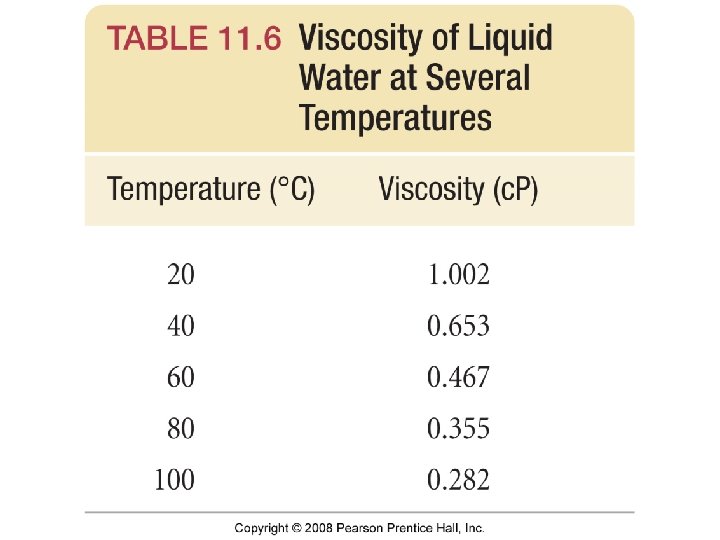
Viscosity • Resistance to flow • If a liquid has strong intermolecular interactions then particles will not flow past each other easily and viscosity will be high.



Surface Tension • tendency to minimize surface area

In Orbit (Space Shuttle), water droplets are spherical

• Cohesive Forces – attraction between molecules in a liquid • Adhesive Forces – attraction between liquid molecules and the surface of the tube • Water (red) has stronger adhesive forces. • Mercury has stronger cohesive forces.

Capillary Action • The ability of a liquid to flow against gravity up a narrow tube. Attraction of water to glass walls draws water up tubes

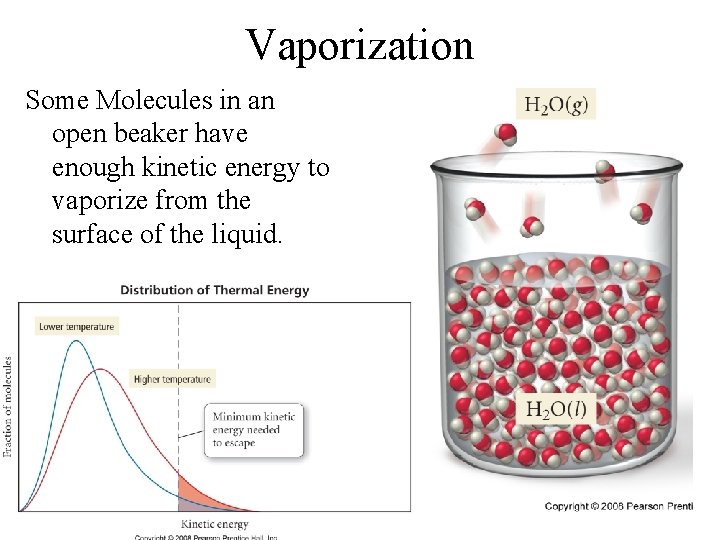
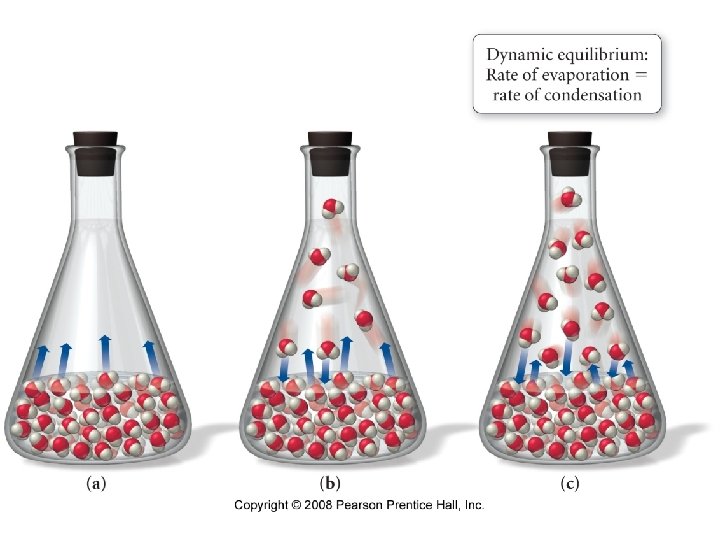
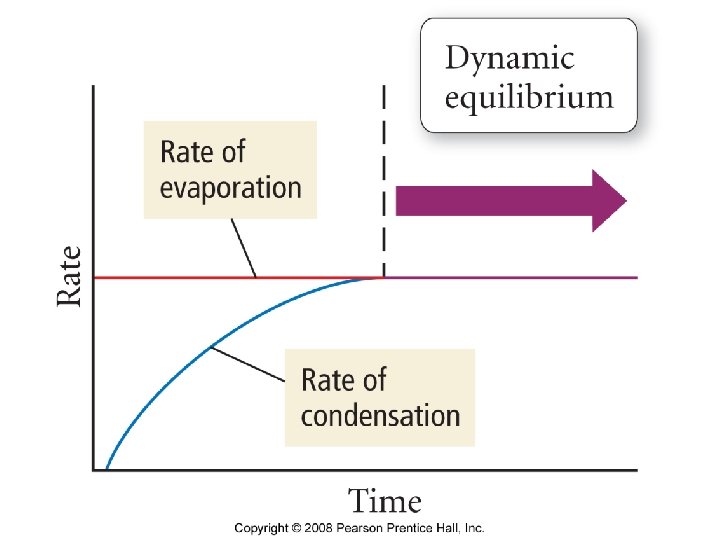
Vaporization Some Molecules in an open beaker have enough kinetic energy to vaporize from the surface of the liquid.



