LIQUID CARBON DIOXIDEBASED LEATHER PROCESS Jeremy Kroon Department

LIQUID CARBON DIOXIDEBASED LEATHER PROCESS Jeremy Kroon Department of Chemistry and Biochemisty South dakota state University

Purpose of Tanning � � � � � To bring about irreversible stabilization of the skin substance that is to prevent putrefaction. stabilize it against enzymatic degrading and increase its resistance to chemicals raise its shrinking temperature and increase its resistance to hot water reduce or eliminate its ability to swell enhance its strength properties lower its density by isolating the fibers reduce its deformability reduce its shrinkage in volume, area and thickness enhance the porosity of its fiber texture.
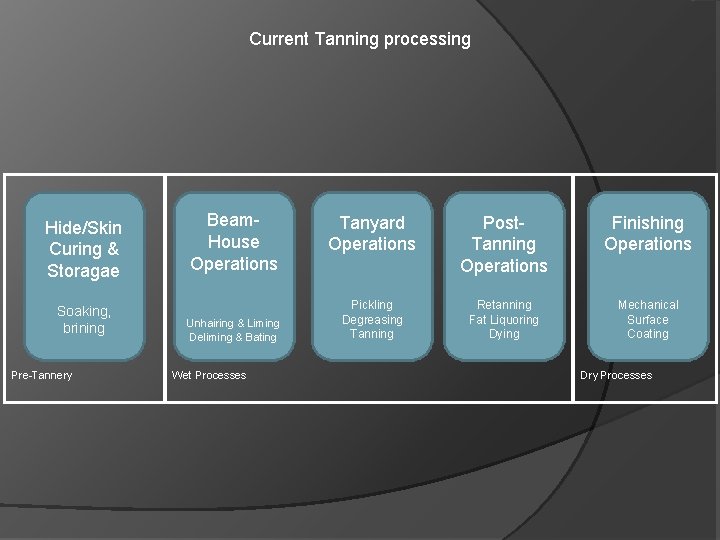
Current Tanning processing Hide/Skin Curing & Storagae Soaking, brining Pre-Tannery Beam. House Operations Unhairing & Liming Deliming & Bating Wet Processes Tanyard Operations Post. Tanning Operations Finishing Operations Pickling Degreasing Tanning Retanning Fat Liquoring Dying Mechanical Surface Coating Dry Processes
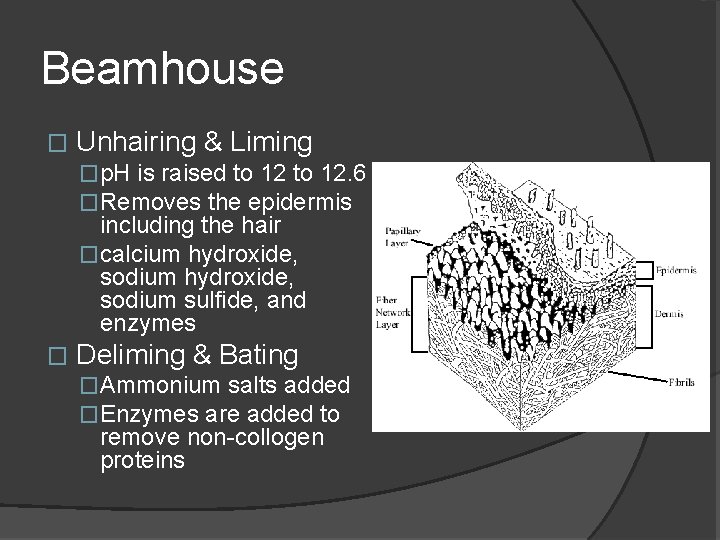
Beamhouse � Unhairing & Liming �p. H is raised to 12. 6 �Removes the epidermis including the hair �calcium hydroxide, sodium sulfide, and enzymes � Deliming & Bating �Ammonium salts added �Enzymes are added to remove non-collogen proteins
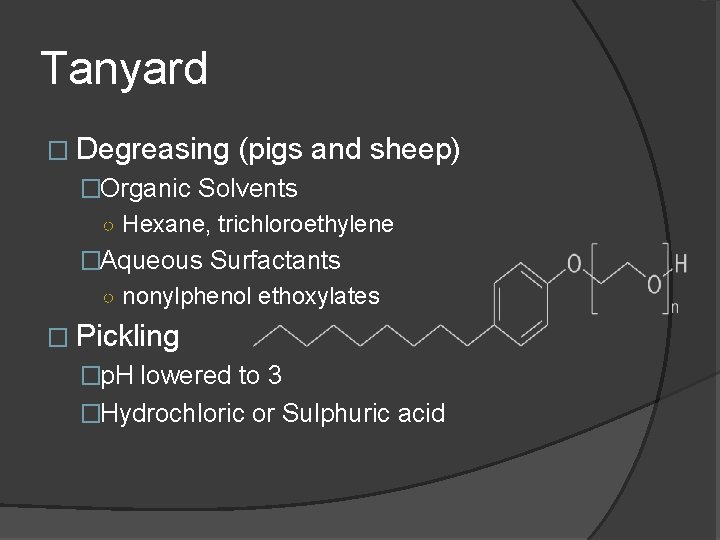
Tanyard � Degreasing (pigs and sheep) �Organic Solvents ○ Hexane, trichloroethylene �Aqueous Surfactants ○ nonylphenol ethoxylates � Pickling �p. H lowered to 3 �Hydrochloric or Sulphuric acid
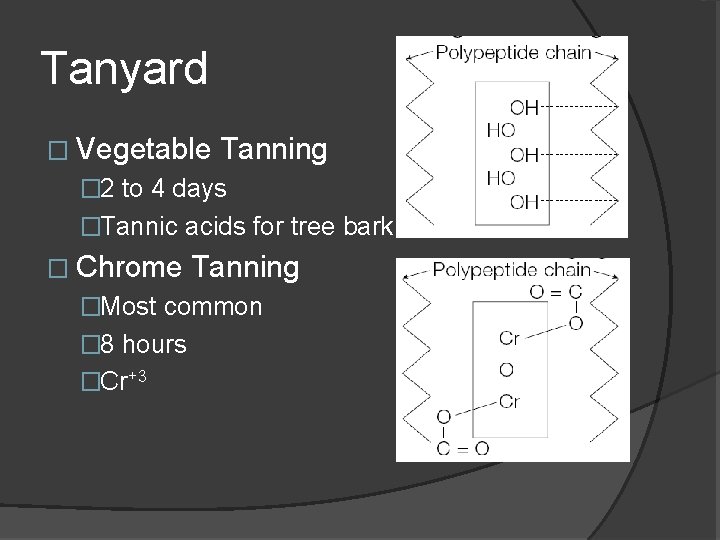
Tanyard � Vegetable Tanning � 2 to 4 days �Tannic acids for tree bark � Chrome Tanning �Most common � 8 hours �Cr+3

