Liquid Chromatography Mass Spectrometry What it is and























![MS-MS of Droloxifene: Products of m/z 388 [M+H]+ 2: Daughters of 388 AP 3881. MS-MS of Droloxifene: Products of m/z 388 [M+H]+ 2: Daughters of 388 AP 3881.](https://slidetodoc.com/presentation_image/3859c4f6c516f6f773b4ff66a9d82ede/image-25.jpg)










![PGE 2 agonist process-related impurity n if m/z 747 is [2 M+H]+, ms-ms should PGE 2 agonist process-related impurity n if m/z 747 is [2 M+H]+, ms-ms should](https://slidetodoc.com/presentation_image/3859c4f6c516f6f773b4ff66a9d82ede/image-46.jpg)


![PGE 2 agonist process-related impurity n Isotope pattern for the m/z 747 [M+H]+. P+2 PGE 2 agonist process-related impurity n Isotope pattern for the m/z 747 [M+H]+. P+2](https://slidetodoc.com/presentation_image/3859c4f6c516f6f773b4ff66a9d82ede/image-53.jpg)



- Slides: 57

Liquid Chromatography Mass Spectrometry What it is and How to do it Thomas R. Sharp Analytical R&D Pfizer Global Research & Development Groton, CT


Everything You Need to Know About Mass Spectrometry (in 10 minutes or less) n J. J. Thomson (1910) – parabola mass spectrograph – positive and negative ions – isotopes of neon

Mass Spectrometric Alphabet Soup EI CI FAB FIB FD APCI ESP TSP FI ECIN NICI PPINICI GCMS LCMS ICPMS PB MSMS LD MALDI TOF QQQ EBEB BEEB EBQQ ITMS FTMS RIMS LAMMA PDMS ICPMS FTMS ROOMS

Some Concepts to Clarify n n n n relative molecular mass chemical vs monoisotopic molecular mass what the mass spectrometer really measures isotope patterns accurate mass vs. high resolution accurate mass measurements one does not prove a structure: one can only disprove it!

Atomic Weight vs Relative Atomic Mass n n n Weight measures the influence of an external gravitational field on a quantity of matter Mass is a measure of the amount of matter, independent of any gravitational field mass of an atom of 16 O = 1. 65979 x 10 -24 grams 12 C is the current standard the dalton is the accepted atomic mass unit

The Oxygen Standard n n n the a. m. u. was originally defined to be 1/16 the atomic mass of oxygen discovered (1929) to include two minor isotopes – 17 O (0. 04%) and 18 O (0. 2%) chemists’ scale based on wet chemical measure of oxygen’s mass, “in error” because of the presence of the minor isotopes physicists’ scale focused on intrinsic mass of the 16 O isotope scales differed by 0. 0044 amu IUPAC standardization efforts began in 1956

The Carbon Standard IUPAC adopted the 12 C standard in 1960 n amu = 1/12 of the mass of a 12 C atom n defined in reference to a specific isotope rather than on an element (including all of its naturally occurring isotopes) n simple conversion of all chemists’ and physicists’ tabulations to the 12 C standard n


I conceive, therefore, that when we say the atomic weight of, for instance, calcium is 40, we really express the fact that, while the majority of calcium atoms have an actual atomic weight of 40, there are not a few of which are represented by 39 or 41, a less number by 38 or 42, and so on. Is it not possible… that these heavier and lighter atoms may have been… sorted out by a process resembling chemical fractionation? This sorting out may have taken place… while atomic matter was condensing. . . , but also… in geological ages by successive solutions and reprecipitations of the various earths. … I do not think it beyond the power of chemistry to test this feasibility. W. Crookes (1886) address to the British Association, Birmingham

Monoisotopic vs. Chemical Relative Molecular Mass n n n n relative molecular mass (r. m. m. ), in daltons chemical (average) -- using periodic table atomic masses (weighted averages) monoisotopic -- using the lowest mass stable isotope (an arguable rule) nominal (integral) mass -- no decimal places accurate (exact) mass -- four decimal places mass defect (sufficiency or deficiency) which one to use?

