LiquidLiquid Equilibrium Ternary System Most practical situations involving

Liquid-Liquid Equilibrium Ø Ternary System Ø Most practical situations involving liquid-liquid equilibrium involve three or more components. Ø Our attention is with three component systems. In this process, a solute is removed from a feed stream by contacting it with a solvent. Ø The solute is quite soluble in the solvent, while the other component in the feed is less soluble. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Liquid-Liquid Equilibrium Ø Terminology Ø Solute ≡ Component (1) Ø Original solvent ≡ Component (2) Ø Extractive solvent ≡ Component (3) Ø x 1 S, x 2 S and x 3 S are the composition of the three components in (solvent rich phase) 1, 2, 3 respectively. Ø x 1 R, x 2 R and x 3 R are the composition of the Three components in the (raffinate phase) 1, 2, 3 respectively. Feed (component +original solvent) Raffinate-rich phase (x 1 R 1, x 2 R 1 , x 3 R 1) solvent-rich phase Extractive solvent Ch. E 334: Separation Processes (x 1 S 1, x 2 S 1 , x 3 S 1) Dr Saad Al-Shahrani

Liquid-Liquid Equilibrium Ø The solvent phase is rich in solvent and soaks up component 1 (the solute), which we are trying to separate from the other component in the feed (component 2, raffinate). Ø The raffinate phase is the liquid phase which is rich in the component 2 (raffinate) and from which the solute (component 1) is being removed. Ø The original feed is usually a mixture of solute (component 1) and raffinate (component 2). Ø The solvent-rich phase contains mostly solvent (component 3) and solute (component 1) and only a small amount of raffinate (component 2) Ø The raffinate-rich phase contains mostly solute (component 1) and raffinate (component 2), but also possibly some small amount of solvent. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
