LIQUID DOSAGE FORM T N Saifullah S Laboratorium

LIQUID DOSAGE FORM T N Saifullah S Laboratorium Teknologi Farmasi Fakultas Farmasi UGM

Difinition • Oral Liquids are homogeneous liquid preparations, usually consisting of a solution, an emulsion or a suspension of one or more medicaments in a suitable vehicle. • All liquid dosage forms are dispersed systems in which medical substance (the internal phase) is dispersed uniformly though-out the external phase. Medical substances in liquid dosage form can be in the different aggregate states: solid, liquid and gas

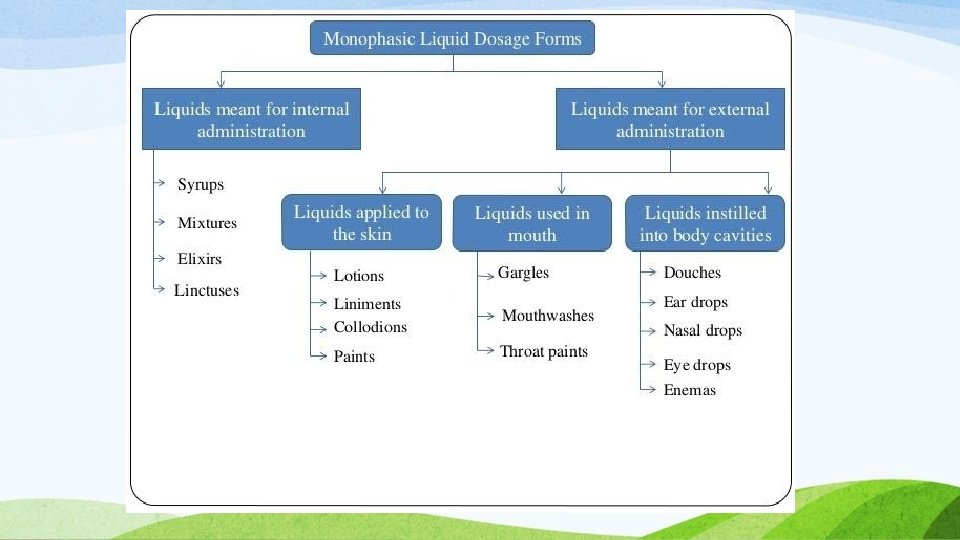
CLASSIFICATION OF LIQUID ORALS Two main types: 1. Monophasic liquids: Solutions Suspensions Elixirs Emulsions Syrup Liquid drops …etc 3 2. Biphasic liquids:


Solution is a homogeneous mixture composed of two or more substances. In mixture, solute is dissolved in another substance, known as a solvent.

Liquid dosage forms for internal use name usually mixtures (from the Latin word of mіxturae - to mix up), the disperse phase - water only. They are measured out to the patients independently by spoons (table (soup)-spoon - 15 ml, dessert-spoon - 10 ml, tea-spoon – 5 ml. ).

By composition By nature of solvent. • Simple- solutions which include only one substance. • Hydrogen's • Multiple- solutions which include a few ingredients. • non-aqueous (alcoholic, glycerin, oily).

Formulation consideration: Solvents Preservatives Antioxidants Solubilizers Organoleptic agents etc. 9

SOLVENTS • Water • Alcohol • Glycerol • Polyethylene glycol • Propylene glycol. 10

Water • Compared to ordinary drinking water , purified water USP is more free of solid impurities. • When evaporated to dryness, it must not yield greater than 0. 001% of residue. • Purified water is obtained by distillation, ion-exchange treatment, reverse osmosis and other relevant method 11