Monoisotopic accurate masses of selected elements n 12 C n 13 C n 1 H n 14 N n 16 O n 18 O n 19 F n 32 S n 34 S n 35 Cl n 37 Cl – 12. 00000 – 13. 00336 – 1. 007825 – 14. 00307 – 15. 99491 – 17. 99916 – 18. 9984 – 31. 9721 – 33. 96787 – 34. 9689 – 36. 9659 n H 0 – 1. 007825 H+ – 1. 007276 n Sources of masses and abundances: n – P. De. Bievre, I. L. Barnes (1985). Int’l J. Mass Spectrometry & Ion Processes 65, 211 -30. – CRC Handbook – O. A. Mamer & A. Lesimple (2004). JASMS 15, 626 – your friendly local mass spectroscopist

An Example: hexatriacontane, C 36 H 74 n n n C 36 x 12. 0000 = 432. 0000 H 74 x 1. 0078 = 74. 5772 –––– 506. 5772 12. 011 = 432. 396 1. 008 = 74. 592 ––––––– 506. 988 65 hydrogen atoms contribute 0. 5 da of excess mass 36 x 12 = 432 74 x 1 = 74 ––––––– 506

An Example: C 14 H 12 NOFCl 2 n n n C 14 x 12. 0000 = 168. 0000 12. 001 = 168. 154 H 12 x 1. 0078 = 12. 0936 1. 008 = 12. 096 N 1 x 14. 0031= 14. 0031 14. 007 = 14. 007 O 1 x 15. 9949 = 15. 9949 15. 999 = 15. 999 F 1 x 18. 9984 = 18. 9984 18. 998 = 18. 998 35 Cl 2 x 34. 9689 = 69. 9378 35. 45 = 70. 900 ––––––– 299. 0278 300. 154

High Resolution vs Accurate Mass Measurement high resolution -- ability to distinguish different elemental compositions n accurate mass measurement -- precise relative mass measurement (to 4 decimal places or more) n high resolution nominal mass measurement n low resolution accurate mass measurement n see K. Biemann (1990) Methods in Enzymology 193, 295 -305 n

LC-MS -- a difficult courtship P. J. Arpino (1982). Trends in Analytical Chemistry 1, 154.

Comparison of Gas Loads Packed column GC -- 3 -5 m. L/min n Capillary column GC -- 0. 5 - 1. 5 m. L/min n Conventional HPLC -- 1 -3 m. L/min n » hexane » chloroform » methanol » water -- 180 - 540 m. L/min -- 280 - 840 m. L/min -- 350 - 1650 m. L/min -- 1250 - 3720 m. L/min

LC-MS Historical Perspective n DLI (direct liquid introduction) – Tal’rose et al. (1968) Russian J Phys Chem 42, 1658 -64. – Baldwin & Mc. Lafferty (1973) Org Mass Spectrom 7, 1111 -12. n Moving belt interface – Scott et al. (1974) J Chromatog 99, 395 -405. – Mc. Fadden et al. (1976) J Chromatog 122, 389 -96. n API and APc. I – Horning et al. (1974) J Chromatog Sci 12, 725 -9.

LC-MS Historical Perspective n Particle Beam (MAGIC) – Willoughby & Browner (1984) Anal Chem 56, 2625 -31. n Continuous flow FAB – Caprioli, Fan & Cottrell (1986) Anal Chem 58, 2949 -54. n Thermospray – Blakley, Carmody & Vestal (1980) Anal Chem 52, 1636 -41. n Electrospray – Yamashita & Fenn (1984) J Phys Chem 88, 4452 -9.

The LC-MS problem domain

“No instrument is functioning so well that it cannot be disassembled, altered, reassembled and perhaps improved. ” M. L. Vestal Salt Lake City circa 1981

APCI & Electrospray n API = Atmospheric Pressure Ionization – APCI = Atmospheric Pressure Chemical Ionization – ESP = Electrospray Soft ionization (minimal fragmentation) n Applicable to polar, water-soluble molecules (i. e. pharmaceuticals) n ESP - most notable direct application to peptides (pharmaceuticals of the future). n

Tandem Mass Spectrometry (MS-MS) Relative molecular mass is important, but insufficient to identify a structure n Structure elucidation and confirmation n Necessary to fragment the molecular ions produced by APCI & ESP n Products, parents, constant neutral loss by collision-induced decompositon (CID) n Ion-genetic relationships -- true MS-MS n “Up Front CID” -- poor man’s MS-MS n

Droloxifene C 26 H 29 NO 2 r. m. m. 387 n Breast cancer n Osteoporosis n 1: Scan AP+ 1. 21 e 6 388 100 % 389 0 80 107121 129 145 100 120 140 390 223 160 180 200 220 240 260 280 300 320 340 360 380 400 420 440 460 Da/e 480
![MSMS of Droloxifene Products of mz 388 MH 2 Daughters of 388 AP 3881 MS-MS of Droloxifene: Products of m/z 388 [M+H]+ 2: Daughters of 388 AP 3881.](https://slidetodoc.com/presentation_image/3859c4f6c516f6f773b4ff66a9d82ede/image-25.jpg)
MS-MS of Droloxifene: Products of m/z 388 [M+H]+ 2: Daughters of 388 AP 3881. 25 e 4 100 n 20 V collision energy % 72 386 0 Da/e 2: Daughters of 388 AP 4. 18 e 3 72 100 n 50 V collision energy % 388 387 388 71 0 57 60 80 100 120 140 160 180 200 220 240 260 280 300 320 340 360 380 Da/e