Liquid-Liquid Equilibrium Ch. E 334: Separation Processes Dr Saad Al-Shahrani
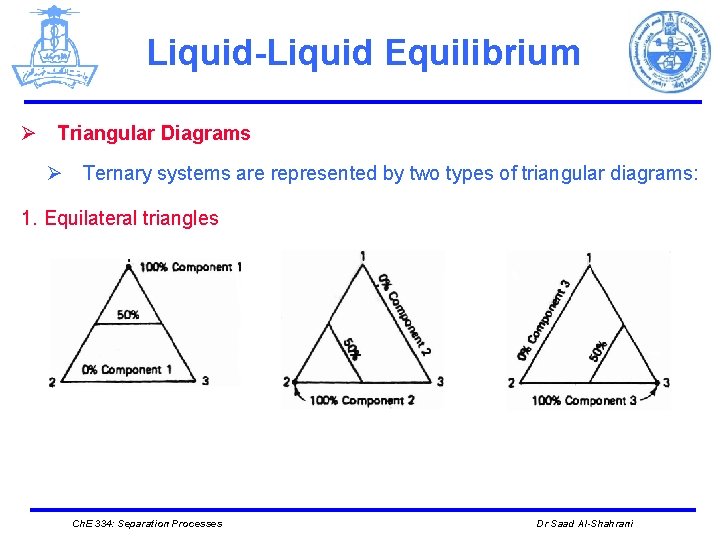
Liquid-Liquid Equilibrium Ø Triangular Diagrams Ø Ternary systems are represented by two types of triangular diagrams: 1. Equilateral triangles Ch. E 334: Separation Processes Dr Saad Al-Shahrani
Liquid-Liquid Equilibrium 2. Right Triangles Ch. E 334: Separation Processes Dr Saad Al-Shahrani
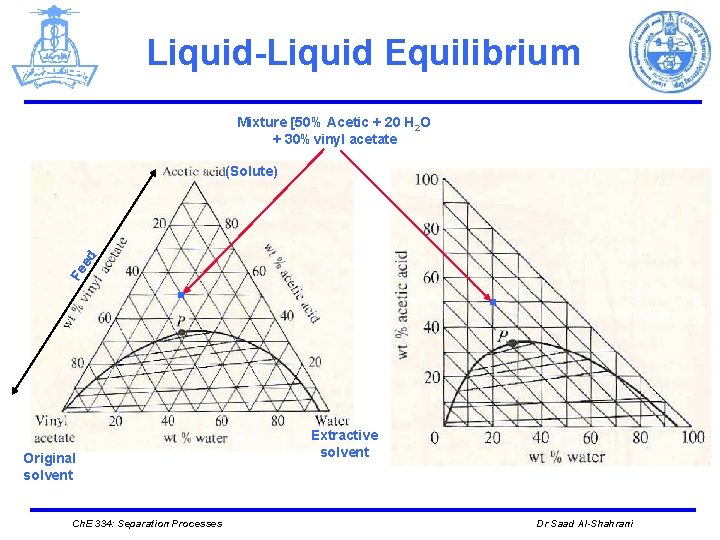
Liquid-Liquid Equilibrium Mixture [50% Acetic + 20 H 2 O + 30%vinyl acetate Fe ed (Solute) . Original solvent Ch. E 334: Separation Processes . Extractive solvent Dr Saad Al-Shahrani
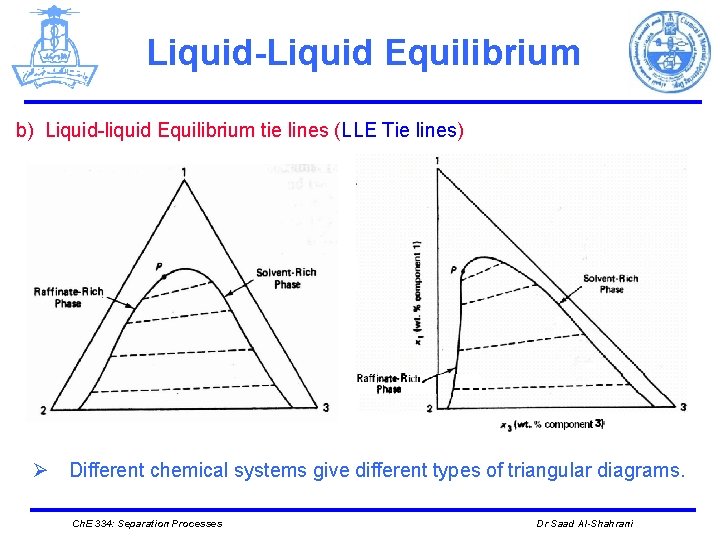
Liquid-Liquid Equilibrium b) Liquid-liquid Equilibrium tie lines (LLE Tie lines) Ø Different chemical systems give different types of triangular diagrams. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
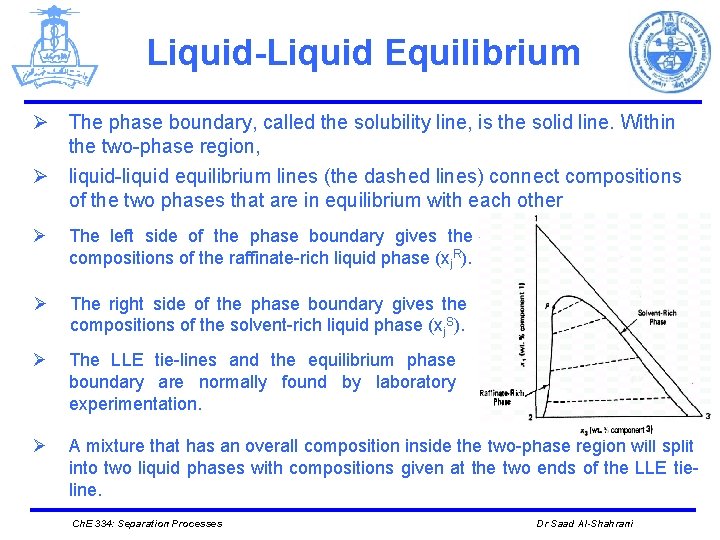
Liquid-Liquid Equilibrium Ø The phase boundary, called the solubility line, is the solid line. Within the two-phase region, Ø liquid-liquid equilibrium lines (the dashed lines) connect compositions of the two phases that are in equilibrium with each other Ø The left side of the phase boundary gives the compositions of the raffinate-rich liquid phase (xj. R). Ø The right side of the phase boundary gives the compositions of the solvent-rich liquid phase (xj. S). Ø The LLE tie-lines and the equilibrium phase boundary are normally found by laboratory experimentation. Ø A mixture that has an overall composition inside the two-phase region will split into two liquid phases with compositions given at the two ends of the LLE tieline. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