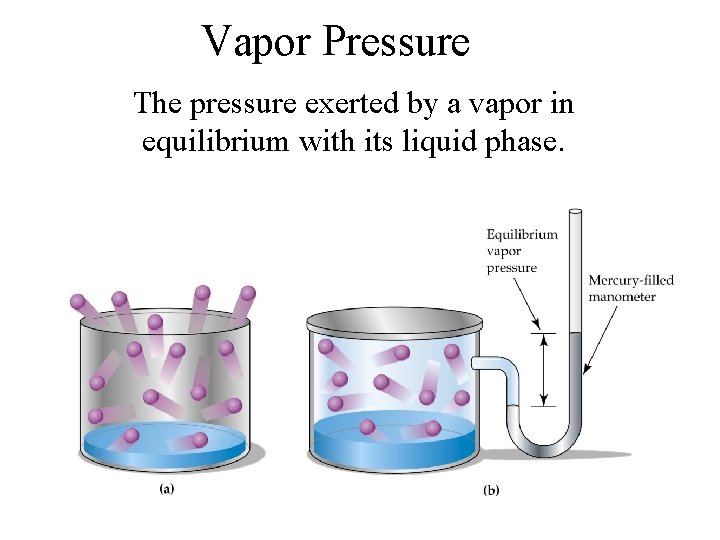
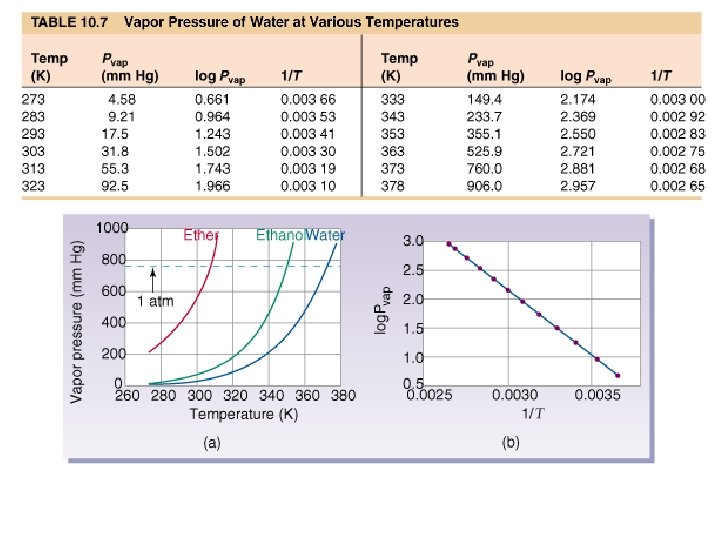
Vapor Pressure The pressure exerted by a vapor in equilibrium with its liquid phase.