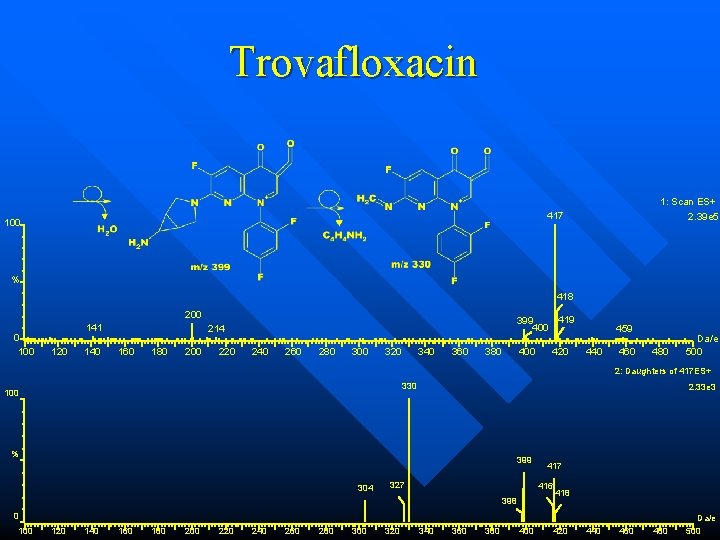
Trovafloxacin “Quinolone” antibiotic n C 20 H 15 N 4 O 3 F 3 r. m. m. 416 n 1: Scan AP+ 4. 28 e 5 417 100 % 373 102 0 100 292 304 120 140 160 180 200 220 240 260 280 300 342344 367 320 340 360 374 380 418 399 400 419 420 458 440 460 480 u/e 500

Trovafloxacin 1: Scan ES+ 2. 39 e 5 417 100 % 418 200 0 100 141 120 140 419 399 400 214 160 180 200 220 240 260 280 300 320 340 360 380 400 420 459 440 460 480 Da/e 500 2: Daughters of 417 ES+ 330 100 2. 33 e 3 % 399 304 327 416 398 418 0 100 120 140 160 180 200 220 240 260 280 300 320 340 360 380 400 420 440 460 480 Da/e 500

HPLC Considerations for LC-MS: Compatible Mobile Phases n Volatile buffer salts! – NH 4 formate, NH 4 acetate (p. H ~4 to 9) – 0. 1% (v/v) trifluoroacetic acid (down to p. H 2) or ammonium trifluoroacetate n Some volatile buffers don’t work well – TFA suppression and the TFA fix – Triethylamine suppression n Substitution of nonvolatile buffer salts

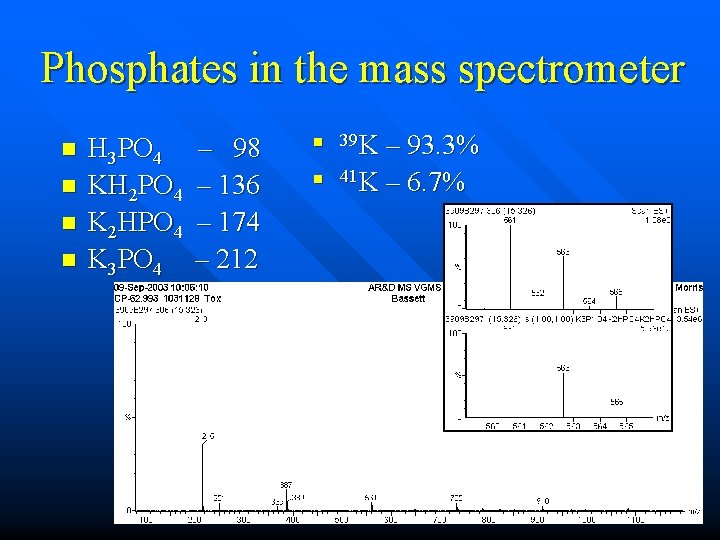
No Phosphates!!!!!! ESP LC-MS using a potassium phosphate mobile phase n Accumulation on the striker plate n A stalagmite or a stalactite? n

