Lipidsoluble vitamins They are nonpolar hydrophobic isoprene derivatives
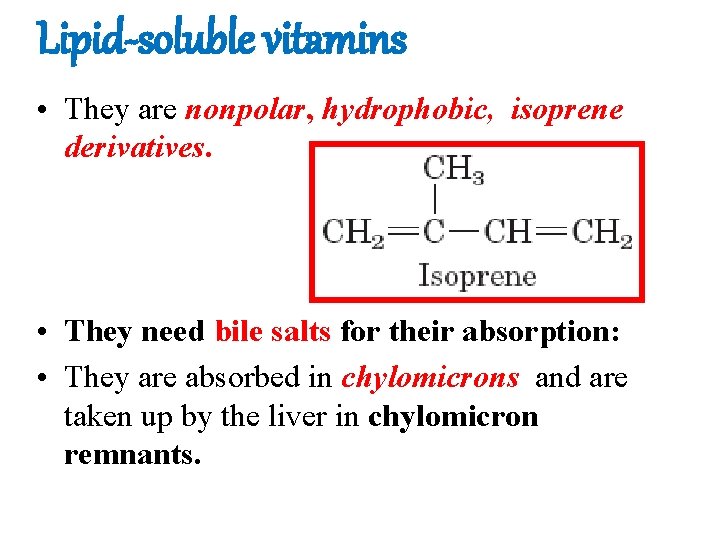
Lipid-soluble vitamins • They are nonpolar, hydrophobic, isoprene derivatives. • They need bile salts for their absorption: • They are absorbed in chylomicrons and are taken up by the liver in chylomicron remnants.

Storage • They are stored in the body (mainly in the liver) and their excess intake causes toxicity (vitamin E is an exception)

Transport • They are transported in blood in association with lipoproteins: VLDL and LDL (vitamin E and K) • or in association with specific binding proteins: retinol-binding protein (RBP) for vitamin A and vitamin D-binding protein (DBP) for vitamin D.
Vitamin A (Anti-xerophthalmic, Anti-night blindness)
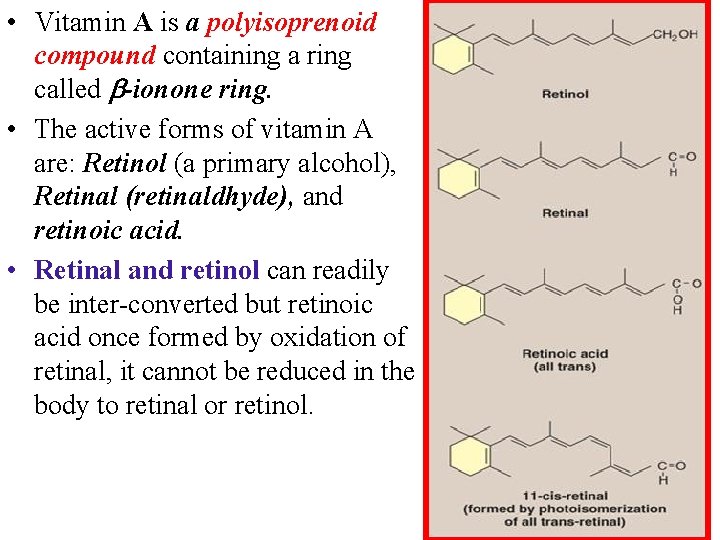
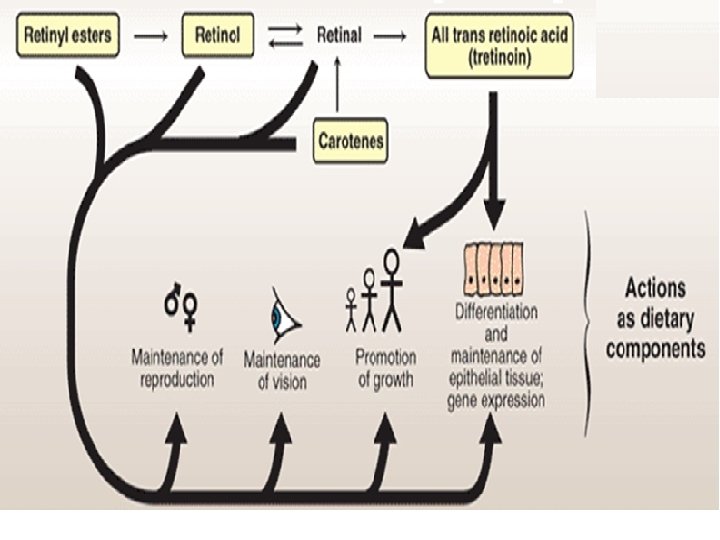
• Vitamin A is a polyisoprenoid compound containing a ring called -ionone ring. • The active forms of vitamin A are: Retinol (a primary alcohol), Retinal (retinaldhyde), and retinoic acid. • Retinal and retinol can readily be inter-converted but retinoic acid once formed by oxidation of retinal, it cannot be reduced in the body to retinal or retinol.