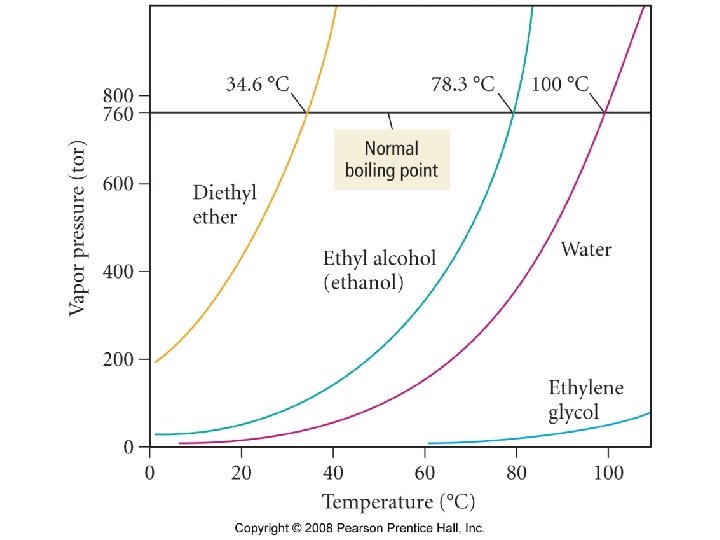
A liquid boils when thermal energy is high enough to cause molecules in the interior of the liquid to become gaseous, forming bubbles that rise to the surface.

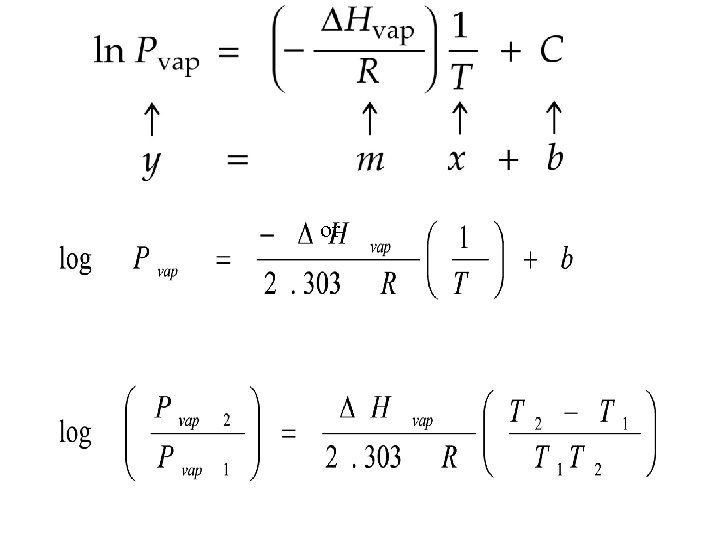
or

Clausius Clapeyron Equation or

Dry ice sublimes at – 78 o. C and has a Hsub of 25. 2 k. J/mol. Calculate the vapor pressure of o CO 2 at – 100 C.


• Calculate the boiling point of water at the summit of Pikes Peak in Colorado where the atmospheric pressure is 447.


