Liquid Crystal Materials Broad Classification Lyotropics Thermotropics amphiphilic
Liquid Crystal Materials
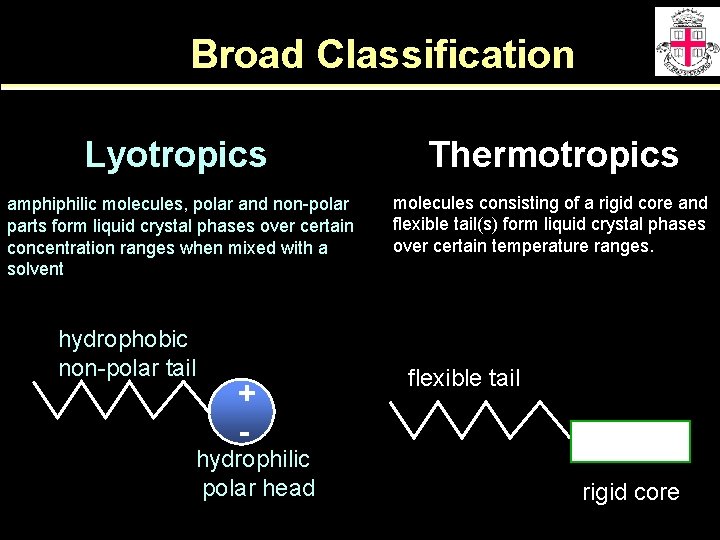
Broad Classification Lyotropics Thermotropics amphiphilic molecules, polar and non-polar parts form liquid crystal phases over certain concentration ranges when mixed with a solvent molecules consisting of a rigid core and flexible tail(s) form liquid crystal phases over certain temperature ranges. hydrophobic non-polar tail + - hydrophilic polar head flexible tail rigid core
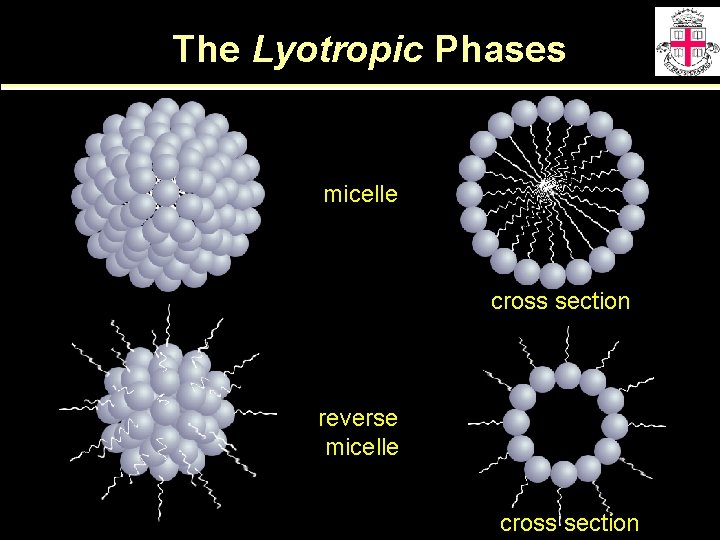
The Lyotropic Phases micelle cross section reverse micelle cross section
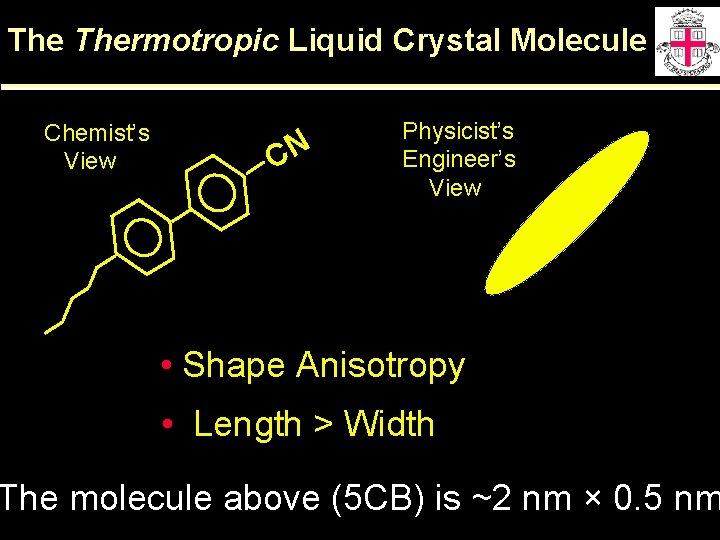
The Thermotropic Liquid Crystal Molecule Chemist’s View CN Physicist’s Engineer’s View • Shape Anisotropy • Length > Width The molecule above (5 CB) is ~2 nm × 0. 5 nm
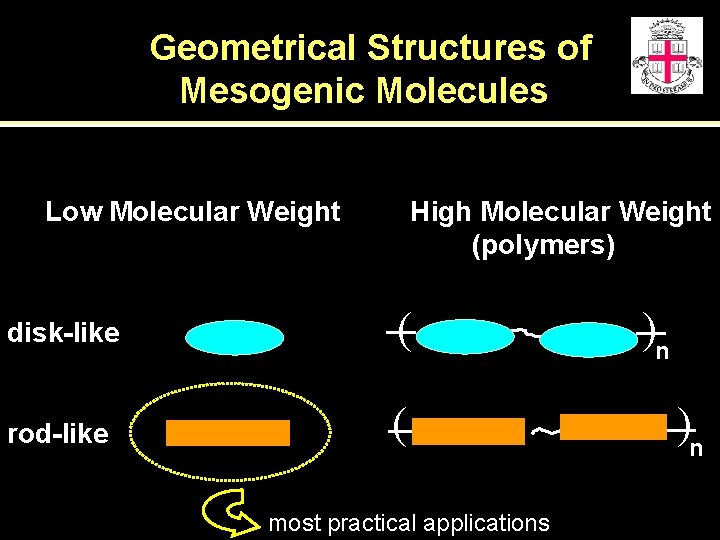
Geometrical Structures of Mesogenic Molecules Low Molecular Weight High Molecular Weight (polymers) disk-like ( rod-like ( most practical applications )n )n
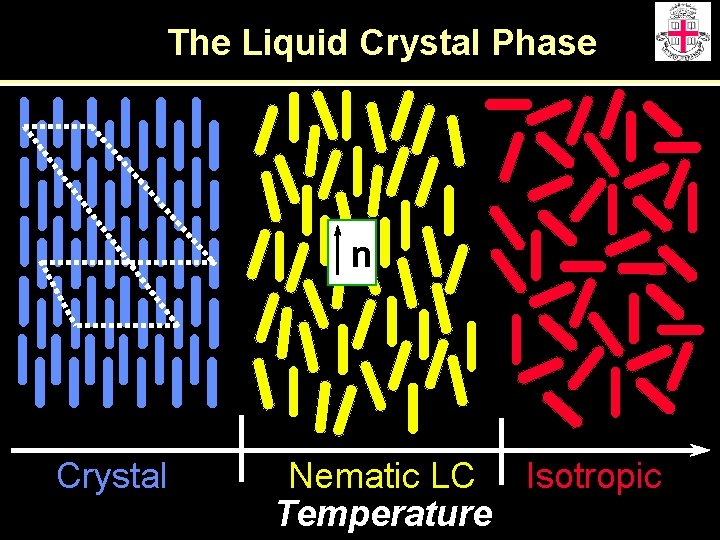
The Liquid Crystal Phase n Crystal Nematic LC Isotropic Temperature
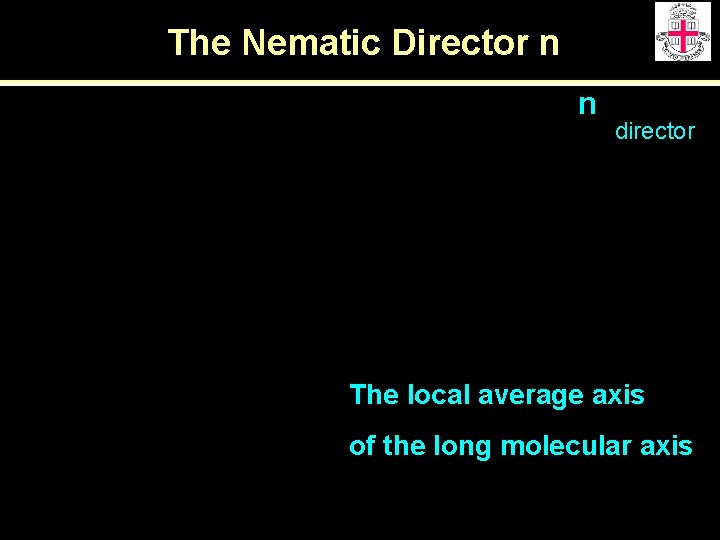
The Nematic Director n n director The local average axis of the long molecular axis
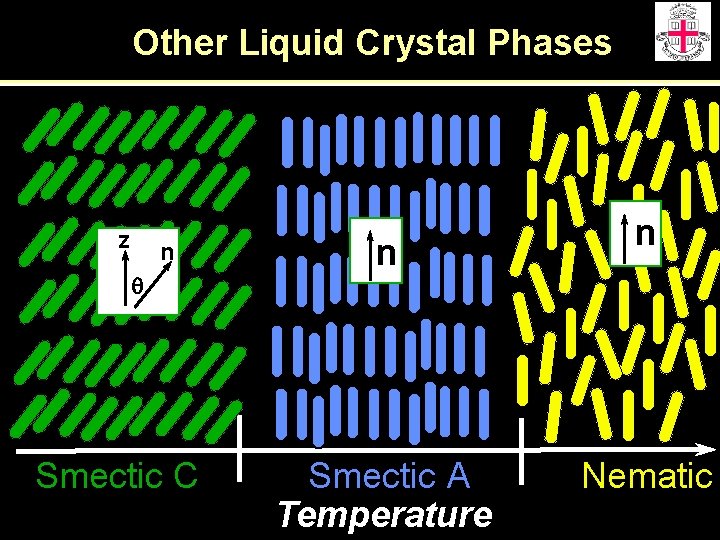
Other Liquid Crystal Phases z n q Smectic C n Smectic A Temperature n Nematic
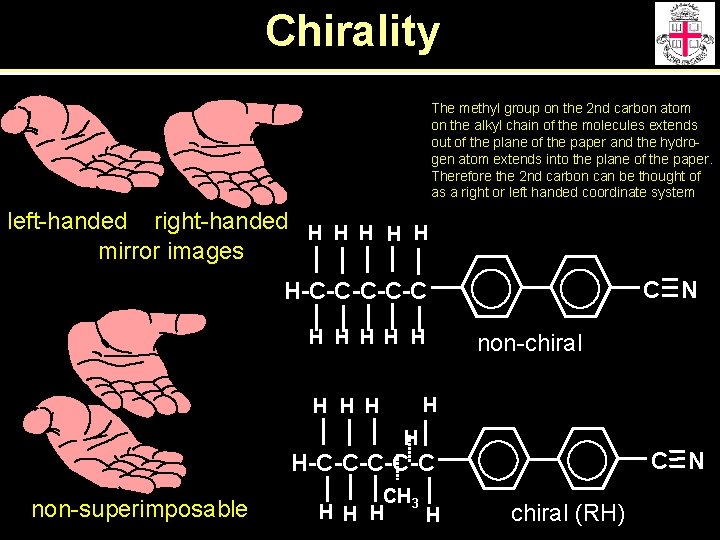
Chirality The methyl group on the 2 nd carbon atom on the alkyl chain of the molecules extends out of the plane of the paper and the hydrogen atom extends into the plane of the paper. Therefore the 2 nd carbon can be thought of as a right or left handed coordinate system left-handed right-handed H H H mirror images C N H-C-C-C H H H non-chiral H H H C N H-C-C-C non-superimposable CH 3 H H chiral (RH)
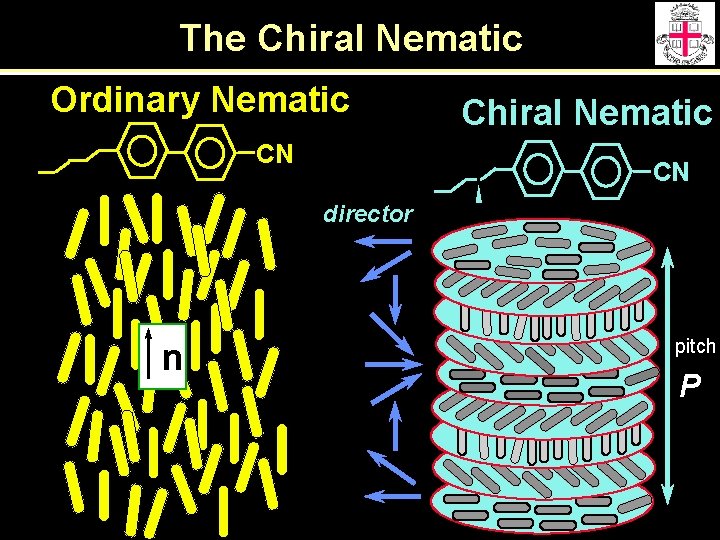
The Chiral Nematic Ordinary Nematic CN Chiral Nematic CN director n pitch P