CAROTENOIDS: • Are -carotene-like plant compounds which yield retinoids upon metabolism. • -carotene is called provitamin A. • In the intestinal mucosal cells, -carotene is cleaved into 2 molecules of retinal by carotenase enzyme. • This conversion is inefficient in humans, and the vitamin A activity of β-carotene is only about one twelfth that of retinol.

SOURCES: 1 -Carotenoids (provitamin A): present in dark green leafy and pigmented vegetables and fruits (carrots, peatches, apricots, sweet potato). Carotenoids are also present in cow milk 2 - Animal sources (active form of vitamin A, retinyl esters): esters Cod liver oil, liver, kidney, butter.

DAILY REQUIREMENT: • The RDA for adults is 1, 000 retinol activity equivalents (RAE) for males and 800 RAE for females. • In comparison, 1 RAE = 1 mg of retinol, 12 mg of β-carotene, or 24 mg of other carotenoids.

ABSORPTION AND TRANSPORT: (1) Retinyl esters taken in animal food are hydrolyzed in the intestinal lumen into free retinol and free fatty acids. (2) Free retinol and intact carotenoids, taken in plant food, form micelles which they diffuse into mucosal cells. 3) Inside intestinal mucosal cells, retinol is re-esterified with a long chain fatty acid and is incorporated in chylomicrons. -carotene is cleaved by -carotenase ( -carotene dioxygenase) enzyme into two molecules of retinal (retinaldehyde). • Retinal is subsequently reduced into retinol by retinaldehyde reductase. • Retinol is then esterified and incorporated in chylomicrons
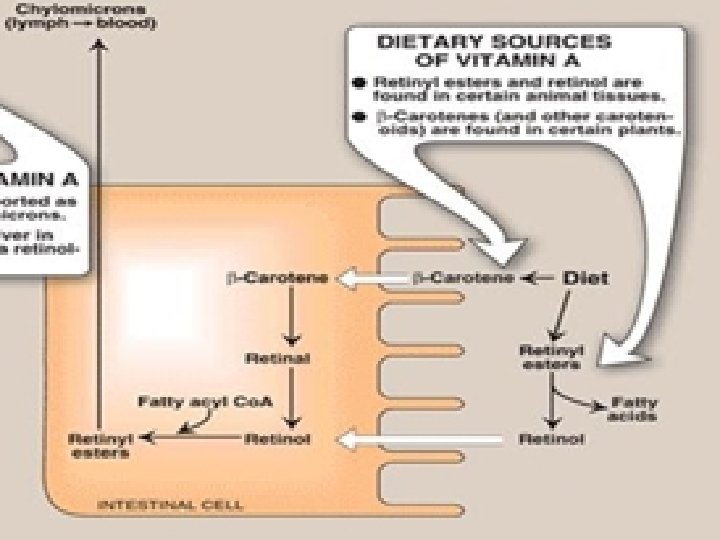