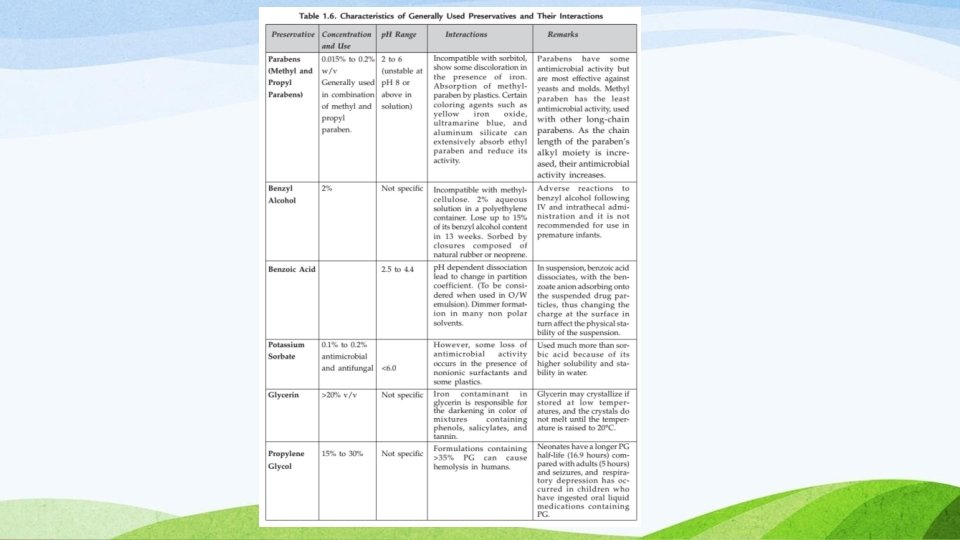
PRESERVATIVES • Preservatives are added to prevent the microbial growth • Preservative are necessary due to chances of microbial growth • Raw material, processing containers & equipments, the manufacturing environment, operators, packing materials & the user. • Phenol, chlorocresol, benzoic acid, etc. 12


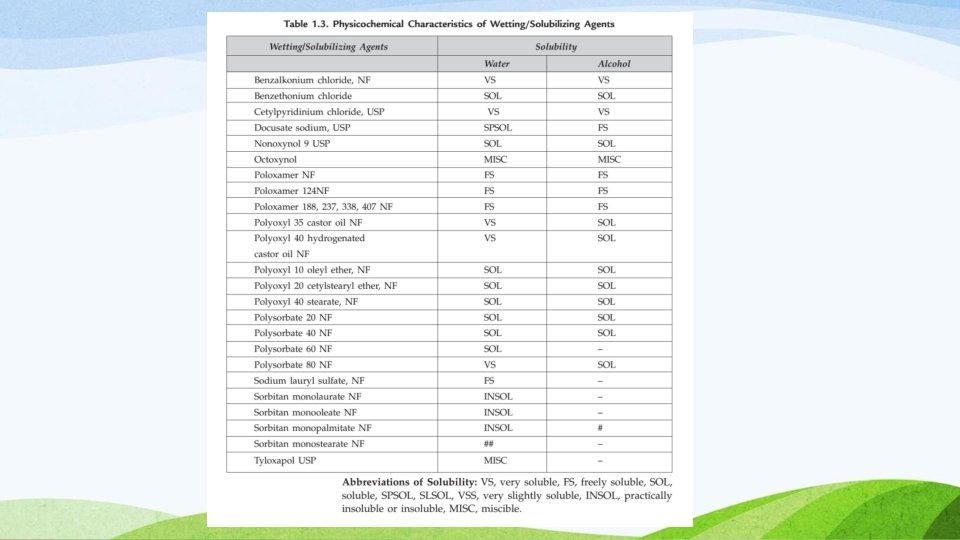
Solubilizers: Wetting Agents and Surfactants Wetting agents– are used in liquid dosage forms to create a homogenous dispersion of solid particles in a liquid vehicle. Wetting agents are Surfactants (HLB Value 7 to 9) that when dissolved in water, lower the contact angle and aid in spreadability of water on the particles surface to displace the air layer at the surface and help in wetting and solubilization 15

p. H Modifiers and Buffering Agents The p. H– of an oral liquid formulation is a key point for API stability, solubilization and also to prevent microbial contamination. Buffers are used to control p. H of liquid orals. Combination of buffers can also be used to gain a wider range of p. H compared to the individual buffer alone. Buffer selection should be judicious to avoid incompatibility.