The Chiral Doped Nematic You can create a cholesteric material by doping a conventional nematic with a chiral dopant. For dilute solutions Chiral Dopant S-811 IS-4651 HTP (mm)-1 -14 -13. 6 - indicates left twist sense For a 10% doping of S-811
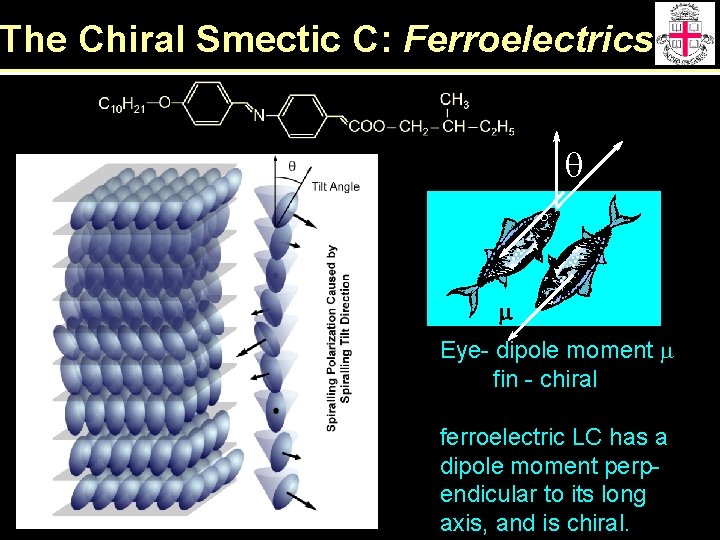
The Chiral Smectic C: Ferroelectrics q m Eye- dipole moment m fin - chiral ferroelectric LC has a dipole moment perpendicular to its long axis, and is chiral.
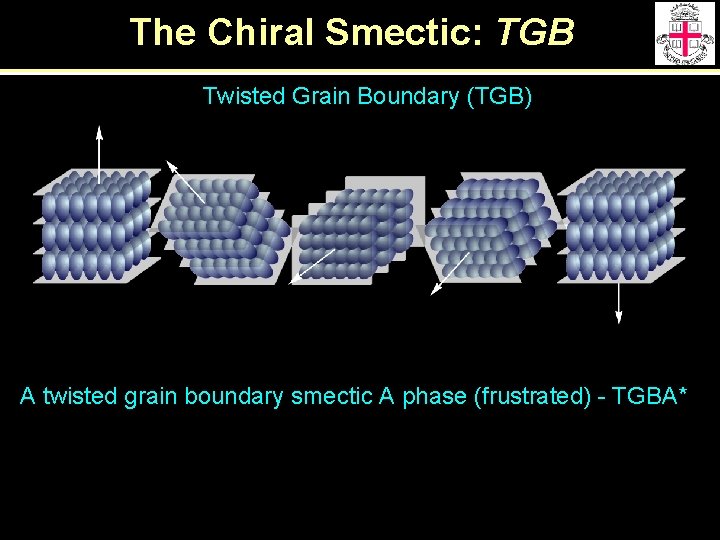
The Chiral Smectic: TGB Twisted Grain Boundary (TGB) A twisted grain boundary smectic A phase (frustrated) - TGBA*
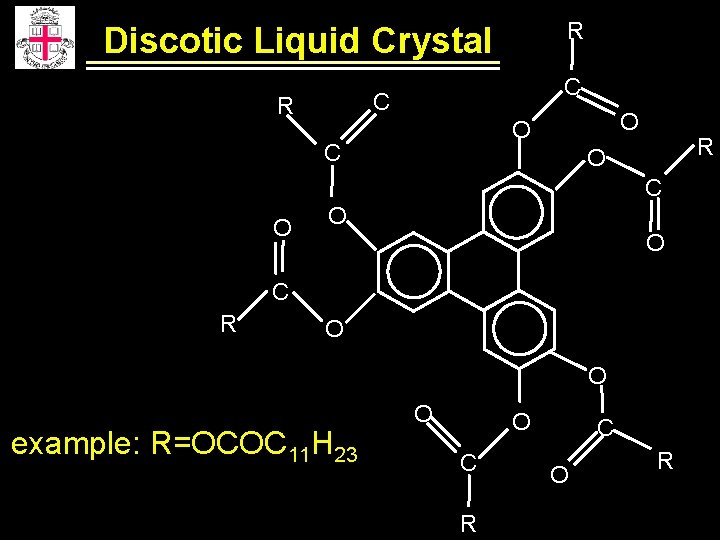
R Discotic Liquid Crystal C C R O O C R O C O O O C R O O example: R=OCOC 11 H 23 O O C R C O R
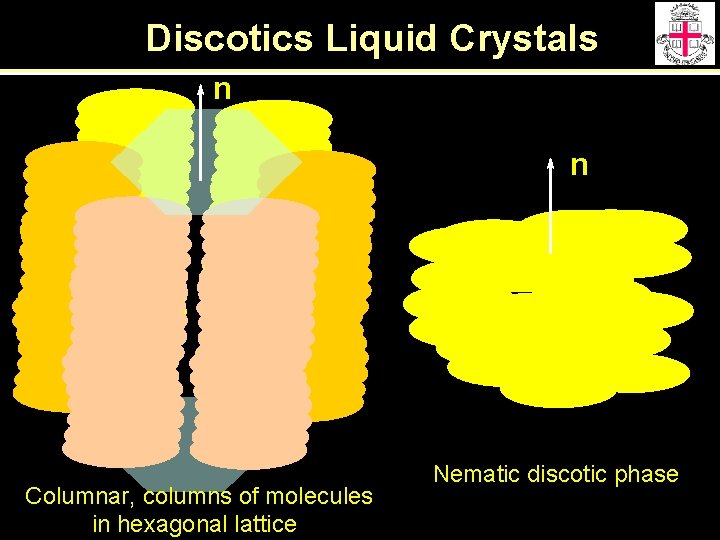
Discotics Liquid Crystals n n Columnar, columns of molecules in hexagonal lattice Nematic discotic phase
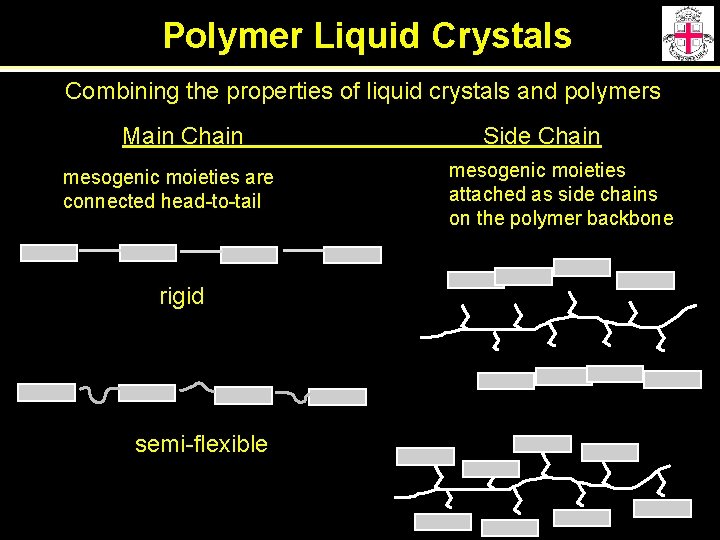
Polymer Liquid Crystals Combining the properties of liquid crystals and polymers Main Chain mesogenic moieties are connected head-to-tail rigid semi-flexible Side Chain mesogenic moieties attached as side chains on the polymer backbone
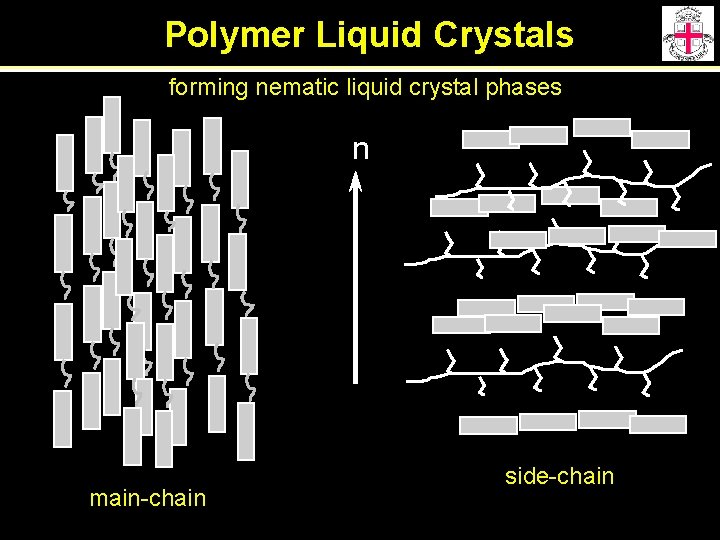
Polymer Liquid Crystals forming nematic liquid crystal phases n main-chain side-chain
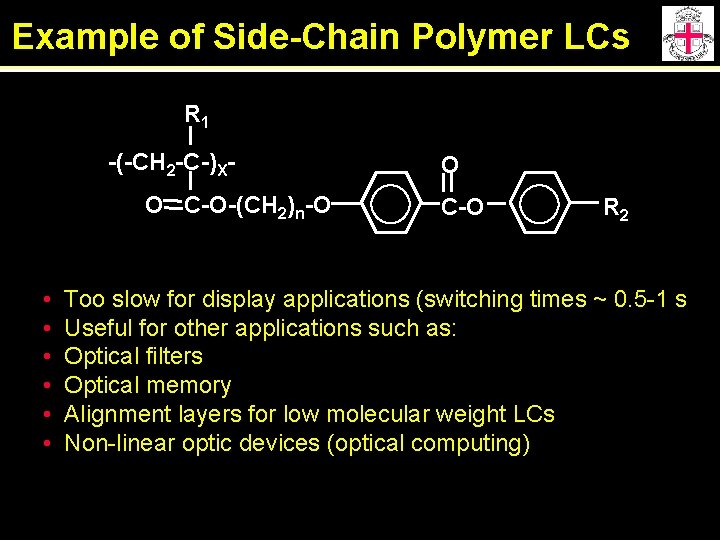
Example of Side-Chain Polymer LCs R 1 -(-CH 2 -C-)XO C-O-(CH 2)n-O • • • O C-O R 2 Too slow for display applications (switching times ~ 0. 5 -1 s Useful for other applications such as: Optical filters Optical memory Alignment layers for low molecular weight LCs Non-linear optic devices (optical computing)
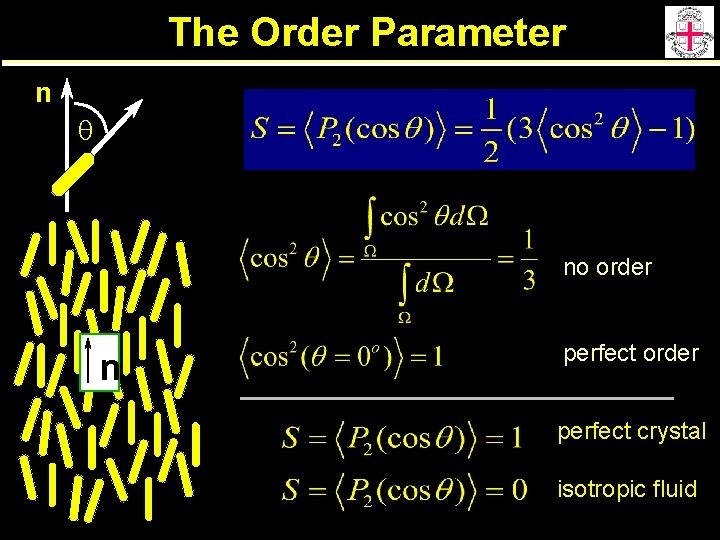
The Order Parameter n q no order n perfect order perfect crystal isotropic fluid