ABSORPTION AND TRANSPORT: (5) Retinyl esters and intact -carotene (which escape carotenase cleavage) are taken by liver (in chylomicron remnants). Liver cells store retinol mainly as retinyl palmitate. (6) retinol is hydrolyzed from its ester and is bound to a retinol-binding protein (RBP). Retinol-RBP complex attaches to specific cell surface receptors on peripheral cells Inside cells, retinol is attached to a cellular RBP which carries it to the nucleus to act in a manner analogous to that of steroid. • N. B. Retinoic acid is not carried by RBP in blood but carried by albumin. Intact -carotene reaching the liver is distributed in VLDL lipoproteins, to be stored in adipose tissue.
FUNCTIONS OF VITAMIN A:
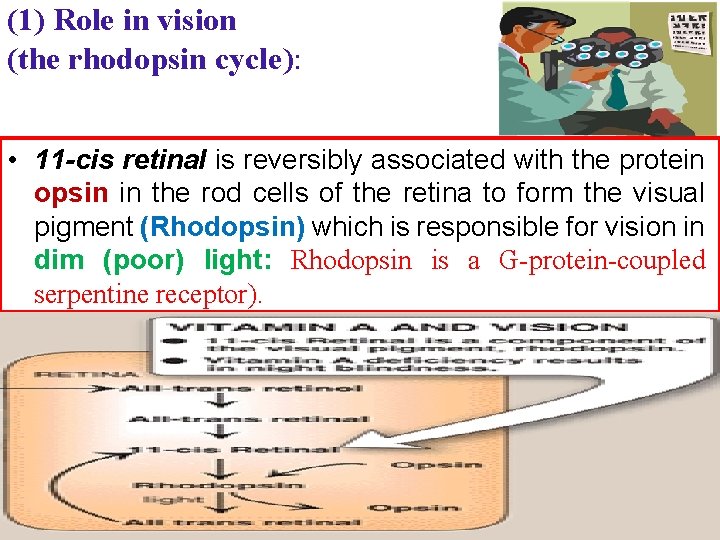
(1) Role in vision (the rhodopsin cycle): • 11 -cis retinal is reversibly associated with the protein opsin in the rod cells of the retina to form the visual pigment (Rhodopsin) which is responsible for vision in dim (poor) light: Rhodopsin is a G-protein-coupled serpentine receptor).
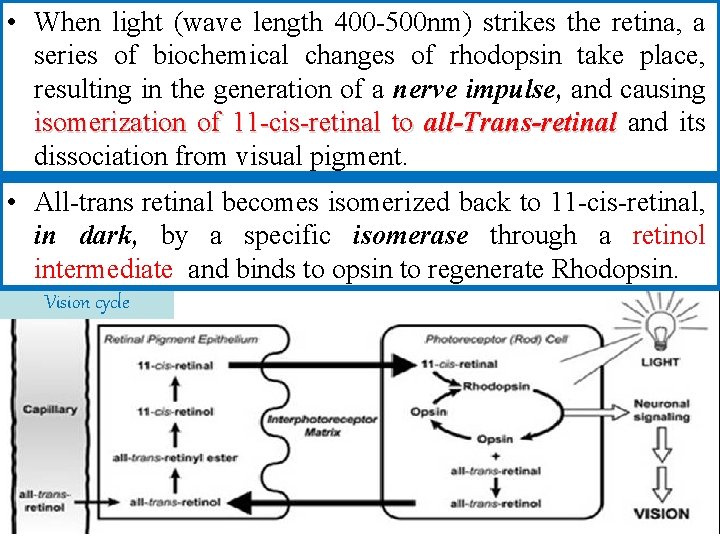
• When light (wave length 400 -500 nm) strikes the retina, a series of biochemical changes of rhodopsin take place, resulting in the generation of a nerve impulse, and causing isomerization of 11 -cis-retinal to all-Trans-retinal and its dissociation from visual pigment. • All-trans retinal becomes isomerized back to 11 -cis-retinal, in dark, by a specific isomerase through a retinol intermediate and binds to opsin to regenerate Rhodopsin. Vision cycle

(2) Role in cell growth and differentiation: • Retinol and retinoic acid (RA) stimulate the transcription of certain genes required for growth and bone development) • N. B. Vitamin A is required for the differentiation of immune cells

(3) Role in maintenance of healthy epithelial cells: • Retinol is converted to retinyl phosphate which serves as glycosyl carrier in the synthesis of glycoproteins and mucopolysaccharide needed for normal mucous secretion

(4) Role in reproduction: • Retinol, but not retinoic acid support spermatogenesis in male and prevent fetal resorption in female rats. • Retinoic acid is inactive in maintaining reproduction and in the visual cycle, but promotes growth and differentiation of epithelial cells; thus, animals given vitamin A only as retinoic acid from birth are blind and sterile.

(5) Role as an antioxidant: • -carotene trap free radicals at low O 2 pressures. • It may protect against cancer and heart attack.