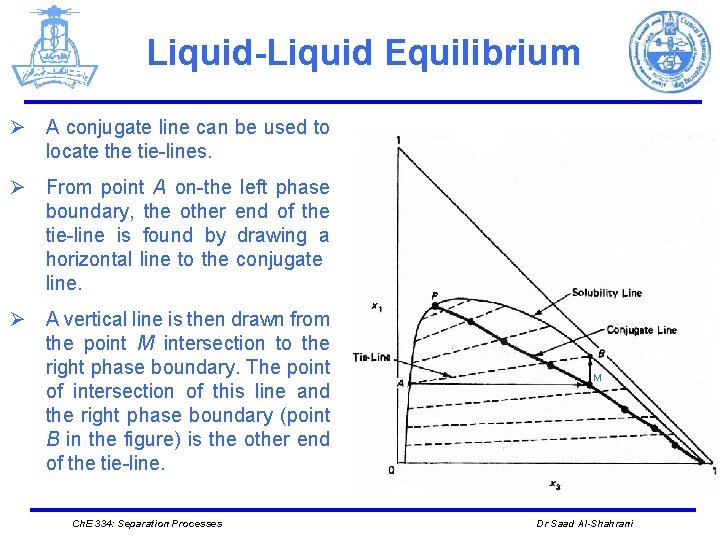
Liquid-Liquid Equilibrium Ø A conjugate line can be used to locate the tie-lines. Ø From point A on-the left phase boundary, the other end of the tie-line is found by drawing a horizontal line to the conjugate line. Ø A vertical line is then drawn from the point M intersection to the right phase boundary. The point of intersection of this line and the right phase boundary (point B in the figure) is the other end of the tie-line. Ch. E 334: Separation Processes M Dr Saad Al-Shahrani

Liquid-Liquid Equilibrium Ø As the system becomes richer in solute, the tie-lines get shorter and ultimately become just a point at the plait point P. Outside the twophase region, a single, homogeneous liquid phase exists. Ø Effect of Temperature on solubility Ø Usually, the solubility increases as the temperature increases, for this reason, most liquid-liquid extraction systems operate at low temperatures and some times even require refrigeration. Ø Pressure, on the other hand, has little effect on solubility. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Liquid-Liquid Equilibrium Ø if we specify only one concentration of one liquid phase, all the other concentrations can be immediately determined from the phase diagram Ø For example, if we fix the concentration of component 1 in the raffinate -rich phase (x 1 R), we can read from the diagram: 1. The concentration of component 3 in the raffinate-rich phase (x 3 R), by using the left side of the solubility curve. 2. The concentrations of components 1 and 3 in the solvent-rich phase that is in equilibrium with the raffinate-rich phase, by going to the other end of the LLE tie-line. The concentrations x 1 S and x 3 S are read from the right side of the solubility curve. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