Maier-Saupe Theory - Mean Field Approach Interactions between individual molecules are represented by a potential of average force n q • {V: minimum} when phase is ordered ( -P 2(cosq)) • {V: V=0} when phase is disordered ( <P 2(cosq)>) • factor for intermolecular strength ( n) y f From Statistical Mechanics (Self Consistency) b=(k. T)-1
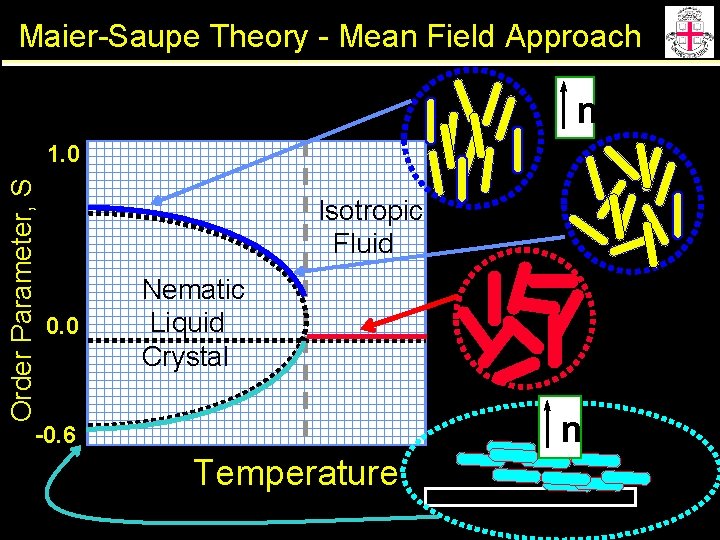
Maier-Saupe Theory - Mean Field Approach n Order Parameter, S 1. 0 Isotropic Fluid 0. 0 Nematic Liquid Crystal n -0. 6 Temperature
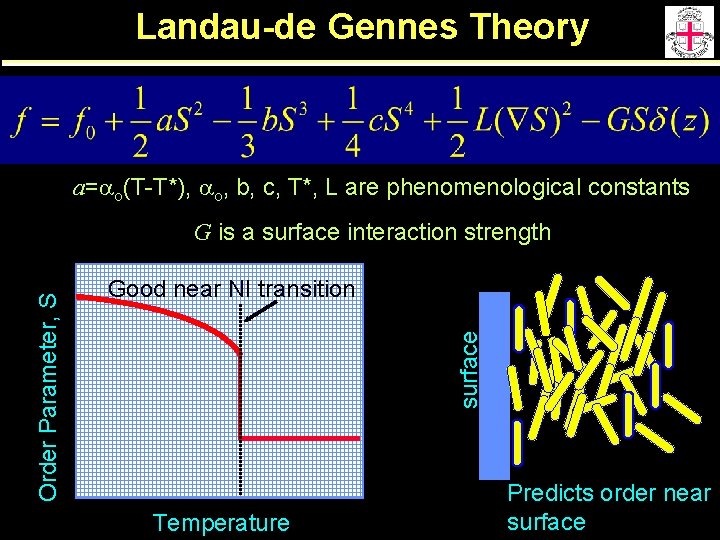
Landau-de Gennes Theory a=ao(T-T*), ao, b, c, T*, L are phenomenological constants Good near NI transition surface Order Parameter, S G is a surface interaction strength Temperature Predicts order near surface

The Order Parameter: How does it affects display performance ? The order parameter, S, is proportional to a number of important parameters which dictate display performance. Parameter Elastic Constant Birefringence Dielectric Anisotropy Magnetic Anisotropy Viscosity Anisotropy Nomenclature Kii Dn De Dc Dh proportional to S 2 S S Example: Does the threshold switching voltage for a TN increase or decrease as the operating temperature increases. Scales as the square root of S therefore lowers with increasing temperature
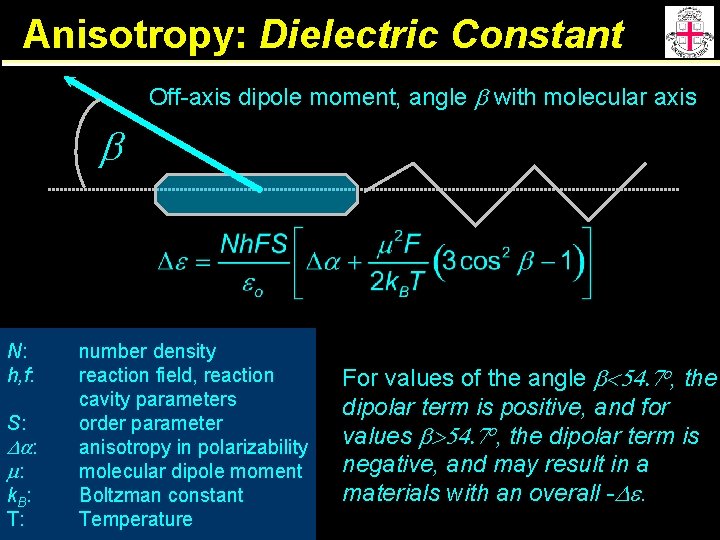
Anisotropy: Dielectric Constant Off-axis dipole moment, angle b with molecular axis b N: h, f: S: Da: m: k. B: T: number density reaction field, reaction cavity parameters order parameter anisotropy in polarizability molecular dipole moment Boltzman constant Temperature For values of the angle b<54. 7 o, the dipolar term is positive, and for values b>54. 7 o, the dipolar term is negative, and may result in a materials with an overall -De.
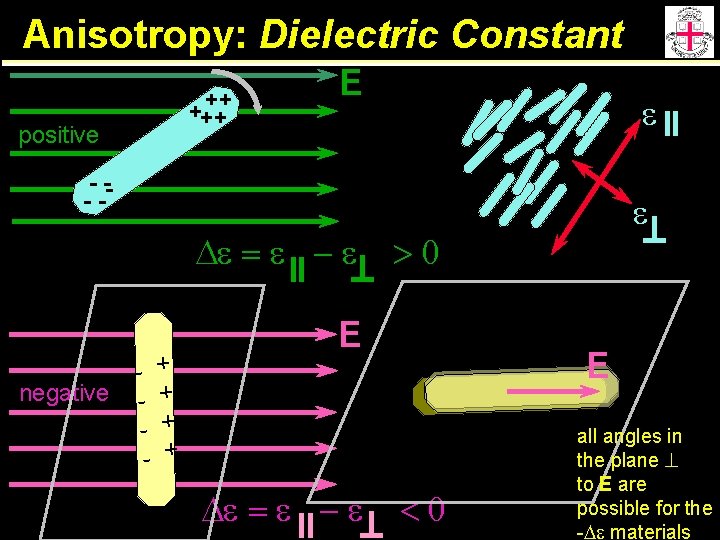
Anisotropy: Dielectric Constant ++ +++ positive E e - --- De = e - e negative - + - + >0 E De = e - e e E <0 all angles in the plane to E are possible for the -De materials
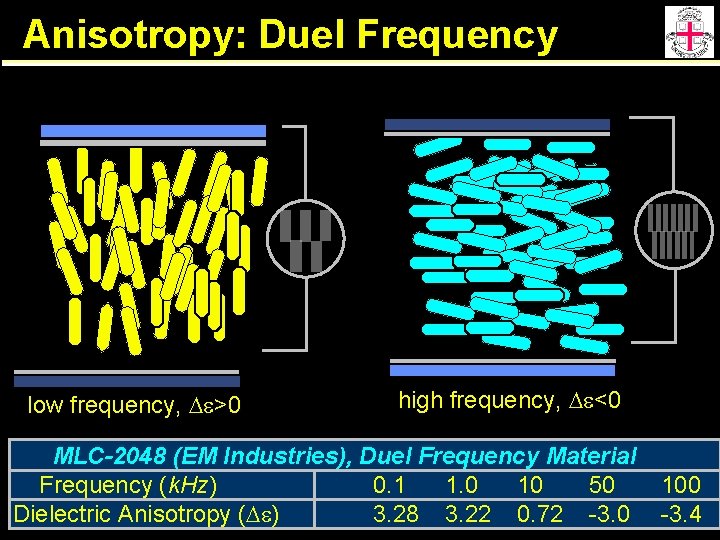
Anisotropy: Duel Frequency low frequency, De>0 high frequency, De<0 MLC-2048 (EM Industries), Duel Frequency Material Frequency (k. Hz) 0. 1 1. 0 10 50 Dielectric Anisotropy (De) 3. 28 3. 22 0. 72 -3. 0 100 -3. 4
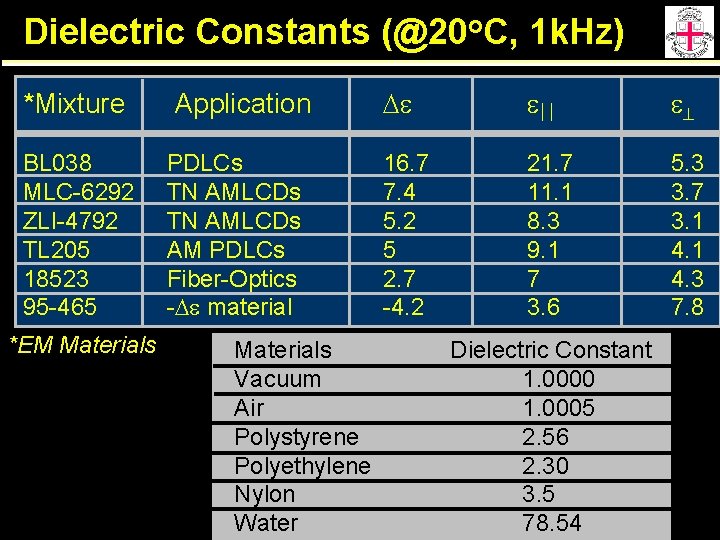
Dielectric Constants (@20 o. C, 1 k. Hz) *Mixture BL 038 MLC-6292 ZLI-4792 TL 205 18523 95 -465 *EM Materials Application PDLCs TN AMLCDs AM PDLCs Fiber-Optics -De material Materials Vacuum Air Polystyrene Polyethylene Nylon Water De e e 16. 7 7. 4 5. 2 5 2. 7 -4. 2 21. 7 11. 1 8. 3 9. 1 7 3. 6 5. 3 3. 7 3. 1 4. 3 7. 8 Dielectric Constant 1. 0000 1. 0005 2. 56 2. 30 3. 5 78. 54
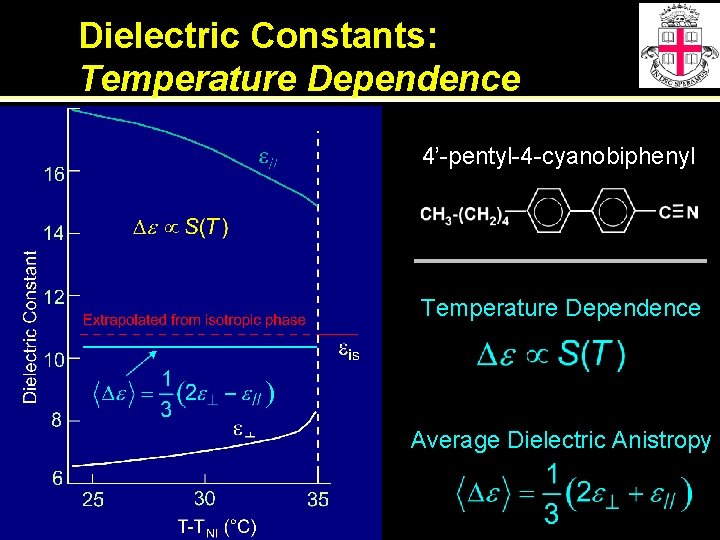
Dielectric Constants: Temperature Dependence 4’-pentyl-4 -cyanobiphenyl Temperature Dependence Average Dielectric Anistropy