DEFICIENCY: Develop only over prolonged periods (2 -3 months) of inadequate intake because vitamin A is stored in liver. may occur due to • Decrease dietary intake • Biliary obstruction • Advanced liver disease (decrease retinol-binding proteins) 1) Night blindness ------------------ 2) Xeroderma (Rough keratinized skin: This causes fissures in the skin which predispose to infection). 3) Growth retardation 4) Increased susceptibility to infection: a) Because of keratinizing metaplasia of mucosal cell lining as well as loss of protective mucin secretion fissures occur. b) Vitamin A deficiency causes impaired immune system because it is important for differentiation of immune cells.

(1) Night blindness • The earliest signs of vitamin A deficiency. • In severe deficiency xeropthalmia (dryness of conjunctiva and cornea due to loss of conjunctival mucin). • If untreated, xerophthalmia results in corneal degeneration and ulceration (keratomalacia) and, ultimately, in blindness because of the formation of opaque scar tissue.

TOXICITY (hypervitaminosis A): • Pregnant women should not ingest excessive quantities of vitamin A because of its potential for causing congenital malformations in the developing fetus.

• Toxicity occurs due to ingestion of massive doses of vitamin A to treat acne or to prevent cold (rare): • Dry, pruritic scaly skin, loss of hair, brittle nails. • Dizziness, nausea, vomiting, • Bone pains. • Enlarged liver (hepatomegaly): accumulation of vitamin A in the liver. • A rise in intracranial pressure may mimic the symptoms of a brain tumor. Notes • Excessive carotene intake has no serious effects. • The skin becomes yellow but the sclera remains white.
Vitamin E (Tocopherol) (Rat Antisterility Vitamin)
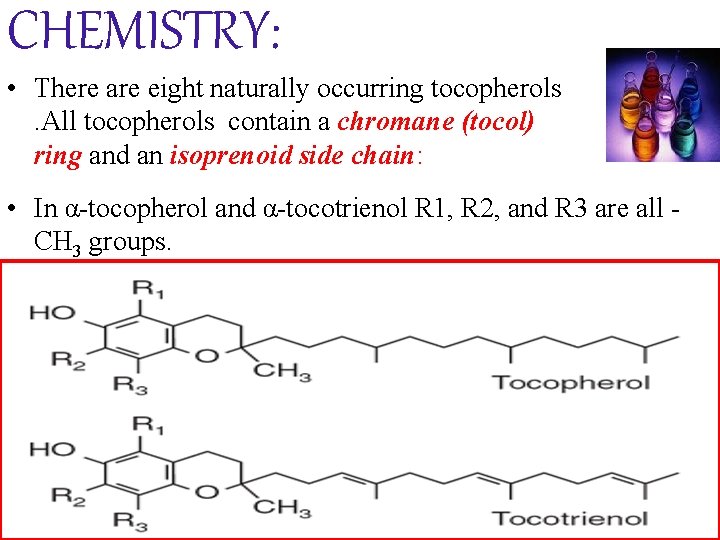
CHEMISTRY: • There are eight naturally occurring tocopherols. All tocopherols contain a chromane (tocol) ring and an isoprenoid side chain: • In α-tocopherol and α-tocotrienol R 1, R 2, and R 3 are all CH 3 groups.

CHEMISTRY: • Tocopherols contain hydroxyl group at C 6 which is responsible for the antioxidant activity of the vitamin. • Tocopherols lose their biologic activity by oxidation (by light and UV rays). • -Tocopherol has the widest distribution and the greatest biological activity

SOURCES: • Vitamin E is synthesized only by plant. • The richest sources are plant (vegetable) oils: cotton seed, corn, wheat germ and soyabean oils. Food rich in PUFA is also rich in vitamin E • Animal sources: sources egg, meat and liver. • The RDA for α-tocopherol is 10 mg for men and 8 mg for women. • Requirement is increased with increase polyunsaturated fatty acids (PUFA).

ABSORPTION AND TRANSPORT: • It is absorbed in chylomicrons. It is taken up by the liver from chylomicron remnant and released in blood in VLDL. • Tocopherol exchanges rapidly between the lipoproteins and erythrocyte membranes.

FUNCTIONS: • (1) Vitamin E acts as a chain-breaking, free radical trapping antioxidant in cell membranes and plasma lipoproteins. It reacts with the lipid peroxide radicals formed by peroxidation of polyunsaturated fatty acids (PUFA) before they can establish a chain reaction. • The tocopheroxyl free radical product is relatively unreactive and ultimately forms nonradical compounds.