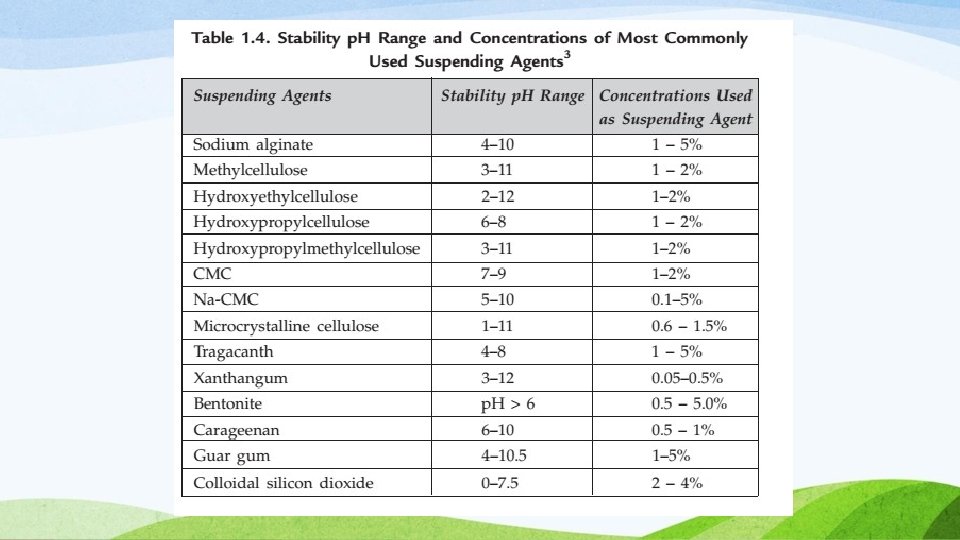
Suspending Agents and Viscosity-modifying Agents Suspending agents– impart viscosity, and thus retard particle sedimentation. Other factors considered in the selection of the appropriate agent include desired rheological property, suspending ability in the system, chemical compatibility with other excipients, p. H stability, length of time to hydrate, batch-to-batch reproducibility, and cost.

Antioxidants Oxidation– may manifest as products with an unpleasant odor, taste, appearance, precipitation, discoloration or even loss of activity. The oxidation of an API in an oral liquid formulation needs to be prevented using anti-oxidents. Antioxidants act as free radicle scavengers and terminator of chain reaction in auto oxidation

Organoleptic agents etc: colour, flavor, sweetening agents Sweetening • Sugar (sucrose) or sugar substitutes (artificial sweeteners) • One of the most popular is Sorbitol Solution USP, which contains 64% w/w sorbitol Flavor The flavours may be of natural origin (e. g. peppermint, lemon, herbs and spices) and are available as oils, extracts, spirits or aqueous solutions. Colours • These are generally natural or synthetic watersoluble, photo-stable ingredients that are selected according to the flavour of the preparation. • For example, mint-flavoured formulations are commonly a green colour, whereas in banana-flavoured solutions a yellow colour is commonly employed.