Magnetic Anisotropy: Diamagnetism: induction of a magnetic moment in opposition to an applied magnetic field. LCs are diamagnetic due to the dispersed electron distribution associated with the electron structure. Delocalized charge makes the major contribution to diamagnetism. Ring currents associated with aromatic units give a large negative component to c for directions to aromatic ring plane. Dc is usually positive since:
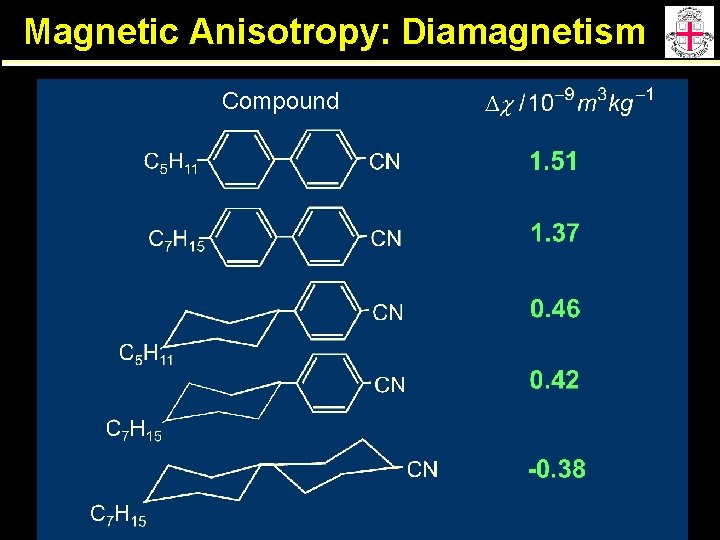
Magnetic Anisotropy: Diamagnetism Compound
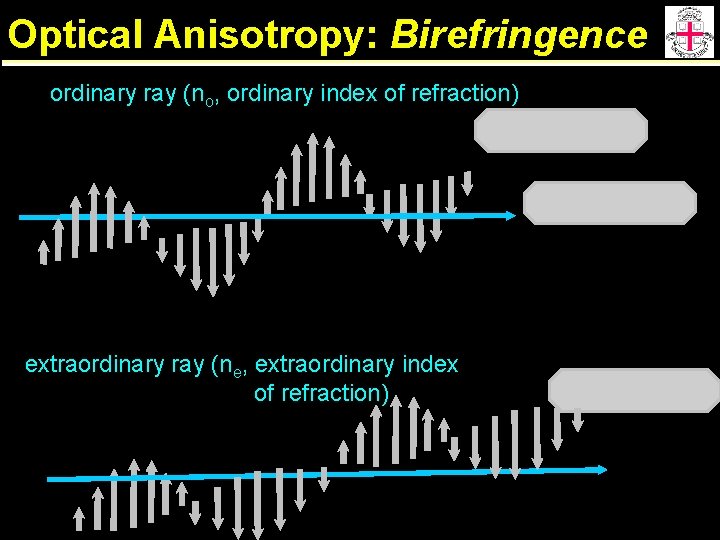
Optical Anisotropy: Birefringence ordinary ray (no, ordinary index of refraction) extraordinary ray (ne, extraordinary index of refraction)
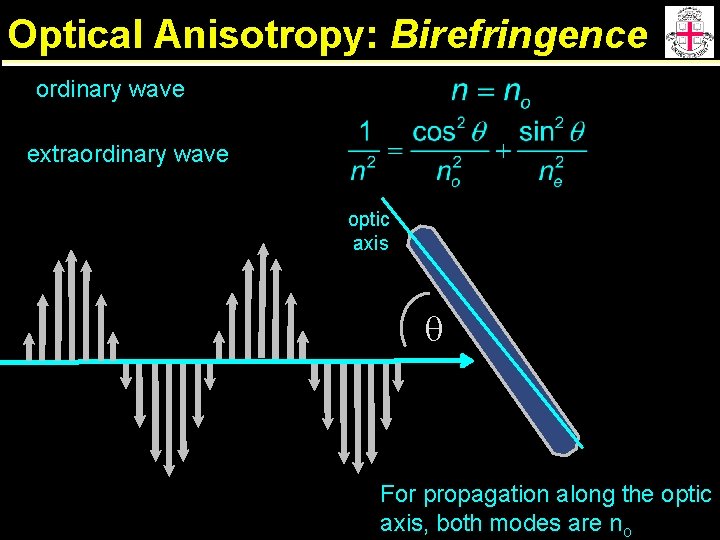
Optical Anisotropy: Birefringence ordinary wave extraordinary wave optic axis q For propagation along the optic axis, both modes are no
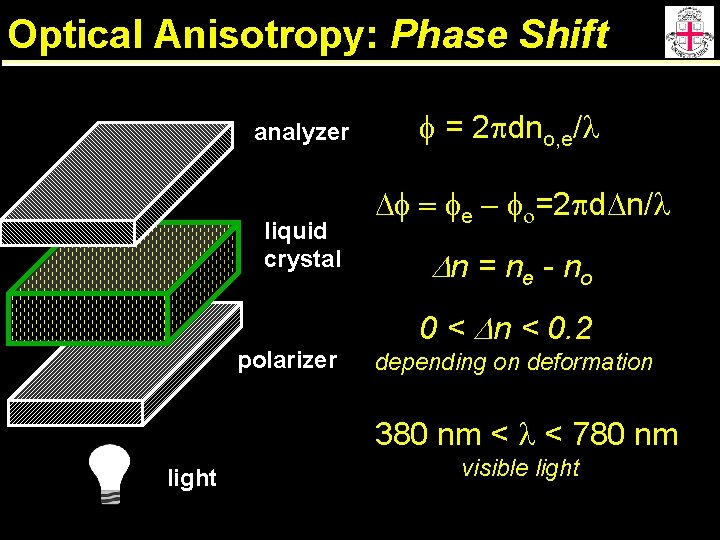
Optical Anisotropy: Phase Shift analyzer liquid crystal polarizer f = 2 pdno, e/l Df = fe - fo=2 pd. Dn/l Dn = ne - n o 0 < Dn < 0. 2 depending on deformation 380 nm < l < 780 nm light visible light
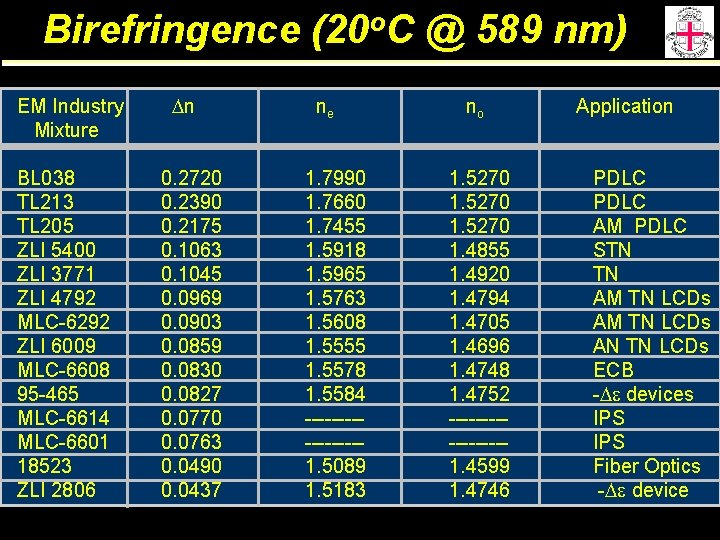
Birefringence (20 o. C @ 589 nm) EM Industry Mixture BL 038 TL 213 TL 205 ZLI 5400 ZLI 3771 ZLI 4792 MLC-6292 ZLI 6009 MLC-6608 95 -465 MLC-6614 MLC-6601 18523 ZLI 2806 Dn 0. 2720 0. 2390 0. 2175 0. 1063 0. 1045 0. 0969 0. 0903 0. 0859 0. 0830 0. 0827 0. 0770 0. 0763 0. 0490 0. 0437 ne 1. 7990 1. 7660 1. 7455 1. 5918 1. 5965 1. 5763 1. 5608 1. 5555 1. 5578 1. 5584 --------1. 5089 1. 5183 no 1. 5270 1. 4855 1. 4920 1. 4794 1. 4705 1. 4696 1. 4748 1. 4752 --------1. 4599 1. 4746 Application PDLC AM PDLC STN TN AM TN LCDs AN TN LCDs ECB -De devices IPS Fiber Optics -De device
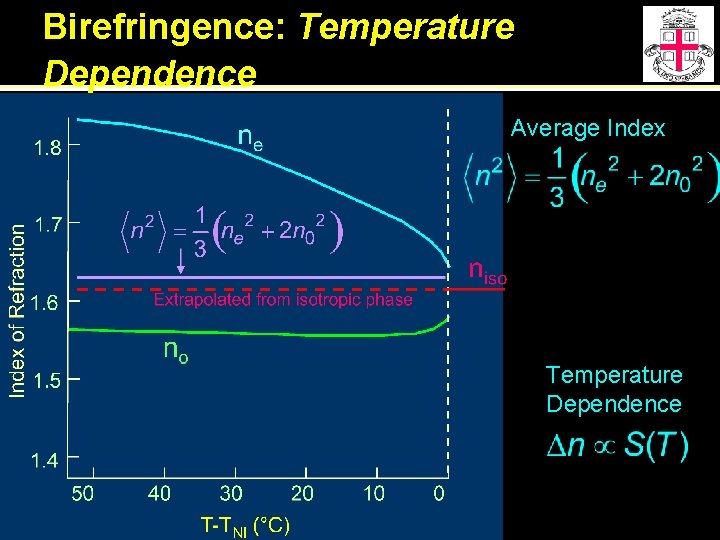
Birefringence: Temperature Dependence Average Index Temperature Dependence
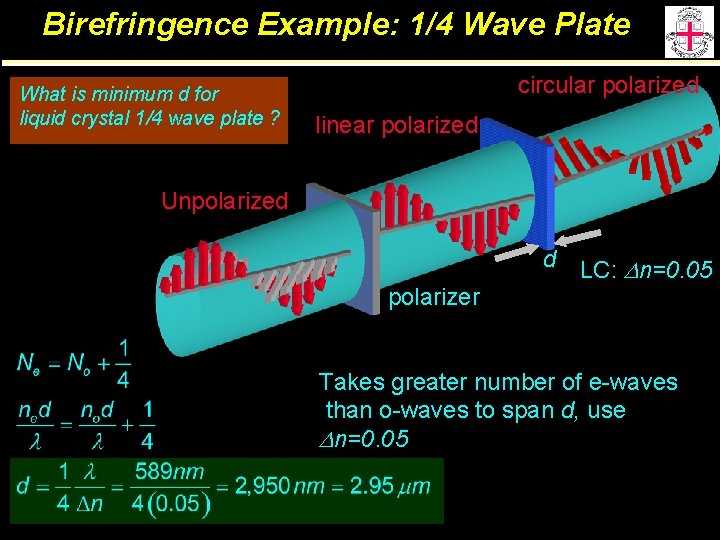
Birefringence Example: 1/4 Wave Plate What is minimum d for liquid crystal 1/4 wave plate ? circular polarized linear polarized Unpolarized polarizer d LC: Dn=0. 05 Takes greater number of e-waves than o-waves to span d, use Dn=0. 05