• It tends to concentrate in cell membranes especially RBCs membranes, So, it protects against hemolysis and damage of tissues. • It is also present in the surface coat of LDL where it prevents its oxidation and, thus, it protects against atherosclerosis and coronary artery diseases.

FUNCTIONS: • (2) Tocopherol is essential for normal spermatogenesis and continuation of pregnancy in animals.

DEFICIENCY: • Is almost entirely restricted to premature infants. • When observed in adults, it is usually associated with defective lipid absorption or transport.

Manifestations: • 1 - Hemolytic anemia of newborn. • It occurs in premature infants who are born with inadequate vitamin E reserves. Their erythrocyte membranes are abnormally fragile as a result of peroxidation. • 2 - Nerve and muscle membrane damage. • 3 - Sterility and abortion in animals (not in human).

TOXICITY OF VITAMIN E: • Vitamin E is the least toxic. • No toxicity has been observed at doses of 300 mg/day.
Vitamin D (Anti-Rickets) (Anti-osteomalcia)
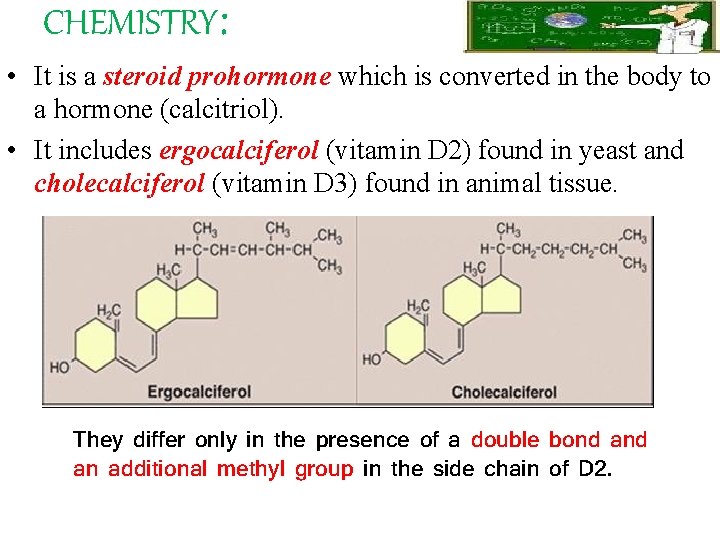
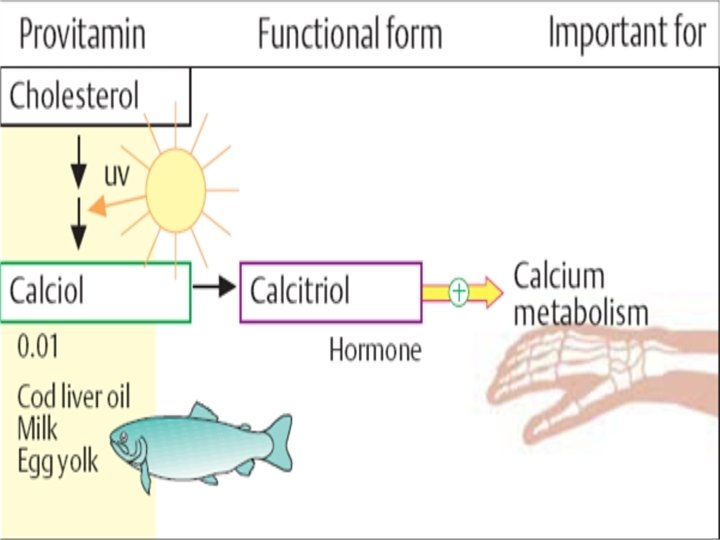
CHEMISTRY: • It is a steroid prohormone which is converted in the body to a hormone (calcitriol). • It includes ergocalciferol (vitamin D 2) found in yeast and cholecalciferol (vitamin D 3) found in animal tissue. They differ only in the presence of a double bond an additional methyl group in the side chain of D 2.