Stages of operations: 1. Tank selection • Material of the tank must not be additive to the product • The shape and size of equipment must be selected according to the batch size • The tanks are usually constructed of polished stainless steel of different grades • Teflon and glass lined tank. • Adequate clean-up procedures developed. 22

2. Mixing • Simple mixing is essential to increase flow of liquids. • If the liquid is of high viscosity, high electrical stirrer may be used. • Addition of ingredients in proper order have vital important. • At high viscosity the chance of air entrapment. 23

Air entrapment Minimize: • By reducing agitator speed • By caring out the mixing procedure in enclosed tank under vacuum • The alternative procedure to the all is versator 24

3. Dispersion • Suspensions and emulsions require considerably greater shear forces • Laboratory formulation is difficult to duplicate at large scale • Dispersion produced by colloidal mill or an immersion homogenizer • Variety of equipment should evaluated for better results. 25 Homogenizer Colloidal mill

4. Filtration and clarification— • Filtration procedure, requires careful evaluation to ensure that pilot scale-ups exhibit the same degree of clarity as their laboratory counterparts. • During the pilot run the clarity of the filtrate should be checked periodically, in order to establish schedule for changing pads, cake, or cartridges, depending on the type of filtration employed. • In filtration, filter pads are used which is made up of asbestos and cellulose. Selection of filtration depends on • The product viscosity • Volumes • Rate requirement 26 will

5. Transfer and filling • Filling – important parameter in the transfer of liquids from tank to tank and into containers. • New batches should not be started until the previous batches are completely filled and the tanks are emptied. 27

Methods for filling of liquids: • The selection of equipment depends on characteristic of liquid such as, viscosity, type of packaging, surface tension. • Gravimetric (specific weight) • Volumetric (specific volume) • Constant volume filling 28 Gravimetric Volumetric