Nematic Elasticity: Frank Elastic Theory 1 2 2 2 Ñ × + × Ñ ´ + ´ Ñ ´ { K ( n ) K ( n n ) } d. V 11 22 33 ò 2 V 1 - ò { K 24 Ñ ×( n × Ñ ´ n + n Ñ × n ) + K 13 Ñ × ( n Ñ × n )} d. V 2 V Dc 1 1 2 Fe = - ò e o D e ( E × n ) d. V - ò o ( B × n ) 2 d. V 2 V 2 V co Fd = Splay, K 11 Twist, K 22 Bend, K 33
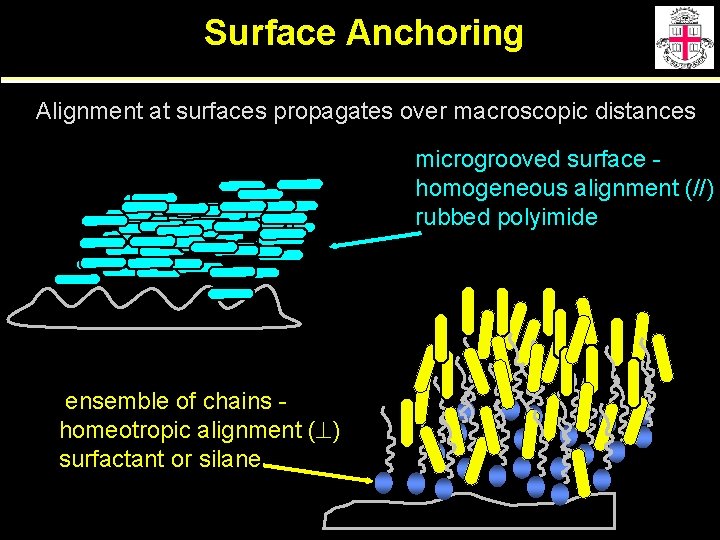
Surface Anchoring Alignment at surfaces propagates over macroscopic distances microgrooved surface homogeneous alignment (//) rubbed polyimide ensemble of chains homeotropic alignment ( ) surfactant or silane
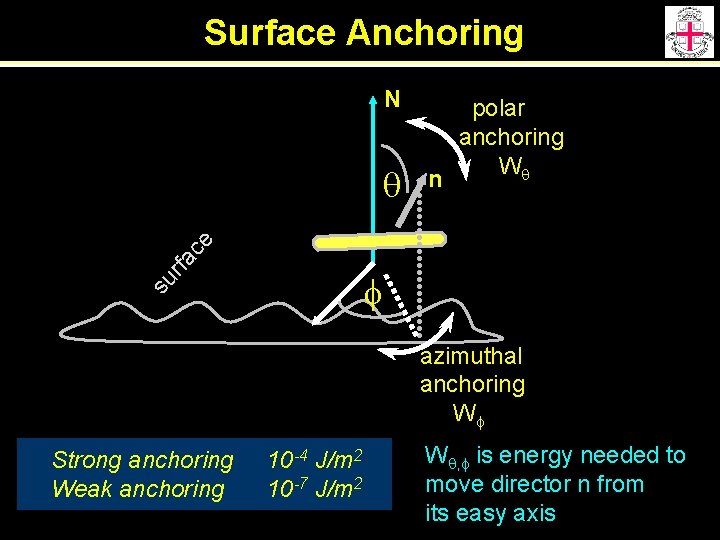
Surface Anchoring N n rfa ce q polar anchoring Wq su f azimuthal anchoring Wf Strong anchoring Weak anchoring 10 -4 J/m 2 10 -7 J/m 2 Wq, f is energy needed to move director n from its easy axis
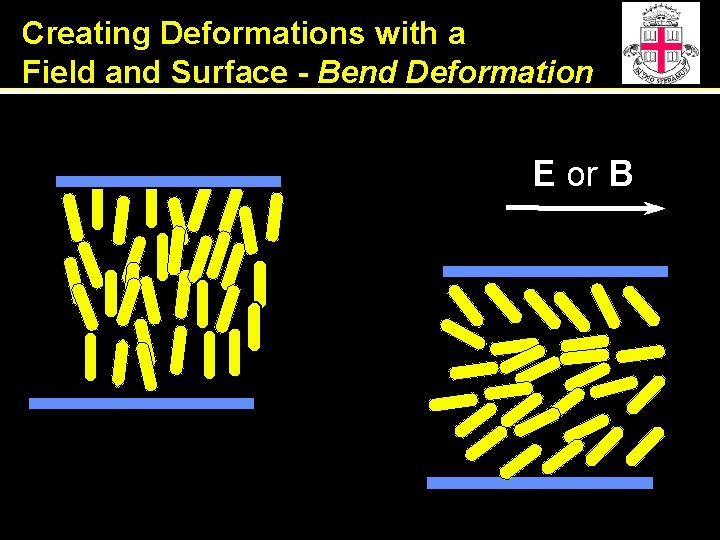
Creating Deformations with a Field and Surface - Bend Deformation E or B
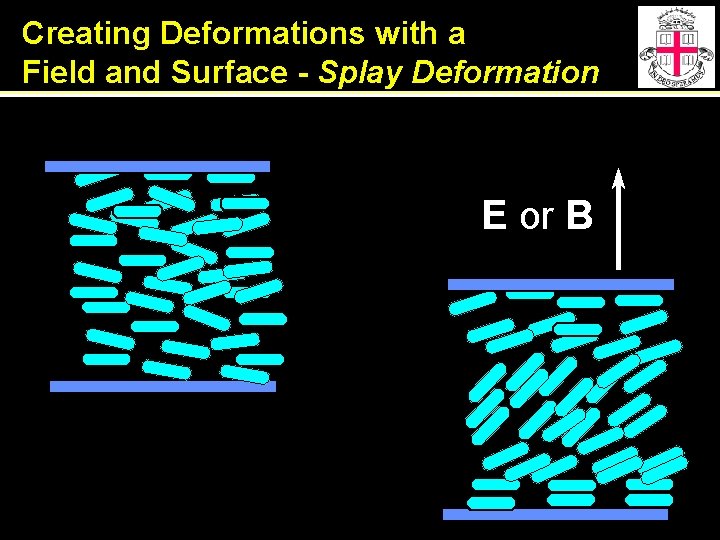
Creating Deformations with a Field and Surface - Splay Deformation E or B
Creating Deformations with a Field and Surface - Twist Deformation E or B
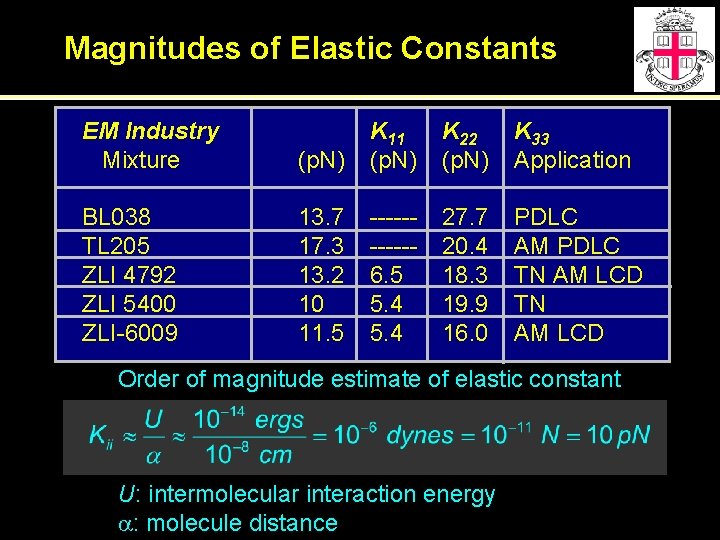
Magnitudes of Elastic Constants EM Industry Mixture BL 038 TL 205 ZLI 4792 ZLI 5400 ZLI-6009 (p. N) K 11 (p. N) K 22 (p. N) K 33 Application 13. 7 17. 3 13. 2 10 11. 5 -----6. 5 5. 4 27. 7 20. 4 18. 3 19. 9 16. 0 PDLC AM PDLC TN AM LCD Order of magnitude estimate of elastic constant U: intermolecular interaction energy a: molecule distance

Elastic Constant K 22: Temperature Dependence
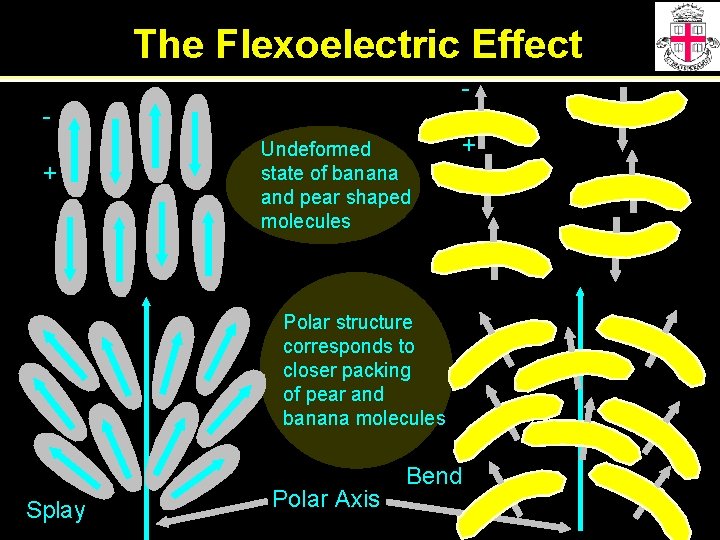
The Flexoelectric Effect + Undeformed state of banana and pear shaped molecules Polar structure corresponds to closer packing of pear and banana molecules Splay Polar Axis Bend +

Effects of an Electric Field n E q y x e e Electric Free Energy Density Electric Torque Density Using De = 5 and E=0. 5 V/mm

Effects of an Magnetic Field n B q y x c c Magnetic free energy density Magnetic torque density Using Dc = 10 -7 m 3 kg-1 and B= 2 T = 20, 000 G
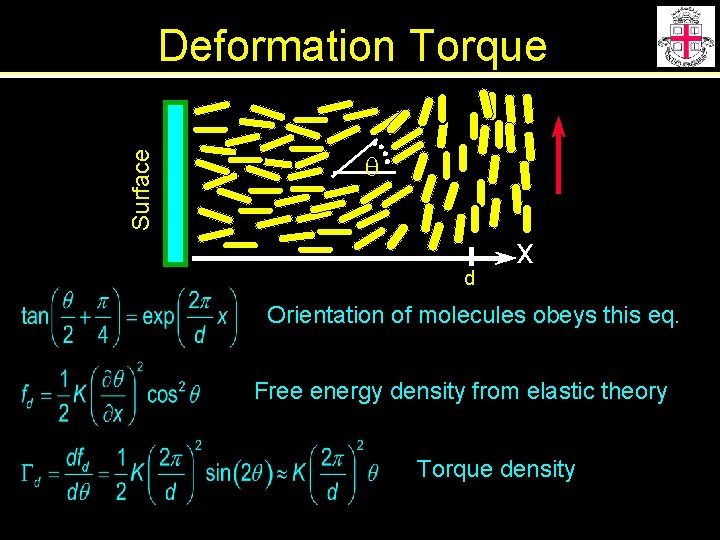
Surface Deformation Torque q d x Orientation of molecules obeys this eq. Free energy density from elastic theory Torque density
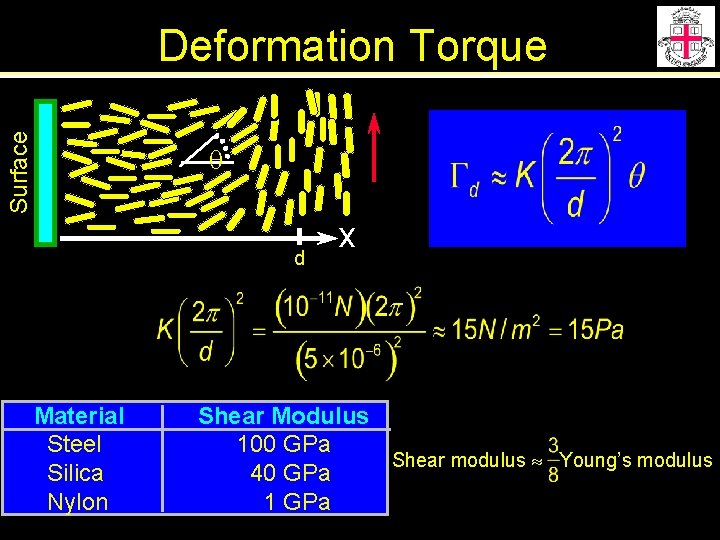
Surface Deformation Torque q d Material Steel Silica Nylon x Shear Modulus 100 GPa Shear modulus Young’s modulus 40 GPa 1 GPa
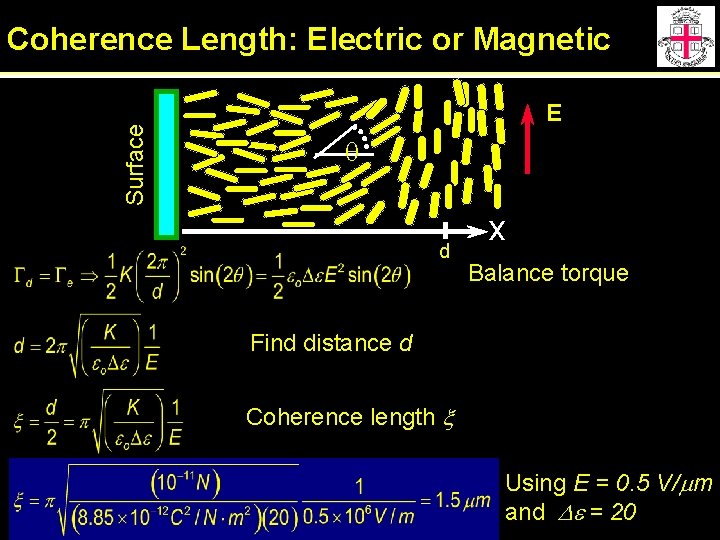
Surface Coherence Length: Electric or Magnetic E q d x Balance torque Find distance d Coherence length x Using E = 0. 5 V/mm and De = 20
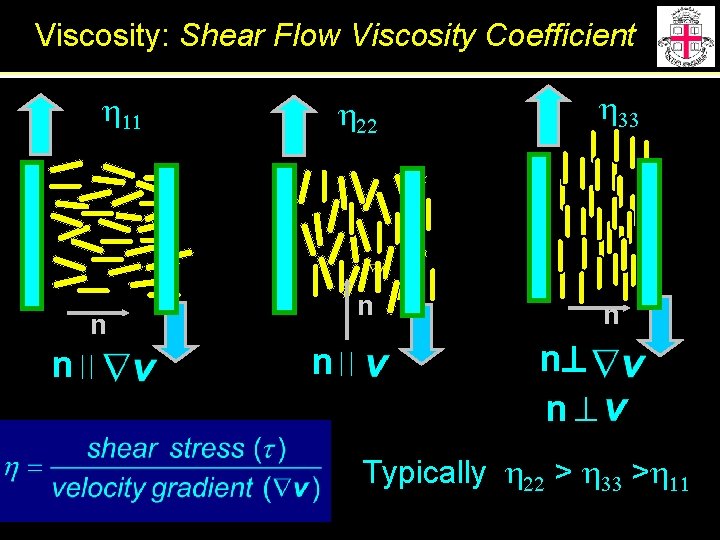
Viscosity: Shear Flow Viscosity Coefficient h 11 n n h 22 h 33 n n n Typically h 22 > h 33 >h 11