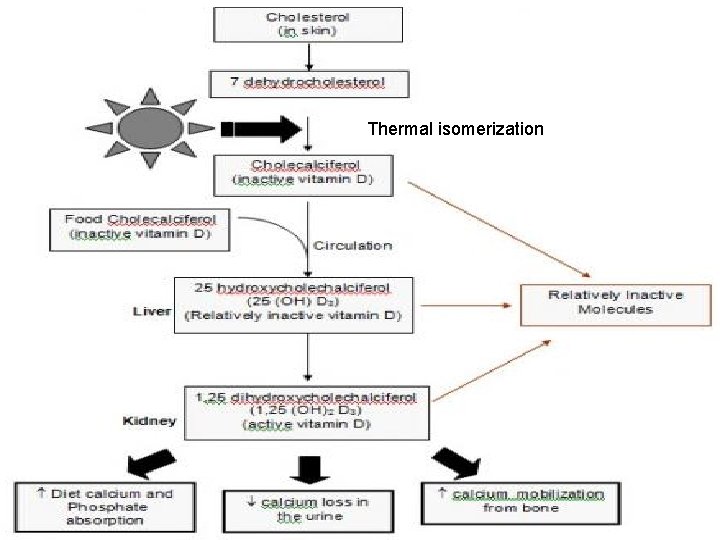
SOURCES: 1. Endogenous Vitamin D 3 (Cholecalciferol) Is produced in the skin by UV irradiation of 7 dehydrocholesterol, dehydrocholesterol an intermediate in cholesterol synthesis, present in subcutaneous fat. If the body is exposed to adequate sunlight, there's no dietary requirement. 2. Dietary source: a)Animal sources: provide vitamin D 3 (cholecalciferol) e. g. salt water fish (Salmon, Sardines), liver egg yolk, cod liver oil. Milk, unless fortified is not a good source of vitamin D b) Yeast: provide vitamin D 2 (ergocalciferol). Vitamin D is very stable in food and is stable to storage, processing and cooking.
Thermal isomerization

DAILY REQUIREMENT: • The RDA for adults is 5 mg of vitamin D (cholecalciferol), or 200 international units (IU) of vitamin D. • If the body is exposed to adequate sunlight, there is almost no dietary need.

ABSORPTION AND METABOLISM: • Vitamin D is absorbed in chylomicrons and is transferred from chylomicron surface to its specific binding protein in plasma (vitamin Dbinding protein = DBP). Vitamin D is taken up by the liver, in chylomicron remnants, hydroxylated at C 25 and released in plasma bound to DBP. • Ergosterol is poorly absorbed from intestine.
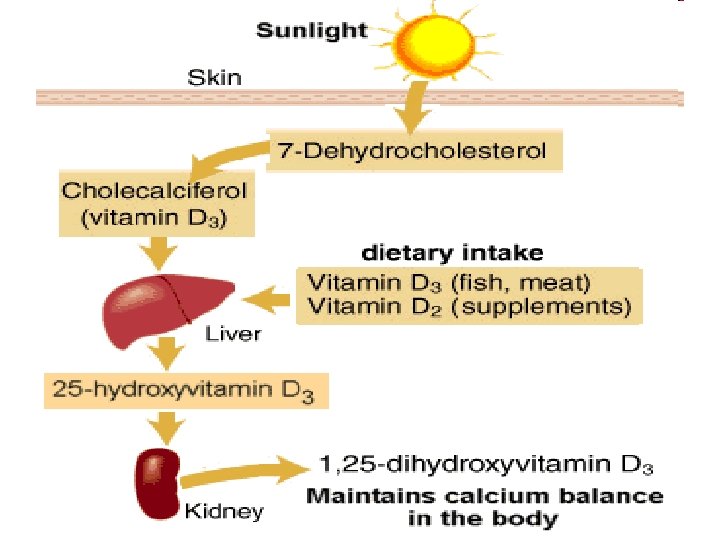

Important • Vitamins D 3 (and D 2) are converted in vivo to the active form of the D vitamin by two sequential hydroxylation reactions:

st 1 Hydroxylation reaction • The first hydroxylation occurs in the liver, at C 25. The product of the reaction, 25 hydroxycholecalciferol (25 -OH-D 3), (25 -OH-D 3) is the predominant form of vitamin D in the plasma and the major storage form of the vitamin.

nd 2 Hydroxylation reaction 25 -OH-D 3 is further hydroxylated at C 1 position by a specific 25 -hydroxycholecalciferol 1 -hydroxylase found primarily in the kidney, resulting in the formation of 1, 25 di. OH-D 3 (calcitriol).