Post Tanning Operations � Neutrilizing � Brings hides back to neutral p. H � Retanning � Vegetable tannins, syntans � Used to impart specific characteristics � Dyeing � Anionic dyes � Fatliquoring � Oils added back to leather

Disadvantages � metric ton of salted cattle hides � 15 -50 m 3 of wastewater ○ 5 -6 kg of chromium ○ 10 kg of sulfide � 40 kg of volatile organics (VOC) �. 8 x 106 to 4. 0 x 107 BTU of energy � Capital investment for pollution control purposes can be as high as 50% of the total value of plant

Leather and CO 2 � Replacing Water with liquid CO 2 �diffusion advantage �simple adjustments of temperature and pressure may result in the precipitation of residual leather treatment agents ○ allows subsequent recycling or disposal of these materials �cheap and readily accepted

Approach � Dyeing and waterproofing �greatest probability of success � Degreasing and Fat liquoring �Fats and oils are highly soluble � Tanning and retanning �Largest challenge since chrome ions are highly insoluble �Focus largely on vegetable tannins and syntans

Waterproofing � fluorocarbons, silicones, waxes, aqueous-based oils, and fluoropolymers �Each was loaded onto cotton �Extracted using supercritical CO 2 at 2500 psi and 50°C �All were found to be soluble

Waterproofing � Allowed to soak with leather sample � Mass loading showed equal weight to manufactures' method � No caking, or discoloration Oil Based Agent CO 2 treatment Manufactures’ Silicone Based Agent CO 2 treatment Manufactures’

Dyeing � CO 2 dyeing currently commercialized in textile industry ○ Introduced in early 1990’s ○ Even color distribution

Degreasing and Fat Liquoring � Been used in oilseed extraction � Supercritical CO 2 limited use in leather industry � reported degreasing efficiencies up to 94% � With sheep reported no damage to the skin structure
Tanning and Retanning � Tanning agents �Metal tanning ○ Not soluble in CO 2 �Vegetable tanning ○ Soluble ○ Make hard leather � Saddles, belts, etc. �Syntans ○ Soluble ○ Mainly for retanning ○ Used to impart specific characteristics

Conclusion � Our goal for this project is to develop a technique that will make leather production: �FASTER �MORE ECONOMICAL �CLEANER

Acknowledgements � Dr. Raynie � Environmental � BASF Protection Agency

- Slides: 18