Viscosity: Flow Viscosity Coefficient LC specification sheets give kinematic viscosity in mm 2/s Kinematic Viscosity (n) 1 m 2/s Dynamic Viscosity (h) 1 kg/m·s = 1 Pa·s 0. 1 kg/m·s = 1 poise Approximate density
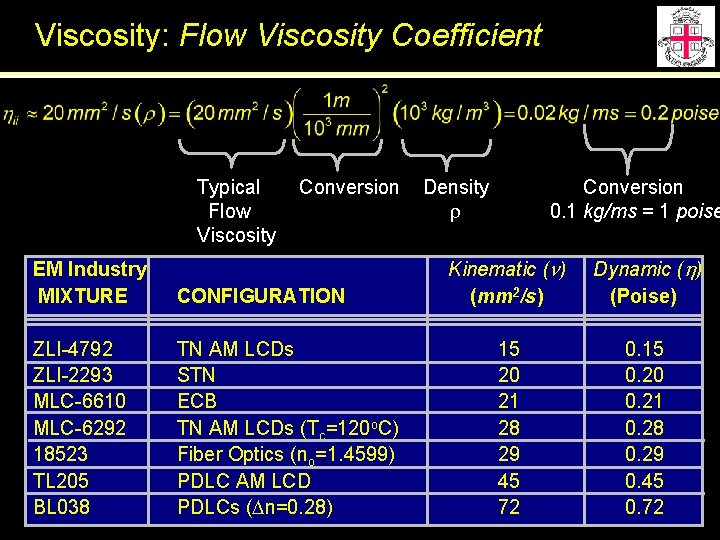
Viscosity: Flow Viscosity Coefficient Typical Flow Viscosity Conversion EM Industry MIXTURE CONFIGURATION ZLI-4792 ZLI-2293 MLC-6610 MLC-6292 18523 TL 205 BL 038 TN AM LCDs STN ECB TN AM LCDs (Tc=120 o. C) Fiber Optics (no=1. 4599) PDLC AM LCD PDLCs (Dn=0. 28) Density r Conversion 0. 1 kg/ms = 1 poise Kinematic (n) (mm 2/s) Dynamic (h) (Poise) 15 20 21 28 29 45 72 0. 15 0. 20 0. 21 0. 28 0. 29 0. 45 0. 72
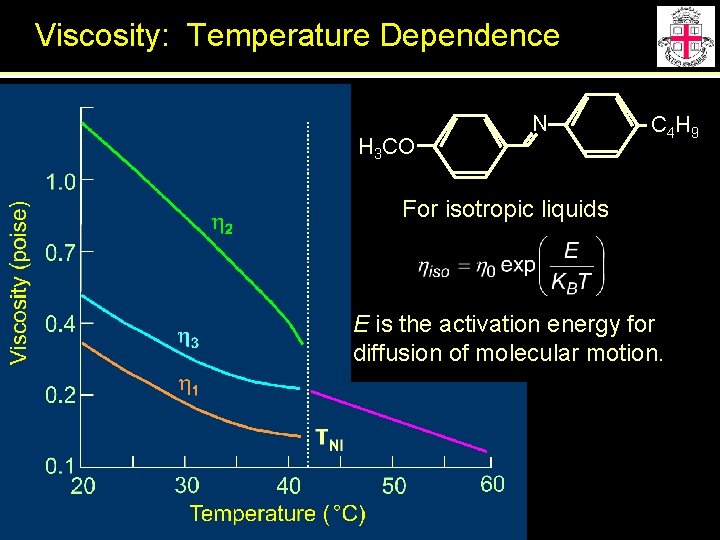
Viscosity: Temperature Dependence H 3 CO N C 4 H 9 For isotropic liquids E is the activation energy for diffusion of molecular motion.
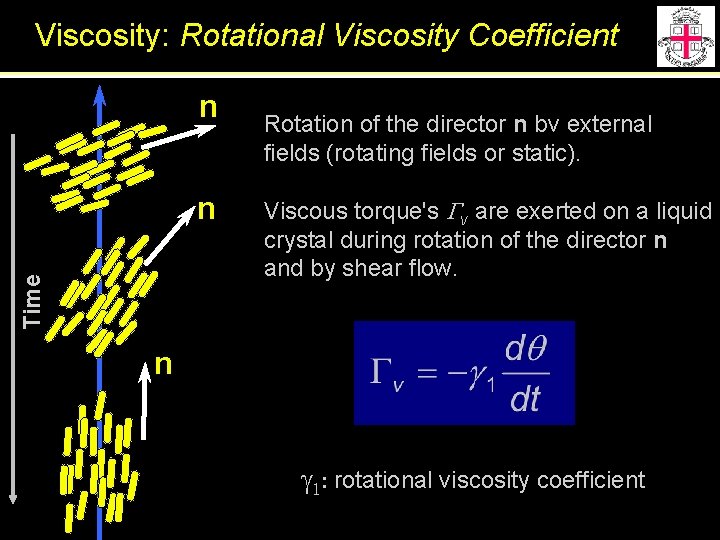
Viscosity: Rotational Viscosity Coefficient n Time n Rotation of the director n bv external fields (rotating fields or static). Viscous torque's Gv are exerted on a liquid crystal during rotation of the director n and by shear flow. n g 1: rotational viscosity coefficient
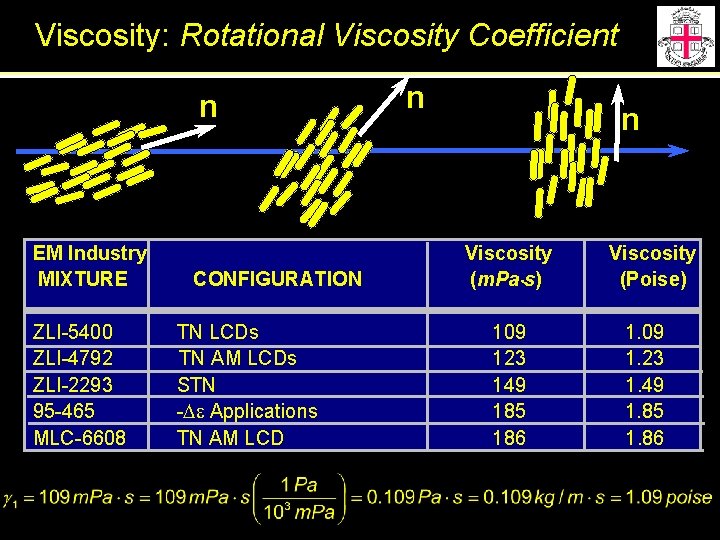
Viscosity: Rotational Viscosity Coefficient n EM Industry MIXTURE ZLI-5400 ZLI-4792 ZLI-2293 95 -465 MLC-6608 CONFIGURATION TN LCDs TN AM LCDs STN -De Applications TN AM LCD n n Viscosity (m. Pa s) 109 123 149 185 186 Viscosity (Poise) 1. 09 1. 23 1. 49 1. 85 1. 86
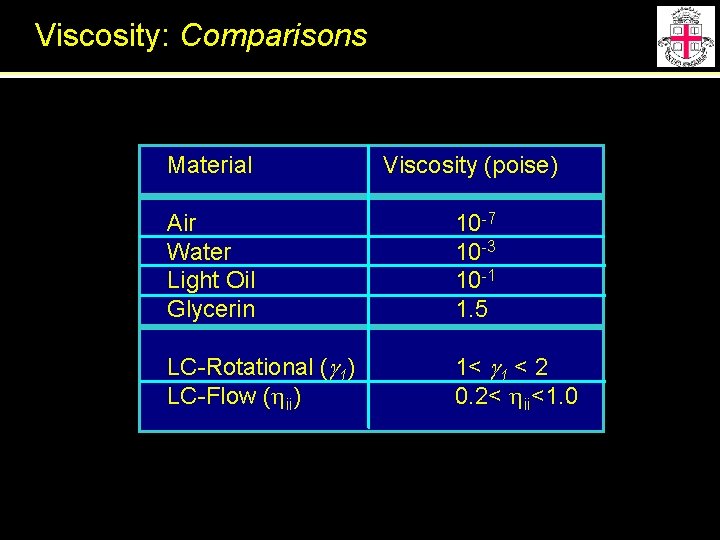
Viscosity: Comparisons Material Viscosity (poise) Air Water Light Oil Glycerin 10 -7 10 -3 10 -1 1. 5 LC-Rotational (g 1) LC-Flow (hii) 1< g 1 < 2 0. 2< hii<1. 0
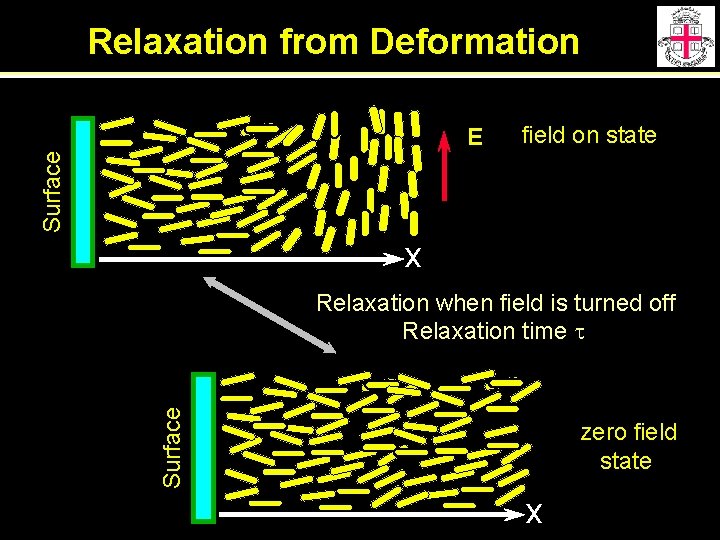
Relaxation from Deformation field on state Surface E x Surface Relaxation when field is turned off Relaxation time t zero field state x