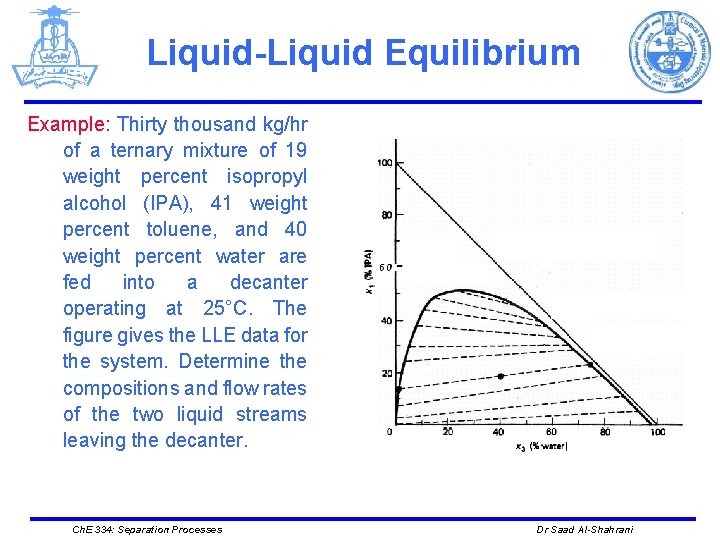
Liquid-Liquid Equilibrium Example: Thirty thousand kg/hr of a ternary mixture of 19 weight percent isopropyl alcohol (IPA), 41 weight percent toluene, and 40 weight percent water are fed into a decanter operating at 25°C. The figure gives the LLE data for the system. Determine the compositions and flow rates of the two liquid streams leaving the decanter. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
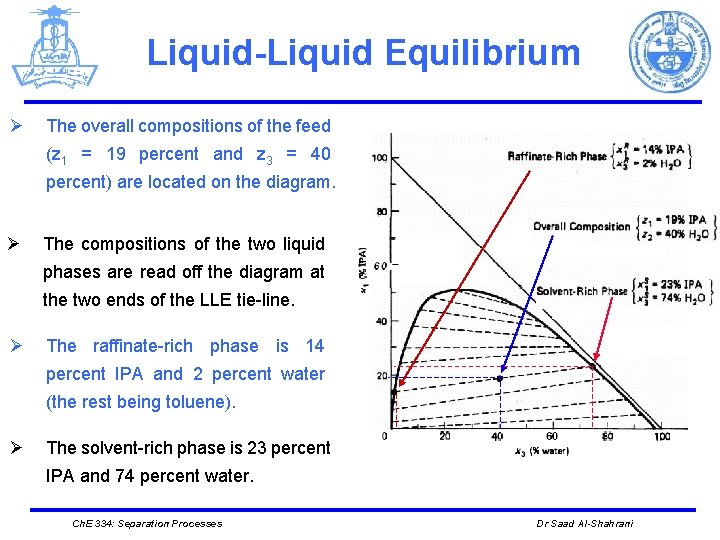
Liquid-Liquid Equilibrium Ø The overall compositions of the feed (z 1 = 19 percent and z 3 = 40 percent) are located on the diagram. Ø The compositions of the two liquid phases are read off the diagram at the two ends of the LLE tie-line. Ø The raffinate-rich phase is 14 percent IPA and 2 percent water (the rest being toluene). Ø The solvent-rich phase is 23 percent IPA and 74 percent water. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Liquid-Liquid Equilibrium Total mass: 30000 = S + R Water = (30000)(0. 4) = S(0. 74) + R(0. 02) Solving the last two equations simultaneously gives S = 15833 kg/h R = 14176 kg/h IPA in = (30000)(0. 19) = 5700 kg/h IPA out = S(0. 23) + R(0. 14) = (15833)(0. 23) + (14176)(0. 14) = 5625 kg/h The difference is due to the accuracy of reading composition from the diagram Ch. E 334: Separation Processes Dr Saad Al-Shahrani

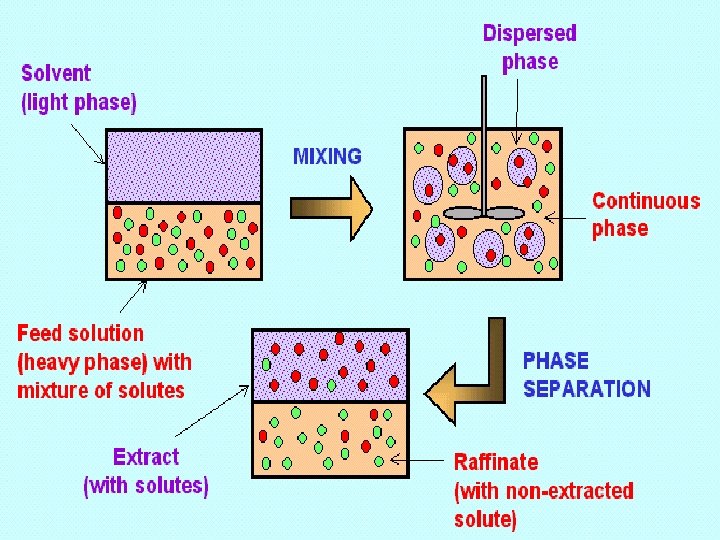