Phosphates in the mass spectrometer n n H 3 PO 4 KH 2 PO 4 K 2 HPO 4 K 3 PO 4 – 98 – 136 – 174 – 212 n n 39 K – 93. 3% 41 K – 6. 7%

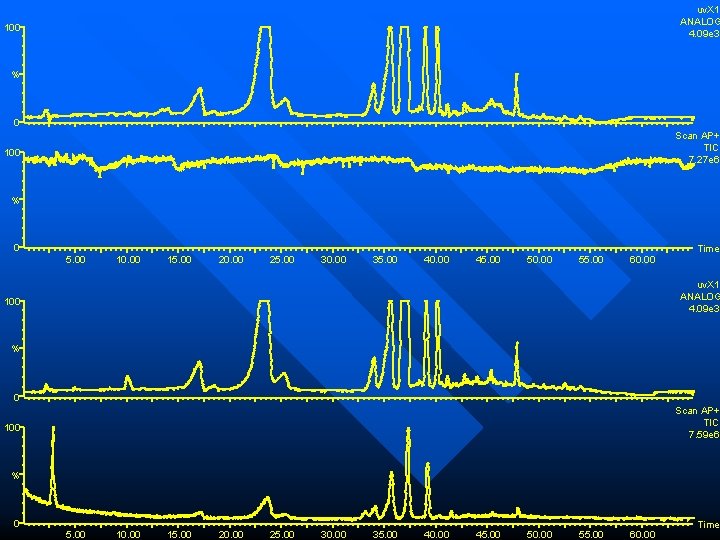
Problems with TEA triethylamine (TEA) a common mobile phase additive for peak shape conditioning n a “volatile” buffer salt n However, it quenches the ion signal!!! n

uv. X 1 ANALOG 4. 09 e 3 100 % 0 Scan AP+ TIC 7. 27 e 6 100 % 0 5. 00 10. 00 15. 00 20. 00 25. 00 30. 00 35. 00 40. 00 45. 00 50. 00 55. 00 60. 00 Time uv. X 1 ANALOG 4. 09 e 3 100 % 0 Scan AP+ TIC 7. 59 e 6 100 % 0 5. 00 10. 00 15. 00 20. 00 25. 00 30. 00 35. 00 40. 00 45. 00 50. 00 55. 00 60. 00 Time

HPLC Considerations for LC-MS: Miscellaneous Points Put a UV-Vis detector in line! n Common organic components are OK n Gradients are OK n Small bore columns are OK n

UV-Active Components that Don’t Respond n “Volatile” small molecules – methyl and propyl parabens – benzoic acid – simple aromatic amines – simple aromatic aldehydes

UV-Silent Components The mass spectrometer is a more universal detector? n Compounds with no UV chromophore n – azithromycin – other azalide antibiotics n UV-silent excipients in drug product formulations

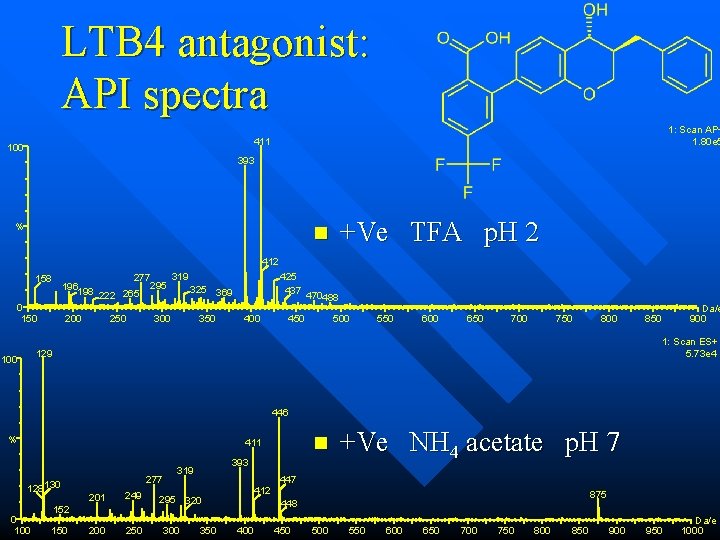
LTB 4 antagonist: API spectra 1: Scan AP+ 1. 80 e 5 411 100 393 % +Ve TFA p. H 2 n 412 319 277 295 196198 325 369 222 265 158 0 150 200 250 300 350 425 437 400 470488 450 500 550 600 650 700 750 800 1: Scan ES+ 5. 73 e 4 129 100 Da/e 900 850 446 % n 411 128 130 0 100 277 201 249 200 250 152 150 393 319 412 295 320 300 350 +Ve NH 4 acetate p. H 7 447 875 448 400 450 500 550 600 650 700 750 800 850 900 950 Da/e 1000