Containers and closures: • Glass • Plastic • It is more important to store the final product in container until its expiration. • Most oral liquids are packed in either amber or flint glass containers with plastic or metal caps. 29
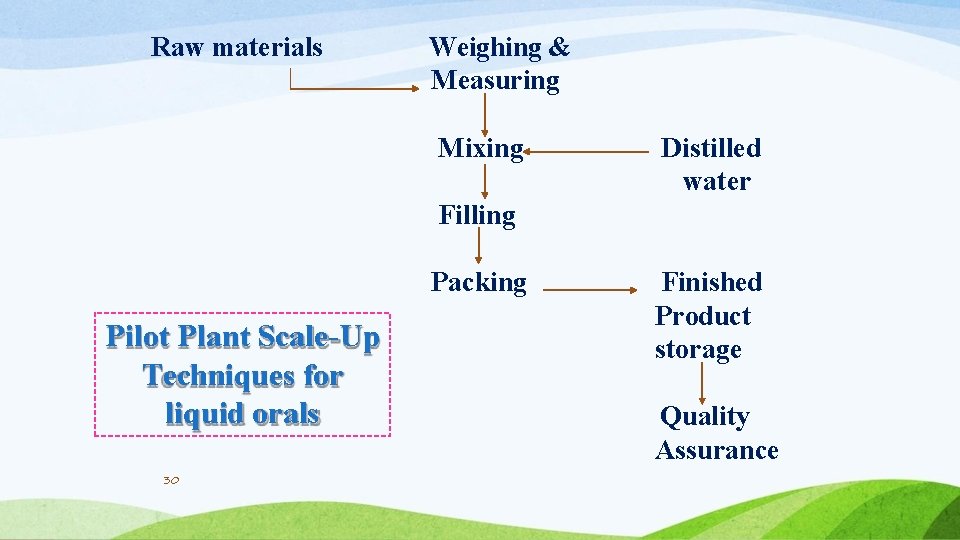
Raw materials Weighing & Measuring Mixing Distilled water Filling Packing Pilot Plant Scale-Up Techniques for liquid orals 30 Finished Product storage Quality Assurance
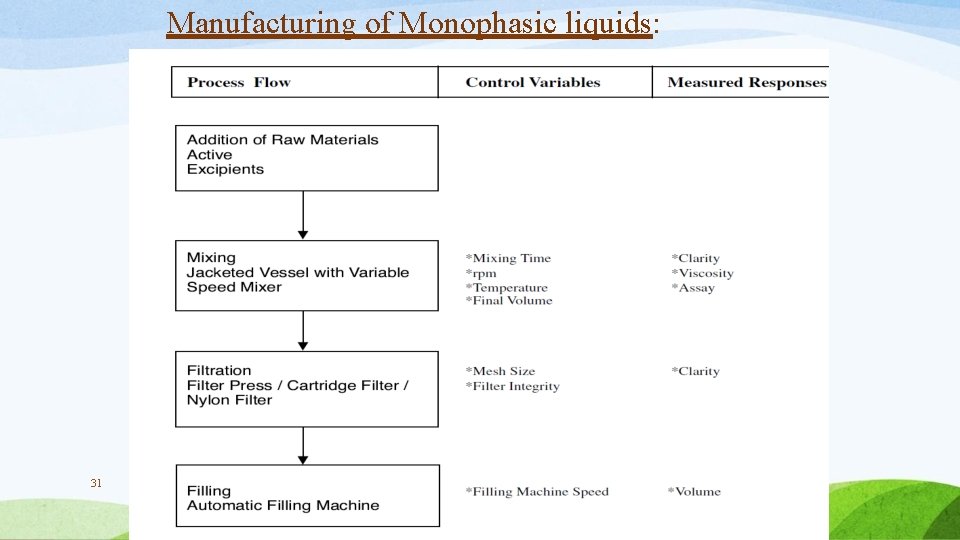
Manufacturing of Monophasic liquids: 31
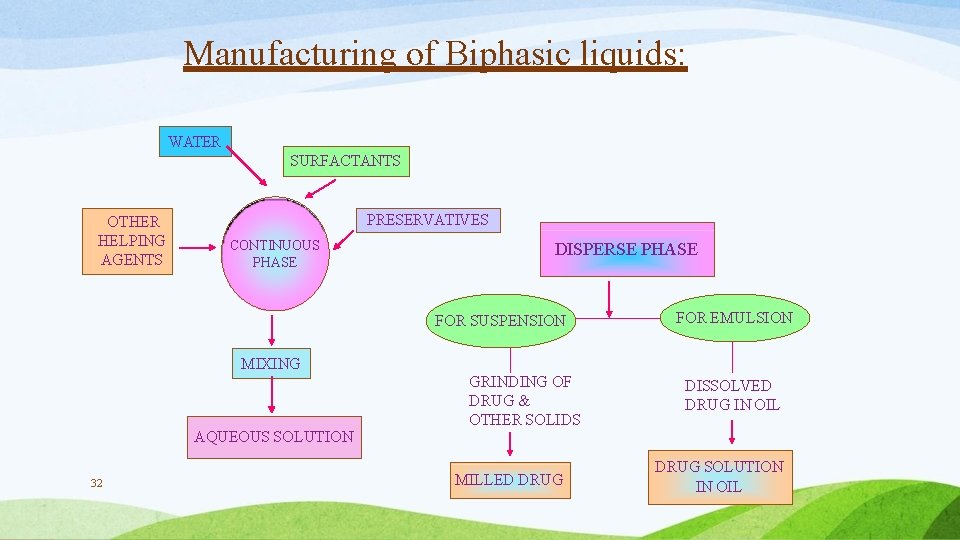
Manufacturing of Biphasic liquids: WATER SURFACTANTS OTHER HELPING AGENTS PRESERVATIVES CONTINUOUS PHASE DISPERSE PHASE FOR SUSPENSION FOR EMULSION MIXING AQUEOUS SOLUTION 32 GRINDING OF DRUG & OTHER SOLIDS MILLED DRUG DISSOLVED DRUG IN OIL DRUG SOLUTION IN OIL