Liquid-Liquid Extraction Ø In liquid-liquid extraction, a liquid of two or more components to be separated is contacted with a second liquid phase, called the solvent, which is immiscible or partially miscible with one or more components of the liquid feed. Ø The simplest liquid-liquid extraction involves only a ternary system. The feed consists of two miscible components, the carrier (C) and the solute (A). Solvent (S) is a pure component. Components (C, S) are at most only partially soluble in each other. Solute (A) is soluble in (C) and completely or partially soluble in S. Ø During the extraction process, mass transfer of (A) from the feed to the solvent occurs, with less transfer of (C) to the solvent, or (S) to the feed. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Liquid-Liquid Extraction Ø Liquid-liquid extraction is used to separate components in situations where: 1. Relative volatilities are quite close to unity ( < 1. 1), making distillation very costly. (Distillation requires tall towers due to the existence of many trays, and high energy consumption because of high reflux ratios. ) e. g. are 80. 1°C and 80. 7°C, respectively, making their separation by distillation impractical 2. Thermally sensitive components will not permit high enough temperatures to produce a vapor-liquid system at reasonable pressures (pressures greater than 10 -50 mm Hg). Ch. E 334: Separation Processes Dr Saad Al-Shahrani

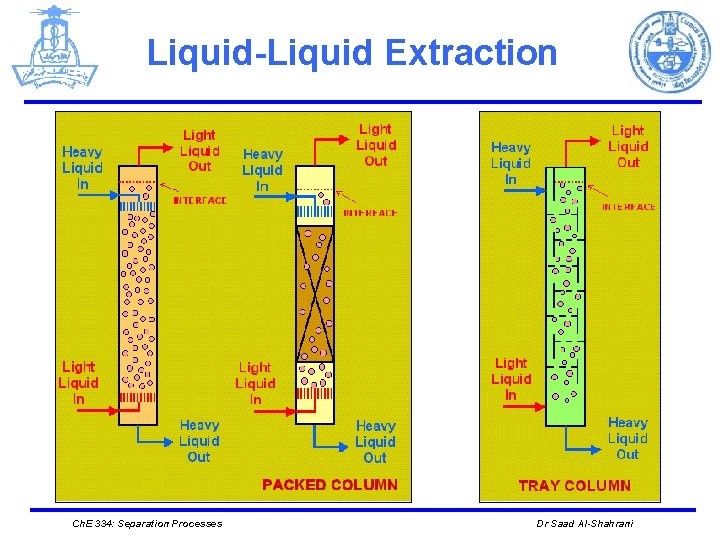
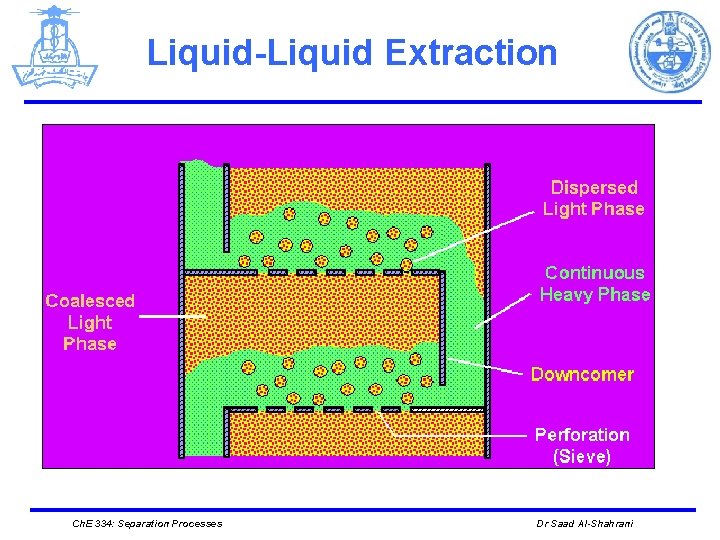
Liquid-Liquid Extraction Ø EQUIPMENT Ø Different mechanical devices are used in liquid-liquid extraction such as: 1. The simplest is a mixer/settler, or decanter, in which the two liquid phases are separated. 