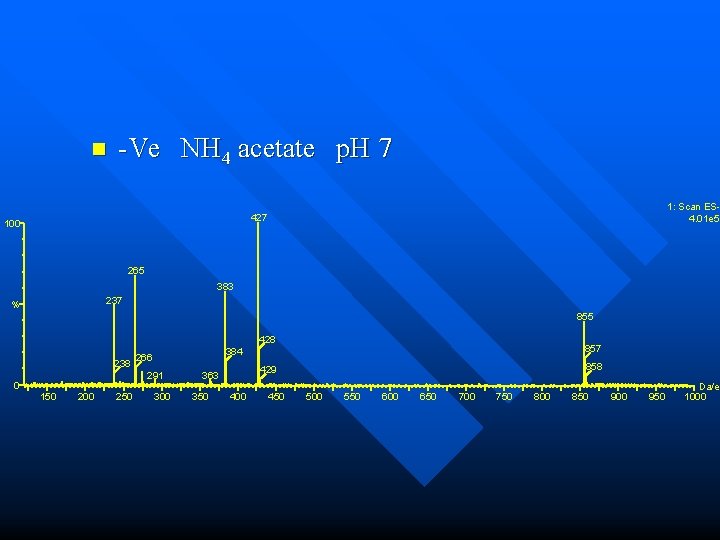
n -Ve NH 4 acetate p. H 7 1: Scan ES 4. 01 e 5 427 100 265 383 237 % 855 428 238 266 291 0 150 200 250 857 384 300 350 858 429 363 400 450 500 550 600 650 700 750 800 850 900 950 Da/e 1000

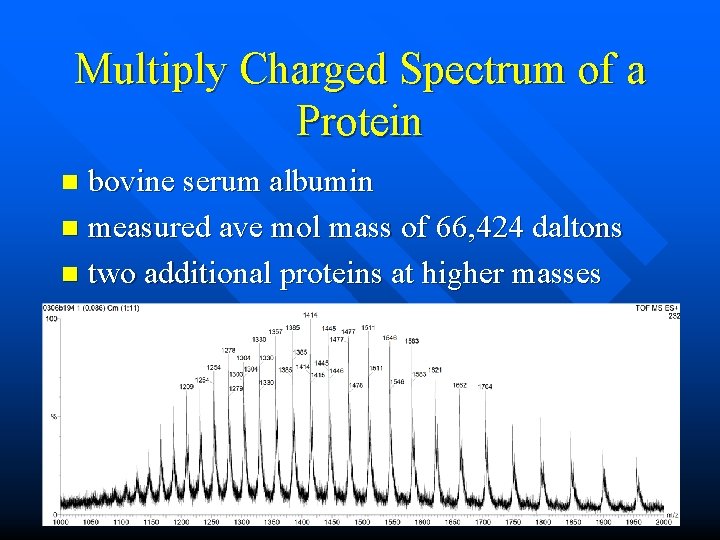
Multiply Charged Spectrum of a Protein bovine serum albumin n measured ave mol mass of 66, 424 daltons n two additional proteins at higher masses n

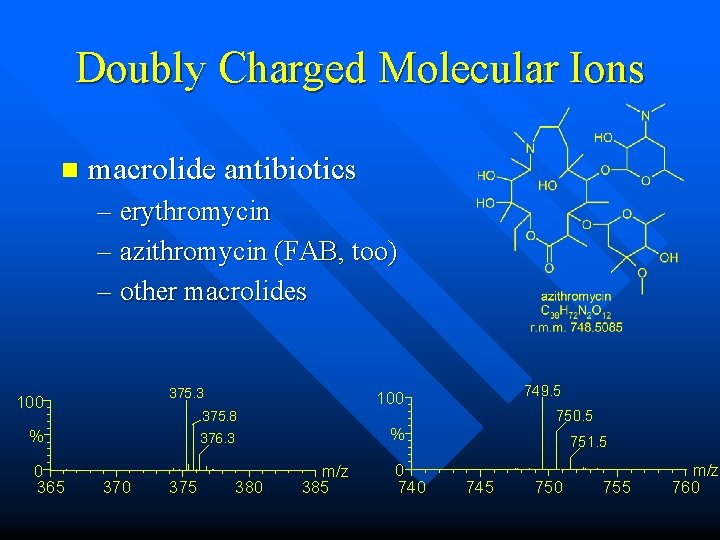
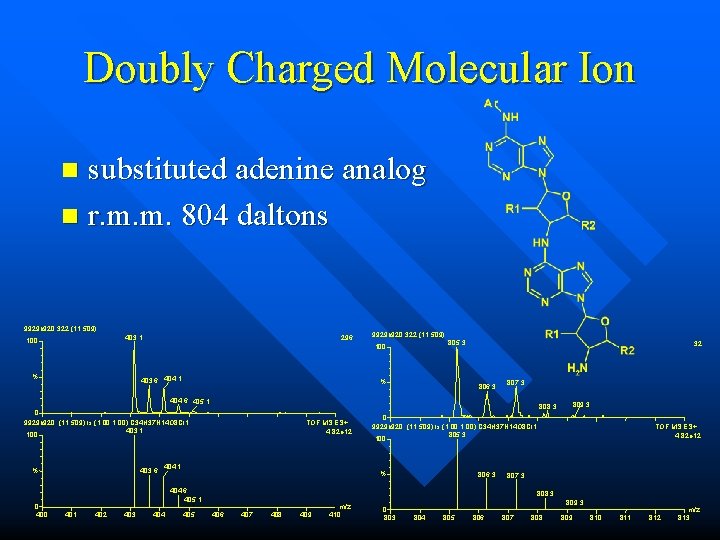
Doubly Charged Molecular Ions n macrolide antibiotics – erythromycin – azithromycin (FAB, too) – other macrolides 375. 3 100 750. 5 375. 8 % 0 365 749. 5 100 % 376. 3 370 375 380 m/z 385 0 740 751. 5 745 750 755 m/z 760