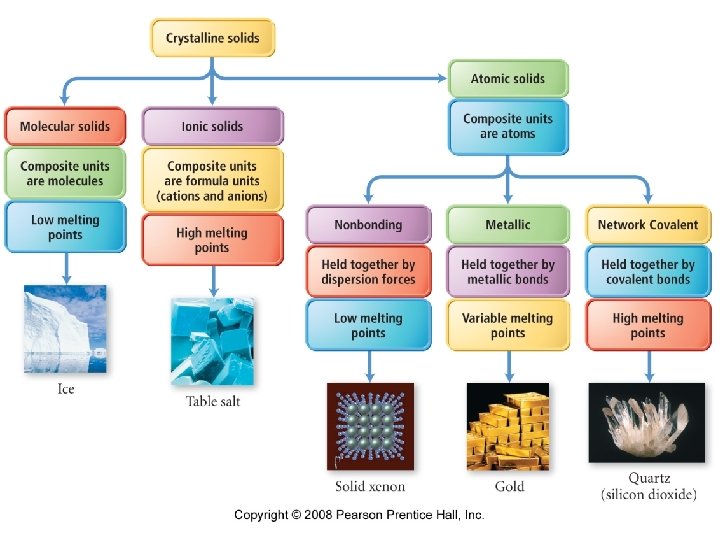
• Crystalline solid – atoms, ions, or molecules lie in an orderly array · typically have flat well defined surfaces called faces. • Amorphous solid – atoms or molecules lie in random jumble.


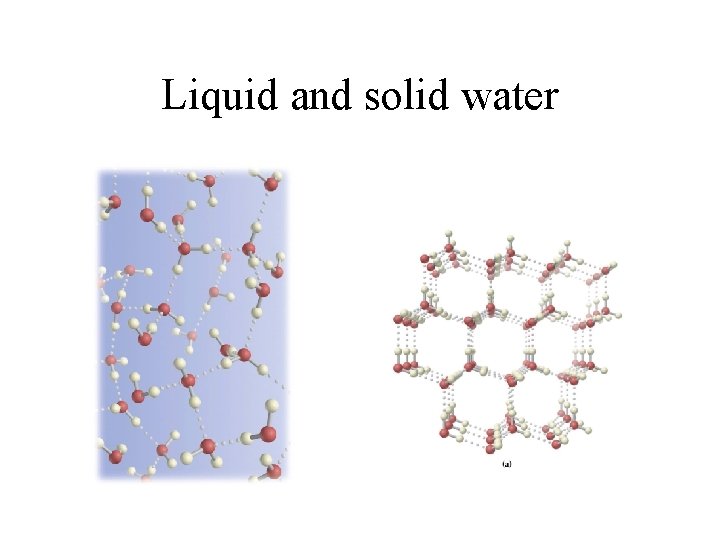
Liquid and solid water

Carbon allotropes






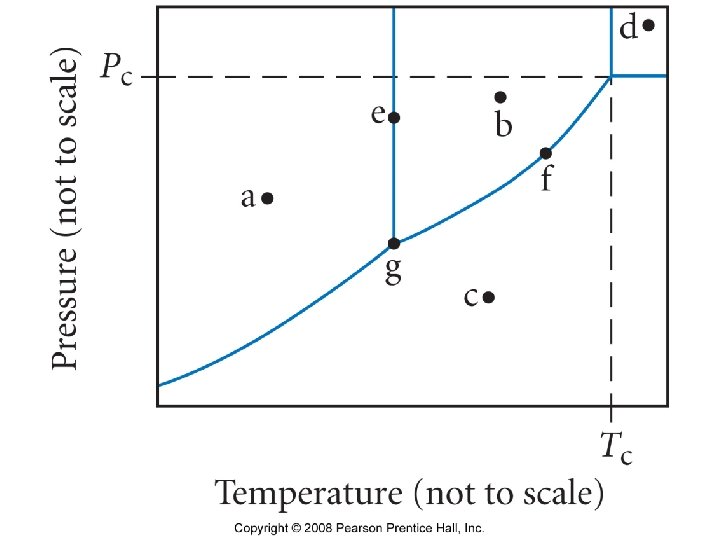
Phase Diagrams • Graphically show conditions under which all phases are stable.