Note: Both hydroxylases employ cytochrome P 450, molecular oxygen, and NADPH

Regulation of vit D • Formation of active vitamin D is tightly regulated by the level of plasma phosphate and calcium ions. • This regulation occurs on kidney 1 hydroxylase:

Regulation of vit D • 1 - ↓ plasma phosphate directly increases 1 -hydroxylase activity. • 2 - ↓ plasma calcium indirectly increases 1 -hydroxylase activity by triggering the release of parathyroid hormone (PTH).

Regulation of vit D 3 - ↑↑ 1, 25 -di. OH-D 3, the product of the reaction, decreases 1 -hydroxylase activity.

Regulation of vit D Growth hormone, estrogen, and prolactin also stimulate 1 hydroxylase. • This increases the rate of vitamin D activation in growing children and in pregnant and lactating women. • 4 -

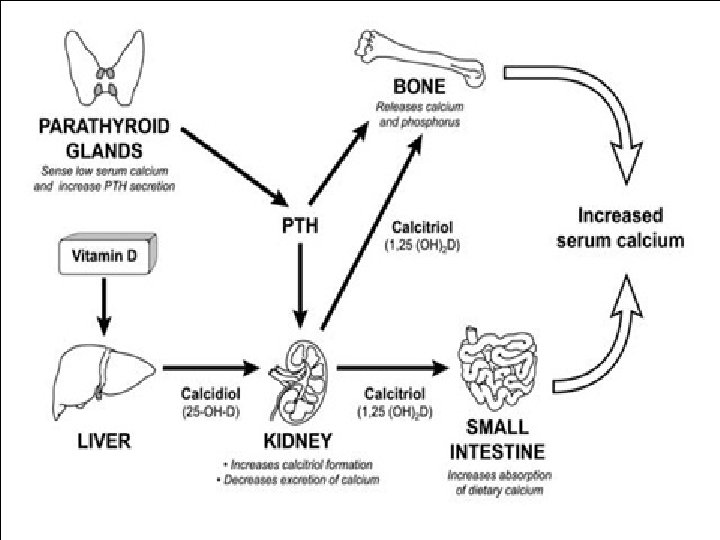
IMPORTANT • Vitamin D can be regarded as a prohormone which is activated to produce calcitriol hormone that acts at nuclear level by a mechanism typical of steroid hormones. • The most prominent action of 1, 25 -di. OHD 3 is to regulate the plasma levels of calcium and phosphorus. •

Regulation of blood calcium level • Calcium level in blood is regulated by the concerted actions of three hormones: • Calcitriol (active vitamin D), Parathyroid hormone, and calcitonin.
I. Active Vitamin D (calcitriol):

(1) Effect of active vitamin D on intestine: • 1, 25 -di-(OH) D (Calcitriol) stimulates Ca 2+ and phosphorus absorption across mucosal cells from lumen to plasma against concentration gradient. It enters the intestinal cell and binds to a cytosolic receptor.
(1) Effect of active vitamin D on intestine: • The 1, 25 -di. OH-D 3–receptor complex then moves to the nucleus where it selectively interacts with the cellular DNA. As a result, there is increased synthesis of a specific calciumbinding protein.
(2) Effect of active vitamin D on kidney: • 1, 25 -(OH)2 -D (calcitiol) stimulates renal reabsorption of phosphate and Ca 2+ in distal tubules.
(3) Effect of active vitamin D on bone mineral turnover: • When serum Ca is lowered (in hypocalcemia), parathormone (PTH) is secreted by the parathyroid gland activates 1 -hydroxylase in the kidney which converts 25 -OH-vitamin D into active vitamin D (calcitriol) which acts synergistically with parathormone to promote bone resorption (demineralization. )

Calcitriol Restores normal serum calcium levels in three different ways: 1) Increasing synthesis of calcium binding protein, thus increasing the absorption of dietary calcium. 2) Increasing the mobilization of calcium from bone into the circulation. 3) Increasing the reabsorption of calcium by the kidneys.
II. Parathyroid hormone (PTH):

II. Parathyroid hormone (PTH): • PTH is produced by 4 glands embeded on the posterior aspect of the thyroid gland. • It is secreted as a prepro-hormone that contains 115 amino acids. Then 25 amino acids are removed to form the pro. PTH followed by removal of an extra 6 amino acids to produce the final PTH form of the hormone.