Doubly Charged Molecular Ion substituted adenine analog n r. m. m. 804 daltons n 9929 b 920 322 (11. 509) 403. 1 100 296 9929 b 920 322 (11. 509) 805. 3 100 % 403. 6 404. 1 % 32 806. 3 807. 3 404. 6 405. 1 % 0 403. 6 TOF MS ES+ 4. 82 e 12 404. 1 402 403 404 0 9929 b 920 (11. 509) Is (1. 00, 1. 00) C 34 H 37 N 14 O 8 Cl 1 805. 3 100 % 404. 6 405. 1 405 809. 3 808. 3 0 9929 b 920 (11. 509) Is (1. 00, 1. 00) C 34 H 37 N 14 O 8 Cl 1 403. 1 100 806. 3 TOF MS ES+ 4. 82 e 12 807. 3 808. 3 406 407 408 409 m/z 410 0 803 809. 3 804 805 806 807 808 809 810 811 812 m/z 813

But how small can you go? n Gaskell (1997) predicts most abundant charge state based on number of basic sites – 1885 Da peptide with 4 “basic” sites predicts +4 charge state most abundant n peptides from digests -- as small as 950 Da – abundant [M+2 H]2+ – dependent upon instrumentation – dependent upon operating conditions

How low can you go?

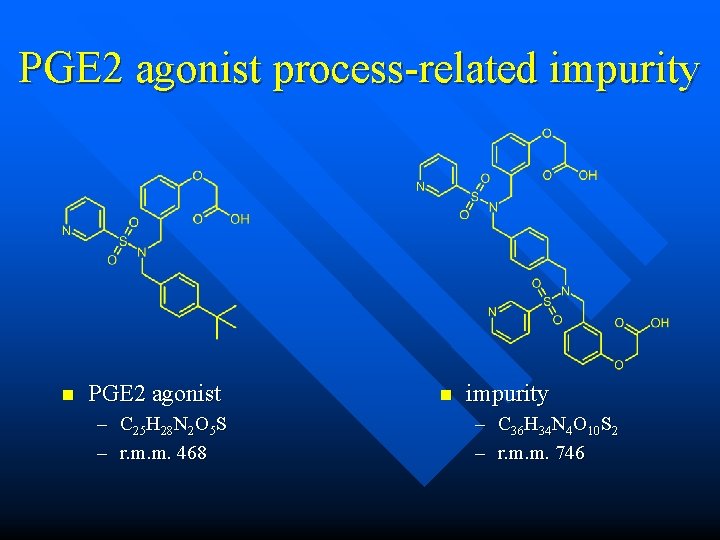
PGE 2 agonist process-related impurity n PGE 2 agonist – C 25 H 28 N 2 O 5 S – r. m. m. 468