Relaxation from Deformation Balance viscous/deformation torque Assume small deformations Solution For 100 mm cell For 5 mm cell
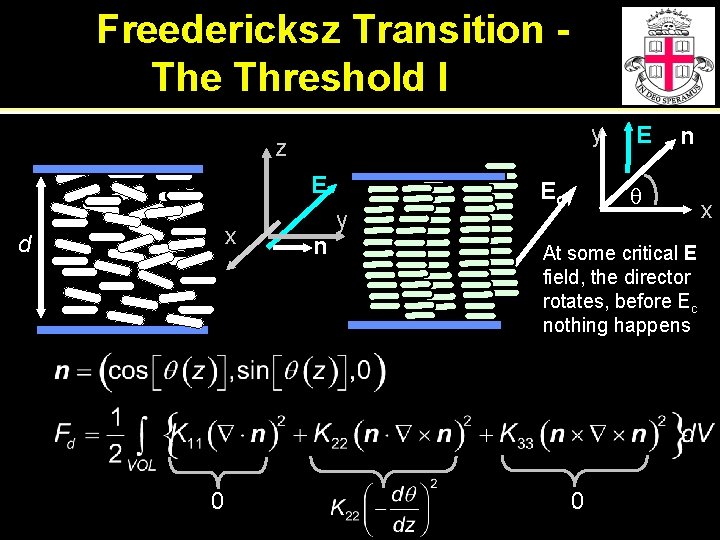
Freedericksz Transition The Threshold I y z E x d 0 n y Ec E n q At some critical E field, the director rotates, before Ec nothing happens 0 x
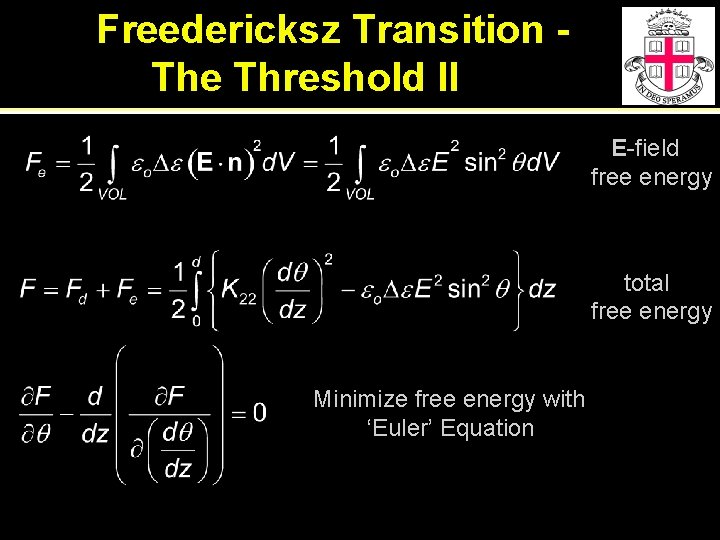
Freedericksz Transition The Threshold II E-field free energy total free energy Minimize free energy with ‘Euler’ Equation

Freedericksz Transition The Threshold III differential equation mid-layer tilt (deg) soln. small q threshold 1. 0 E/Ec

Defects s=1/2 s=+1 s=3/2 s=-1/2 s=+1 s=+2 s=-1 s=+1 The singular line (disclination) is pointing out of the page, and director orientation changes by 2 ps on going around the line (s is the strength)

Estimate Defect Size The simplest hypothesis is that the core or defect or disclination is an isotropic liquid, therefore the core energy is proportional to k. BDTc. Let M be the molecular mass, N Avogadadro’s number and r the density of the liquid crystal.
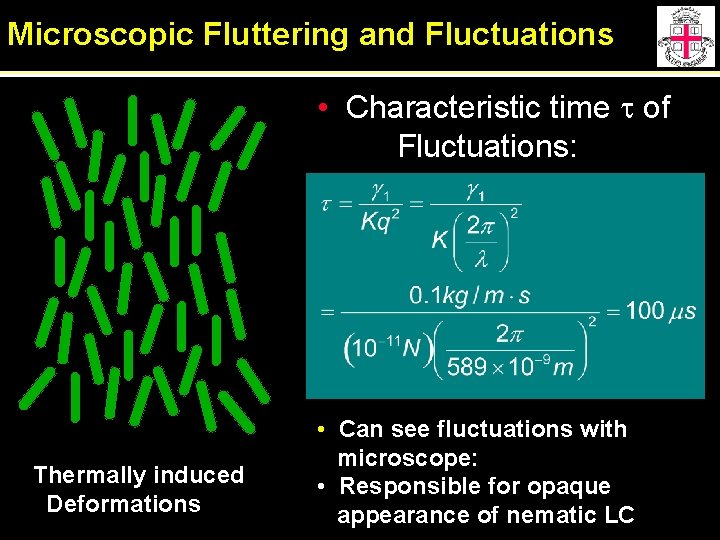
Microscopic Fluttering and Fluctuations • Characteristic time t of Fluctuations: Thermally induced Deformations • Can see fluctuations with microscope: • Responsible for opaque appearance of nematic LC
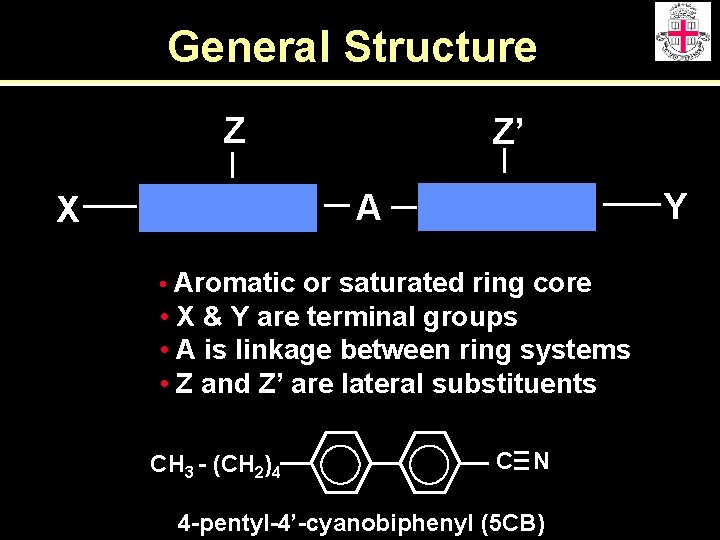
General Structure Z Z’ Y A X • Aromatic or saturated ring core • X & Y are terminal groups • A is linkage between ring systems • Z and Z’ are lateral substituents CH 3 - (CH 2)4 C N 4 -pentyl-4’-cyanobiphenyl (5 CB)
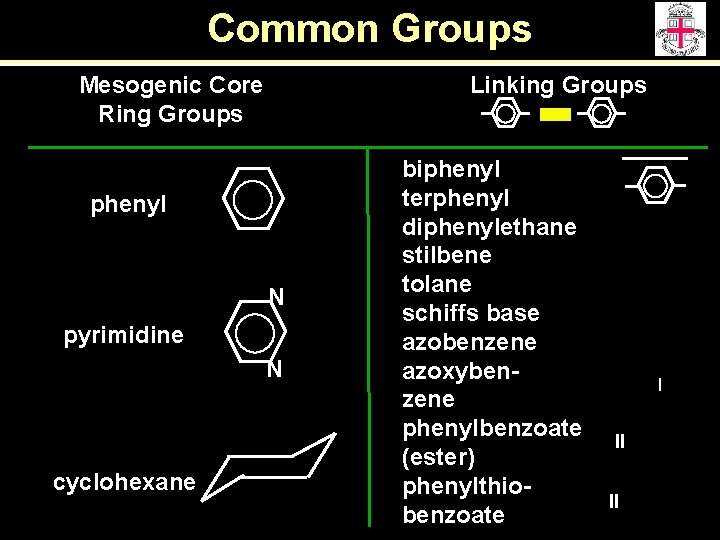
Common Groups Mesogenic Core Ring Groups Linking Groups phenyl N pyrimidine N cyclohexane biphenyl terphenyl diphenylethane stilbene tolane schiffs base azobenzene azoxybenzene phenylbenzoate (ester) phenylthiobenzoate
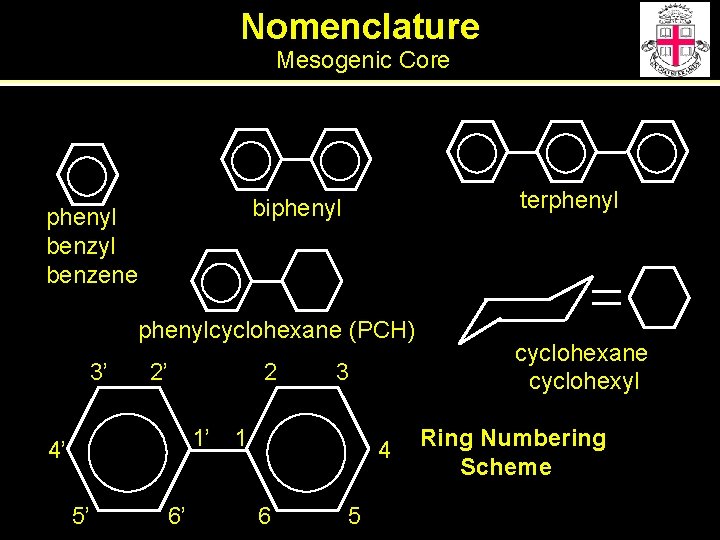
Nomenclature Mesogenic Core terphenyl biphenyl benzyl benzene phenylcyclohexane (PCH) 3’ 2’ 2 3 1’ 1 4’ 5’ 6’ 4 6 5 cyclohexane cyclohexyl Ring Numbering Scheme
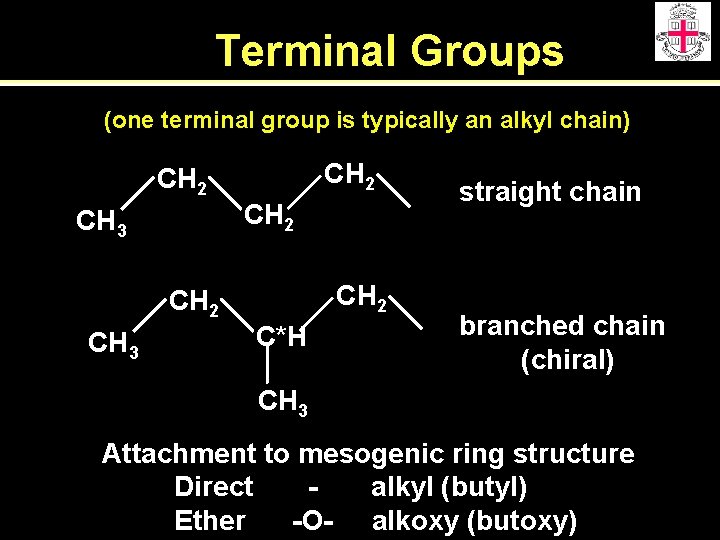
Terminal Groups (one terminal group is typically an alkyl chain) CH 2 CH 3 CH 2 C*H straight chain branched chain (chiral) CH 3 Attachment to mesogenic ring structure Direct alkyl (butyl) Ether -O- alkoxy (butoxy)

Terminal Groups CH 3 - methyl CH 3 -O- methoxy CH 3 -CH 2 - ethyl CH 3 -CH 2 -O- ethoxy CH 3 -(CH 2)2 - propyl CH 3 -(CH 2)2 -O- propoxy CH 3 -(CH 2)3 - butyl CH 3 -(CH 2)3 -O- butoxy CH 3 -(CH 2)4 - pentyl CH 3 -(CH 2)4 -O- pentoxy CH 3 -(CH 2)5 - hexyl CH 3 -(CH 2)5 -O- hexoxy CH 3 -(CH 2)6 - heptyl CH 3 -(CH 2)6 -O- heptoxy CH 3 -(CH 2)7 - octyl CH 3 -(CH 2)7 -O- octoxy