2. Plate towers, packed towers, and mechanically agitated mixers (rotating disk contactors) Ø The number of stages tends to be much smaller than in distillation columns. This is due to the larger settling times required for liquid-liquid separation because of the small density differences between the liquid phases. Ø Liquid-liquid extraction columns are sometimes operated in a pulsed mode. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
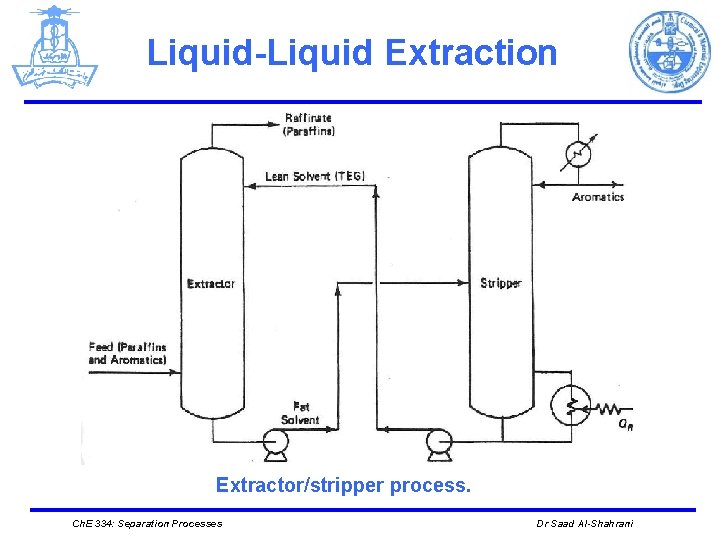
Liquid-Liquid Extraction Extractor/stripper process. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
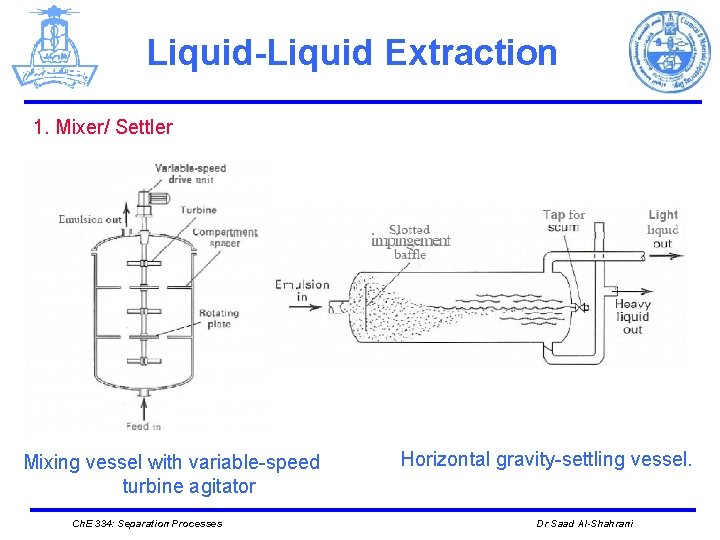
Liquid-Liquid Extraction 1. Mixer/ Settler Mixing vessel with variable-speed turbine agitator Ch. E 334: Separation Processes Horizontal gravity-settling vessel. Dr Saad Al-Shahrani
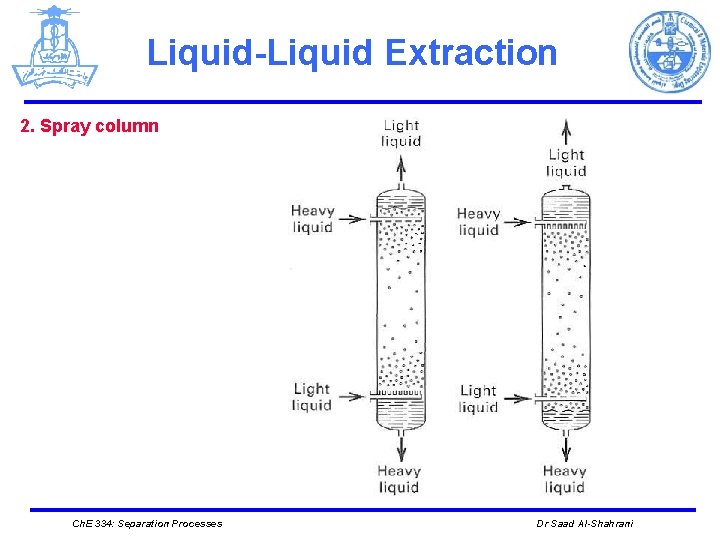
Liquid-Liquid Extraction 2. Spray column Ch. E 334: Separation Processes Dr Saad Al-Shahrani
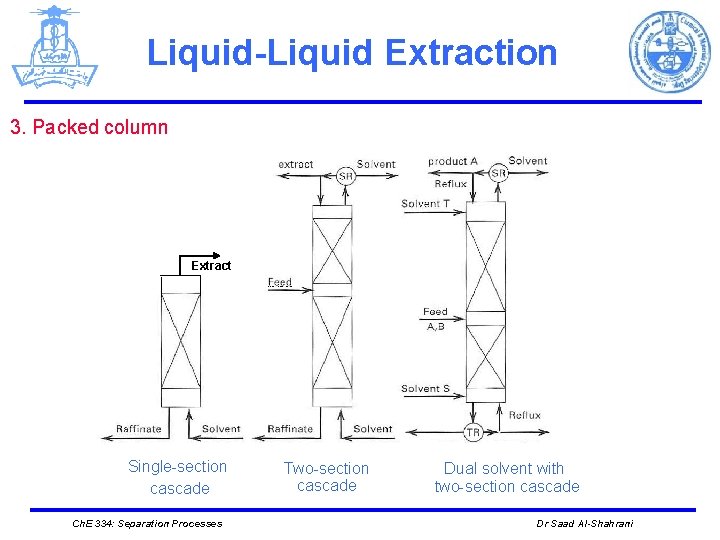
Liquid-Liquid Extraction 3. Packed column Extract Single-section cascade Ch. E 334: Separation Processes Two-section cascade Dual solvent with two-section cascade Dr Saad Al-Shahrani
Liquid-Liquid Extraction Ch. E 334: Separation Processes Dr Saad Al-Shahrani
Liquid-Liquid Extraction Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Liquid-Liquid Extraction Ø GRAPHICAL MIXING RULES If we have two streams that contain three components and mix them together. Let one of these streams be stream A with flow rate FA (kg/h) and composition x 1 A, x 2 A and x 3 A (weight fractions of components 1, 2, and 3), and let the other be stream FB with corresponding composition x 1 B, x 2 B and x 3 B. The mixed stream leaving the mixer will have a flow rate FM and composition x 1 M, x 2 M and x 3 M. A flow diagram is as follows: FA x 1 A, x 2 A , x 