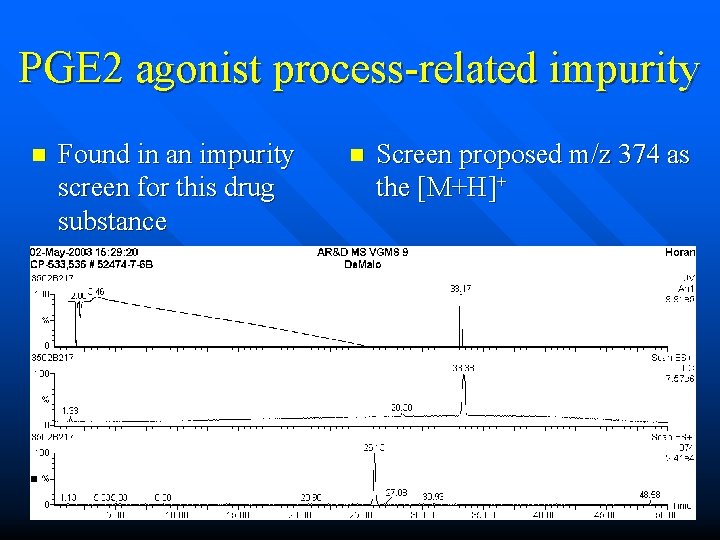
PGE 2 agonist process-related impurity n Found in an impurity screen for this drug substance n Screen proposed m/z 374 as the [M+H]+

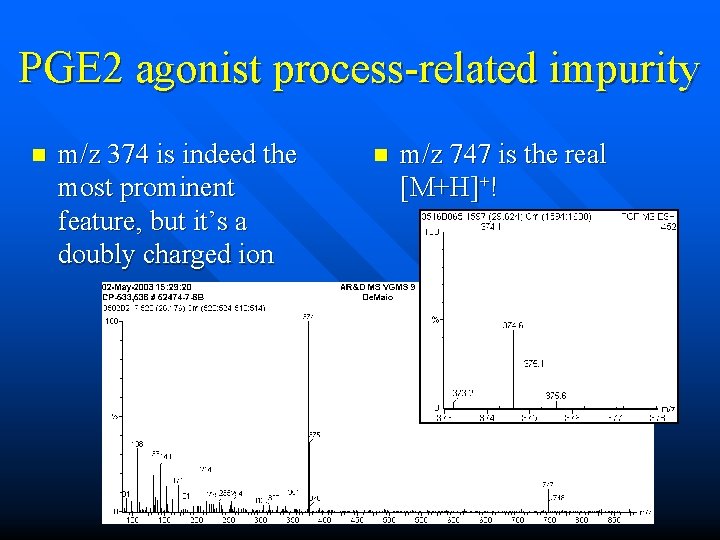
PGE 2 agonist process-related impurity n m/z 374 is indeed the most prominent feature, but is it the real molecular ion? n n molecular ion and protonbound dimer? molecular ion and doubly charged mol ion?
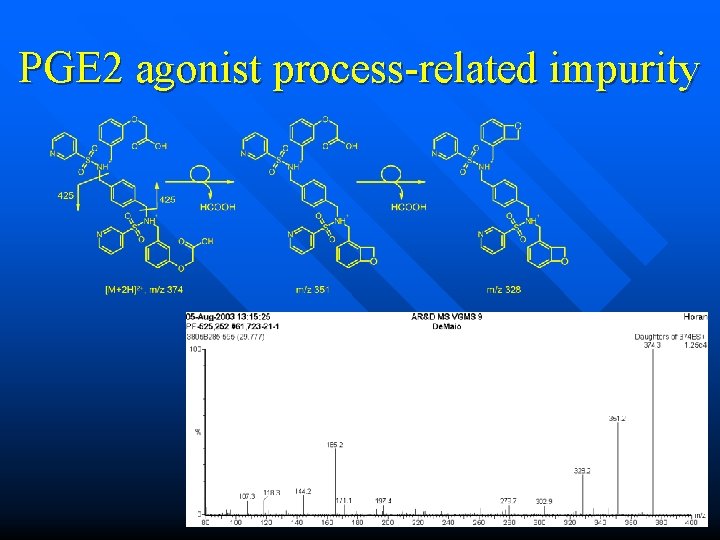
![PGE 2 agonist processrelated impurity n if mz 747 is 2 MH msms should PGE 2 agonist process-related impurity n if m/z 747 is [2 M+H]+, ms-ms should](https://slidetodoc.com/presentation_image/3859c4f6c516f6f773b4ff66a9d82ede/image-46.jpg)
PGE 2 agonist process-related impurity n if m/z 747 is [2 M+H]+, ms-ms should generate m/z 374 n unusual 23 dalton differences in ms-ms of m/z 374

PGE 2 agonist process-related impurity n m/z 374 is indeed the most prominent feature, but it’s a doubly charged ion n m/z 747 is the real [M+H]+!