Second Terminal Group and Lateral Substituents (Y & Z) H F Cl Br I CH 3(CH 2)n CN NH 2 N(CH 3) NO 2 flouro chloro bromo iodo methyl alkyl cyano amino dimethylamino nitro phenyl cyclohexyl
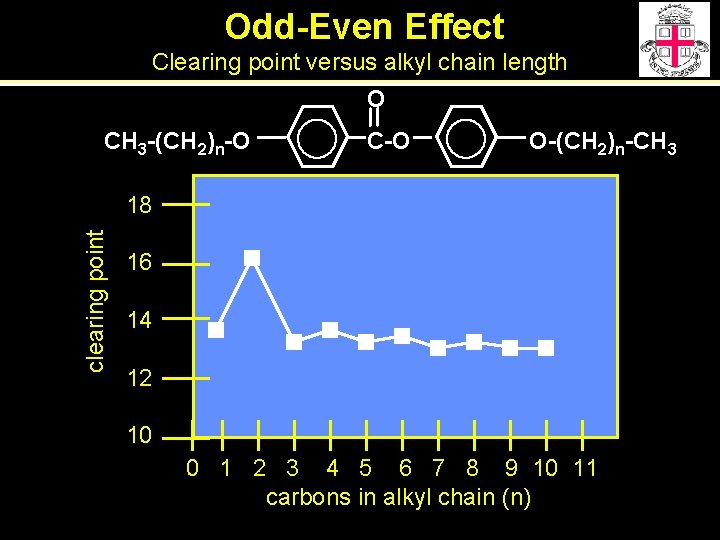
Odd-Even Effect Clearing point versus alkyl chain length O CH 3 -(CH 2)n-O C-O O-(CH 2)n-CH 3 clearing point 18 16 14 12 10 0 1 2 3 4 5 6 7 8 9 10 11 carbons in alkyl chain (n)
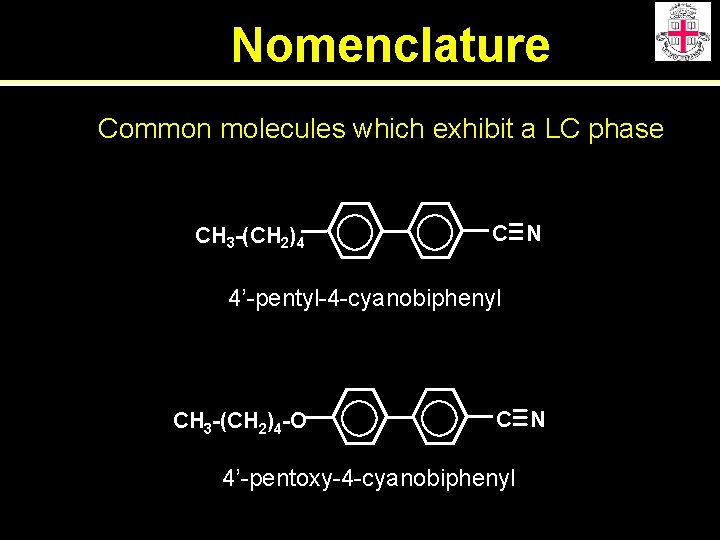
Nomenclature Common molecules which exhibit a LC phase CH 3 -(CH 2)4 C N 4’-pentyl-4 -cyanobiphenyl CH 3 -(CH 2)4 -O C N 4’-pentoxy-4 -cyanobiphenyl
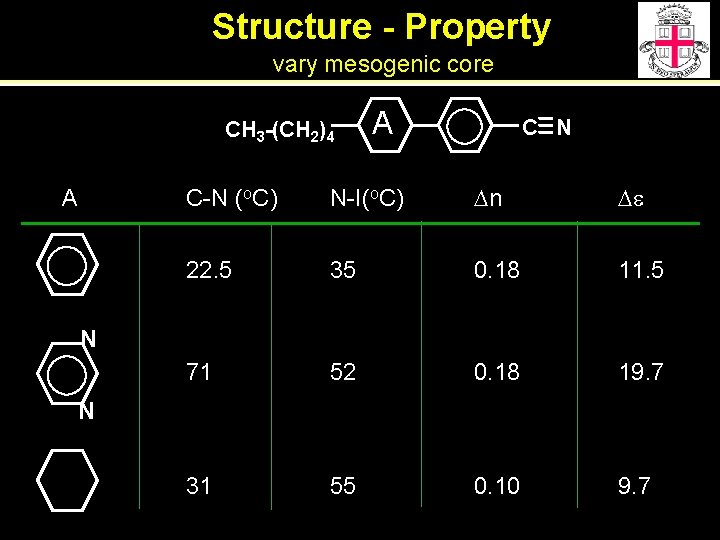
Structure - Property vary mesogenic core CH 3 -(CH 2)4 A A C N C-N (o. C) N-I(o. C) Dn De 22. 5 35 0. 18 11. 5 71 52 0. 18 19. 7 31 55 0. 10 9. 7 N N
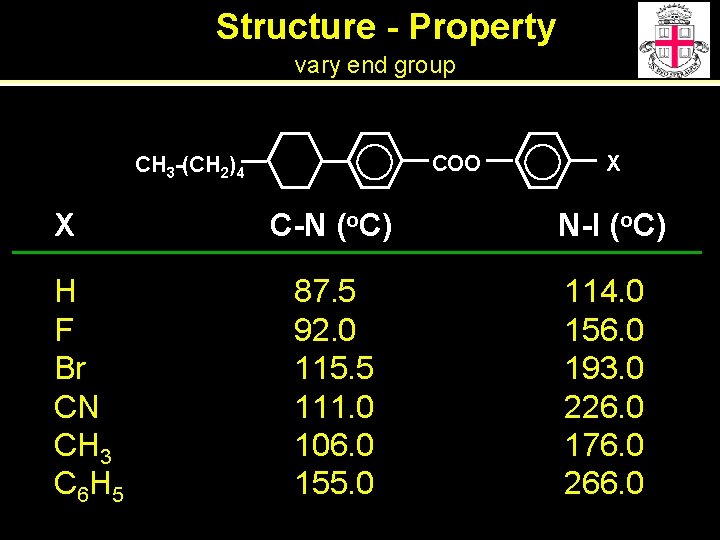
Structure - Property vary end group COO CH 3 -(CH 2)4 X H F Br CN CH 3 C 6 H 5 X C-N (o. C) N-I (o. C) 87. 5 92. 0 115. 5 111. 0 106. 0 155. 0 114. 0 156. 0 193. 0 226. 0 176. 0 266. 0
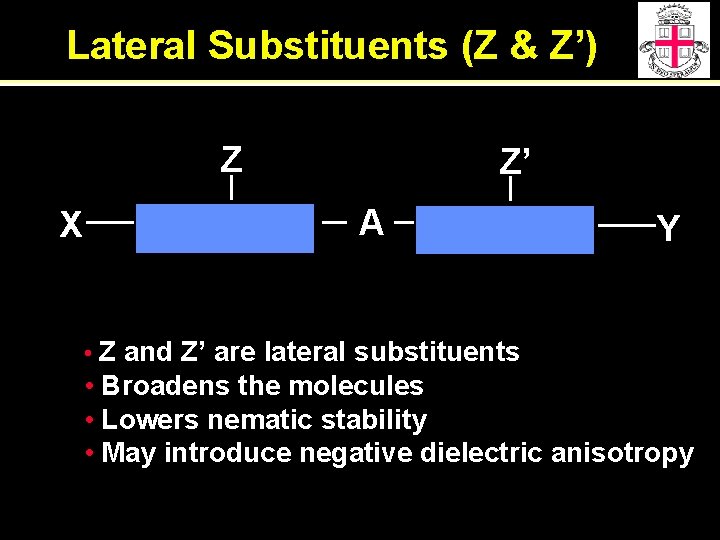
Lateral Substituents (Z & Z’) Z X Z’ A Y • Z and Z’ are lateral substituents • Broadens the molecules • Lowers nematic stability • May introduce negative dielectric anisotropy
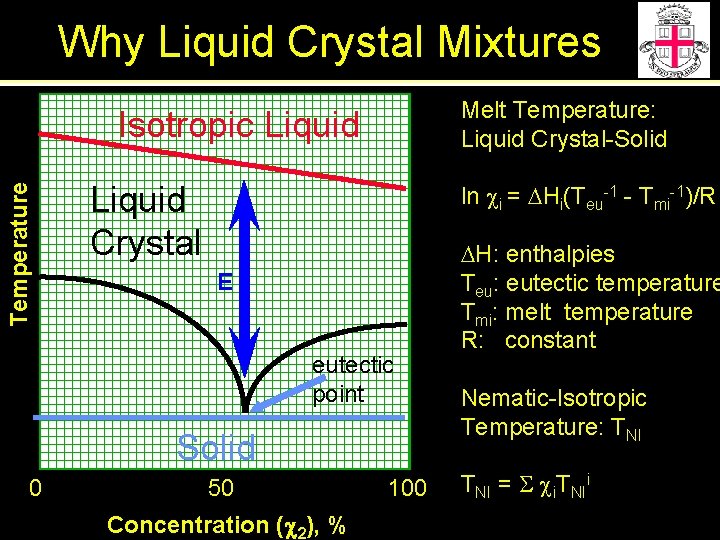
Why Liquid Crystal Mixtures Melt Temperature: Liquid Crystal-Solid Temperature Isotropic Liquid Crystal ln ci = DHi(Teu-1 - Tmi-1)/R E eutectic point Solid 0 50 Concentration (c 2), % 100 DH: enthalpies Teu: eutectic temperature Tmi: melt temperature R: constant Nematic-Isotropic Temperature: TNI = S ci. TNIi
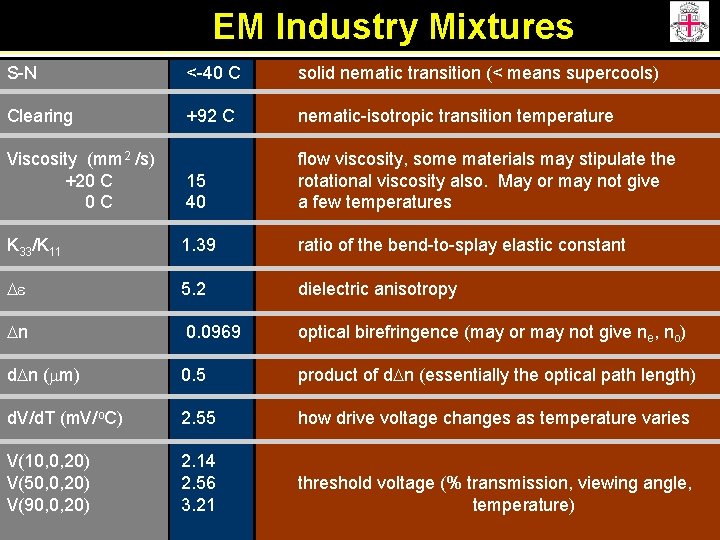
EM Industry Mixtures S-N <-40 C solid nematic transition (< means supercools) Clearing +92 C nematic-isotropic transition temperature Viscosity (mm 2 /s) +20 C 0 C 15 40 flow viscosity, some materials may stipulate the rotational viscosity also. May or may not give a few temperatures K 33/K 11 1. 39 ratio of the bend-to-splay elastic constant De 5. 2 dielectric anisotropy Dn 0. 0969 optical birefringence (may or may not give ne, no) d. Dn (mm) 0. 5 product of d. Dn (essentially the optical path length) d. V/d. T (m. V/o. C) 2. 55 how drive voltage changes as temperature varies V(10, 0, 20) V(50, 0, 20) V(90, 0, 20) 2. 14 2. 56 3. 21 threshold voltage (% transmission, viewing angle, temperature)
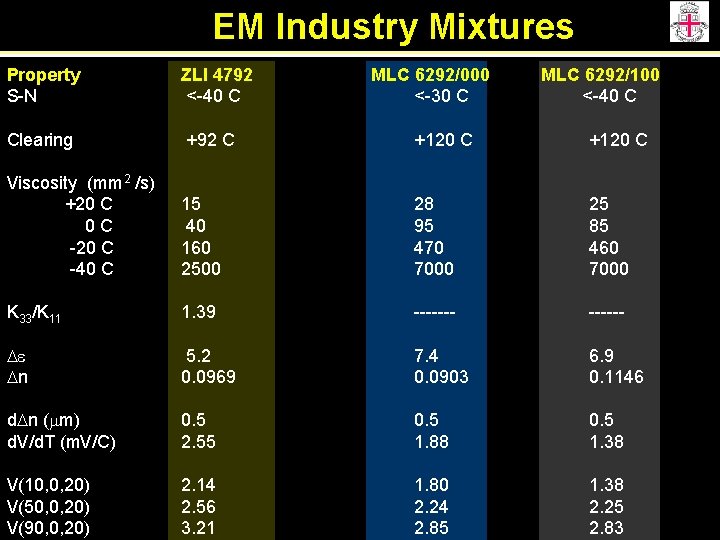
EM Industry Mixtures Property S-N ZLI 4792 <-40 C MLC 6292/000 <-30 C MLC 6292/100 <-40 C Clearing +92 C +120 C Viscosity (mm 2 /s) +20 C 0 C -20 C -40 C 15 40 160 2500 28 95 470 7000 25 85 460 7000 K 33/K 11 1. 39 ------- De Dn 5. 2 0. 0969 7. 4 0. 0903 6. 9 0. 1146 d. Dn (mm) d. V/d. T (m. V/C) 0. 5 2. 55 0. 5 1. 88 0. 5 1. 38 V(10, 0, 20) V(50, 0, 20) V(90, 0, 20) 2. 14 2. 56 3. 21 1. 80 2. 24 2. 85 1. 38 2. 25 2. 83
Summary of Fundamentals • Thermotropic Liquid Crystal • Anisotropy • Nematic phase • Chirality • Order parameters • Dielectric Anisotropy • Diamagnetism • Birefringence • Elastic constants • Surface Anchoring • Viscosity • Threshold • Defects • Eutectic Mixture
- Slides: 80