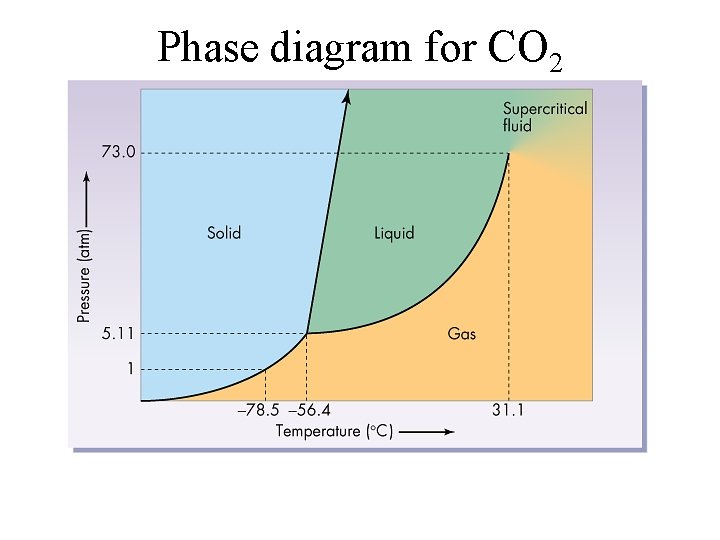
Phase diagram for CO 2

Triple point • A three-way intersection representing the unique temp, pressure where all three phases exist simultaneously.

Critical point • The temperature (critical temperature) where a gas cannot be liquefied no matter what the pressure. · Notice that the triple point for carbon dioxide is at 5. 11 atm pressure. The liquid form doesn’t ever exist below this pressure and this is the reason that dry ice never melts but always sublimes.

Supercritical fluid • Neither a liquid nor a gas. The liquid and gas forms become indistinguishable at this point. • http: //www. nottingham. ac. uk/supercritical/s cintro. html

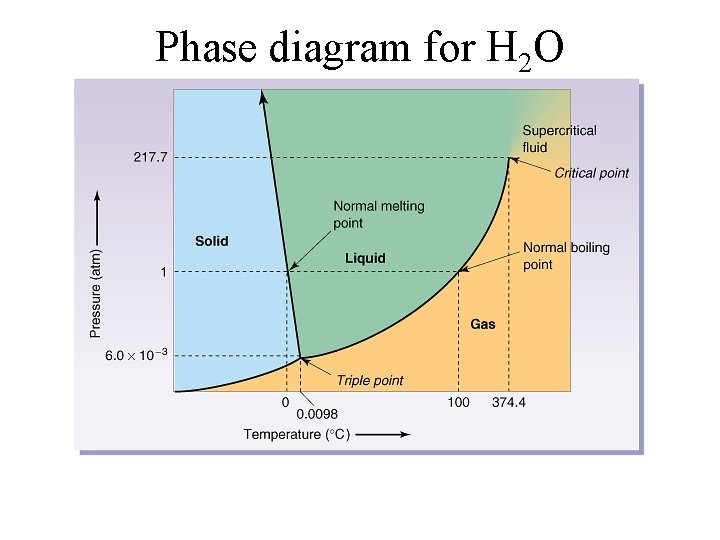
Phase diagram for H 2 O


Water • colorless, odorless, tasteless, liquid at ordinary temperatures • only inorganic compound occurring naturally as a liquid • composes � 65% of mass of living organisms • excellent solvent for many things • abnormally high boiling and melting point • ice is less dense than water (it floats)


Water purification • Hard water -- Contains Ca+2, Mg+2, Fe+3 and other minerals. • Soft water -- Doesn’t contain Ca+2, Mg+2, Fe+3 ions. • Softened water -- metal cations in hard water are replaced by Na+. • Deionized water -- cations are replaced by H+ and anions are replaced by OH-