PGE 2 agonist process-related impurity n PGE 2 agonist – C 25 H 28 N 2 O 5 S – r. m. m. 468 n impurity – C 36 H 34 N 4 O 10 S 2 – r. m. m. 746

PGE 2 agonist process-related impurity

Polyisotopic Elements Chemical atomic mass vs monoisotopic atomic mass n Carbon -- 12. 011 (periodic table) n – 12 C – n - 12. 0000 - 98. 9% 13 C - 13. 0033 - 1. 1% Chlorine -- 35. 453 (periodic table) – 35 Cl – - 34. 9689 - 75. 8% 37 Cl - 36. 9659 - 24. 2%

Isotope patterns for C 14 H 12 NOFCl 2, r. m. m. 299 Da. n m/z 269 is neutral loss of methylamine n m/z 234 is further loss of Cl 1 n isotope patterns match predictions n

PGE 2 agonist process-related impurity n PGE 2 agonist – C 25 H 28 N 2 O 5 S – r. m. m. 468 n impurity – C 36 H 34 N 4 O 10 S 2 – r. m. m. 746
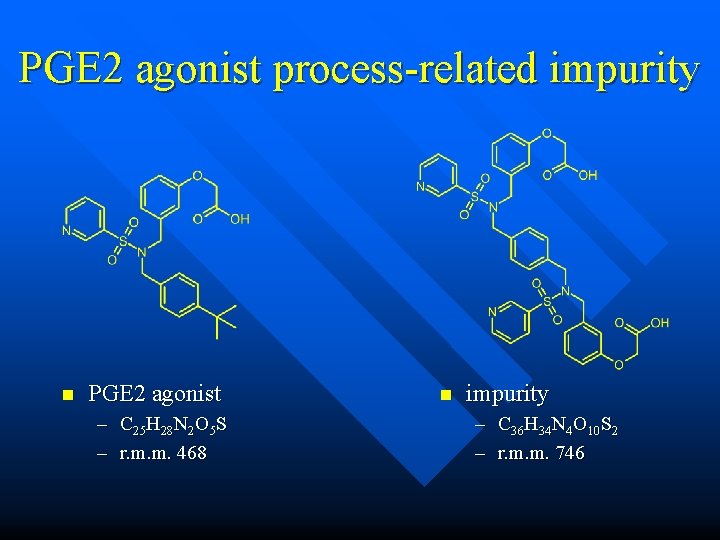
![PGE 2 agonist processrelated impurity n Isotope pattern for the mz 747 MH P2 PGE 2 agonist process-related impurity n Isotope pattern for the m/z 747 [M+H]+. P+2](https://slidetodoc.com/presentation_image/3859c4f6c516f6f773b4ff66a9d82ede/image-53.jpg)
PGE 2 agonist process-related impurity n Isotope pattern for the m/z 747 [M+H]+. P+2 is 24. 6% of P. (34 S = 4. 2%) n prediction for elemental composition with two sulfur atoms n prediction replacing sulfurs with carbon and hydrogen

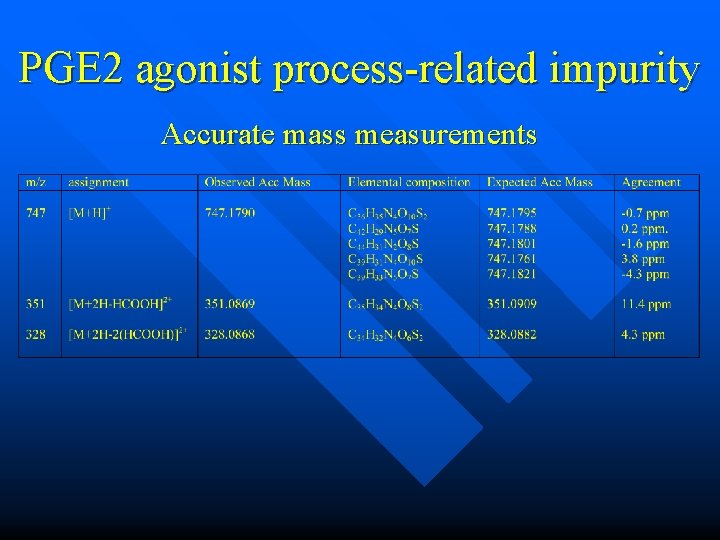
PGE 2 agonist process-related impurity Accurate mass measurements

Concepts Clarified relative molecular mass n chemical vs monoisotopic molecular mass n what the mass spectrometer really measures n isotope patterns n accurate mass vs. high resolution n accurate mass measurements n contributions to a structure identification n


Words of Wisdom: It is as important to understand what a technique CANNOT tell you as it is to understand what it can tell you.