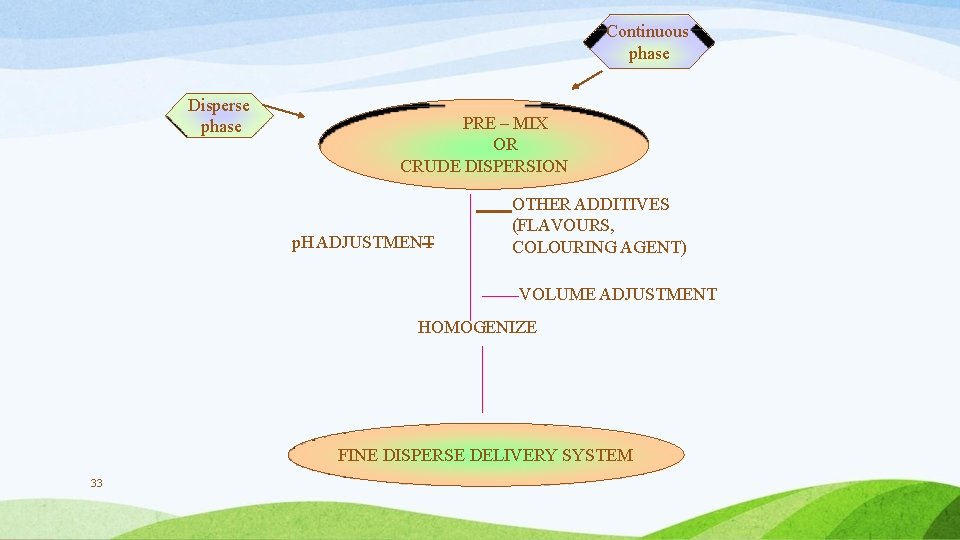
Continuous phase Disperse phase PRE – MIX OR CRUDE DISPERSION p. H ADJUSTMENT OTHER ADDITIVES (FLAVOURS, COLOURING AGENT) VOLUME ADJUSTMENT HOMOGENIZE FINE DISPERSE DELIVERY SYSTEM 33
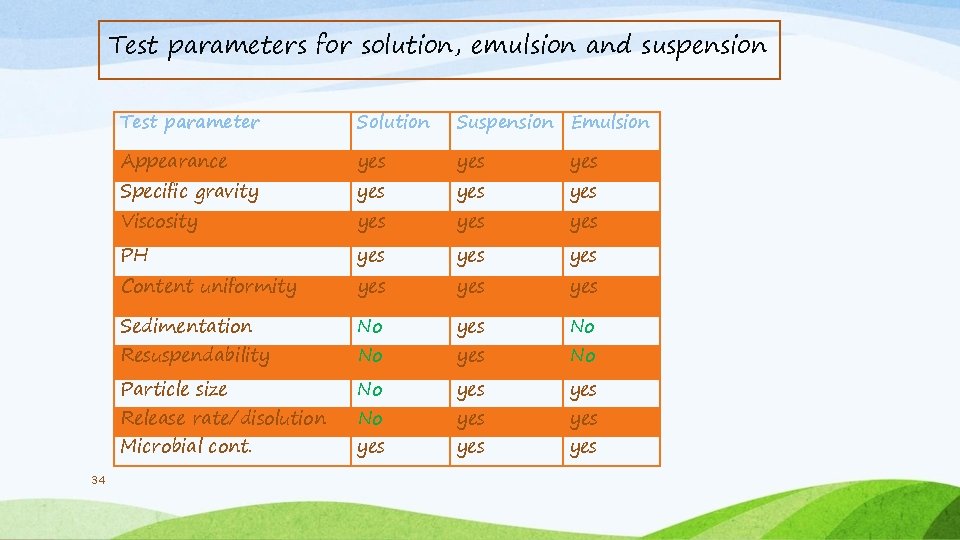
Test parameters for solution, emulsion and suspension Test parameter Solution Suspension Emulsion Appearance yes Specific gravity yes yes PH yes yes Sedimentation No yes No Particle size No yes Microbial cont. yes yes Viscosity Content uniformity Resuspendability Release rate/disolution 34 yes yes No No yes yes yes No yes


Storage area 36

Terimakasih tn. saifullah@gmail. com Hp: 08179421188 WA: 089501332718
- Slides: 37