3 A FM x 1 M, x 2 M , x 3 M FB x 1 B, x 2 B , x 3 B Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Liquid-Liquid Extraction Ø To determine the location of the mixture composition on a graph, since there are three components, only two coordinates are needed to completely specify the composition of any stream. We can use either right or equilateral triangular plots. Ø If we use right-triangular plot. locate point A with coordinates (x 1 A, x 2 A ) and point B with coordinates (x 1 B, x 2 B). The point M with coordinates (x 1 M, x 2 M ) representing the mixture will lie some place on the graph. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Liquid-Liquid Extraction Ø After mixing point M is supposed to lie on a straight line joining the A and B points. If we can show that the angles and in the figure are equal, then M must lie on a straight line between A and B. Ø The total mass balance for the system is (1) Ø Component balances for components 1 and 2 are (2) and Ch. E 334: Separation Processes (3) Dr Saad Al-Shahrani

Liquid-Liquid Extraction Ø Rearranging these two equations, we obtain: Ø Solving for the ratio FAIFB, we have: or Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Liquid-Liquid Extraction Ø These two ratios are the tangents of the angles and , hence, tan = tan . Therefore, = , and we have proven that the line AMB is a straight line. Ø The coordinates of the point M can be solved for analytically by using equations (1), (2), and (3). Alternatively, M can be located graphically where the distance from the point A to the point M divided by the distance from the point M to the point B is equal to the ratio FB/FA. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
- Slides: 29