II. Parathyroid hormone (PTH): • Secretion of the PTH is controlled mainly by the calcium level in the blood. • When calcium level falls, PTH is released and vice versa.
PTH increases calcium level by: • Effect on bone: PTH stimulates the removal of calcium from the bone to increase levels in the blood.

PTH increases calcium level by: • Effect on the kidney: PTH also increases the kidney’s reabsorption of calcium so that less is lost in urine. It also activates vitamin D by stimulating 1α-hydroxylase.
PTH increases calcium level by: • Effect on the intestine: PTH increases calcium absorption from the intestine indirectly, indirectly by stimulating the production of 1, 25 dihydroxycholecalciferol in the kidney.
III. Calcitonin (calcium-lowering hormone)

III. Calcitonin (calcium-lowering hormone) • The thyroid gland secretes calcitonin, in response to high calcium levels in the blood.

III. Calcitonin (calcium-lowering hormone) • It stimulates calcium deposition in the bones. • It also stimulates excretion of calcium and phosphorus by the kidney.

DEFICIENCY OF VITAMIN D: may occur due to: • 1. Decrease exposure to UV rays (in lowincome groups and elderely) • 2. Fat malabsorption (e. g. biliary obstruction, fatty diarrhea) • 3. Severe kidney disease causes vitamin Dresistant rickets due to impaired hydroxylation. • 4. Some drugs e. g. anticonvulsants or alcohol which induce vitamin D catabolism.

MANIFESTATIONS: • (1) Rickets: • Appears in children 6 -24 months of age: • improper mineralization skeletal deformities e. g. bow legs. • Delayed teeth eruption, delayed closure of fontanelles. • Enlarged joints, muscle weakness while alkaline phosphatase is high.
MANIFESTATIONS: • (2) Renal rickets (renal osteodystrophy): • Results from chronic renal failure and which impair 1 -α-hydroxylation activity. 1, 25 -di. OH cholecalciferol (calcitriol) administration is effective replacement therapy.
MANIFESTATIONS: • (3) Osteomalacia: occurs in adults especially pregnant and lactating female. There is demineralization of preexisting bones muscle weakness, bone pain and increased susceptibility to bone fractures.

(4) Hypoparathyroidism: Lack of parathyroid hormone causes hypocalcemia and hyperphosphatemia. These patients may be treated with any form of vitamin D, together with parathyroid hormone.

TOXICITY (hypervitaminosis D): • Vitamin D is the most toxic of all vitamins • High doses (more than 100, 000 IU for weeks or months) cause the following toxic symptoms:

MANIFESTATIONS: • a) Early symptoms: Loss of appetite, nausea, thirst, polyuria. • b) Microcephaly: premature closure of sutures and fontanelles • c) Hypercalcemia: due to increase intestinal Ca absorption and increased bone resorption Ca 2+ deposition in many organs (metastatic calcification) particularly, kidneys and arteries. Hypercalciuria occurs predisposing to renal stones (nephrocalcinosis) which may proceed to renal failure.

Vitamin K (Anti-hemorrhagic Vitamin)

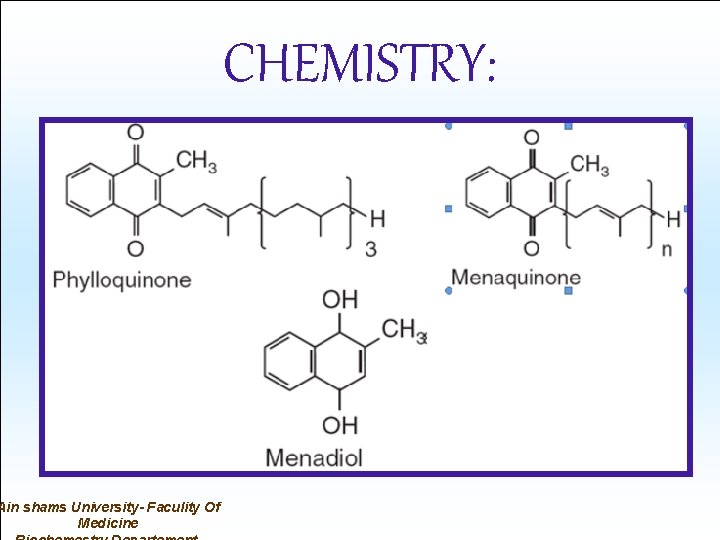
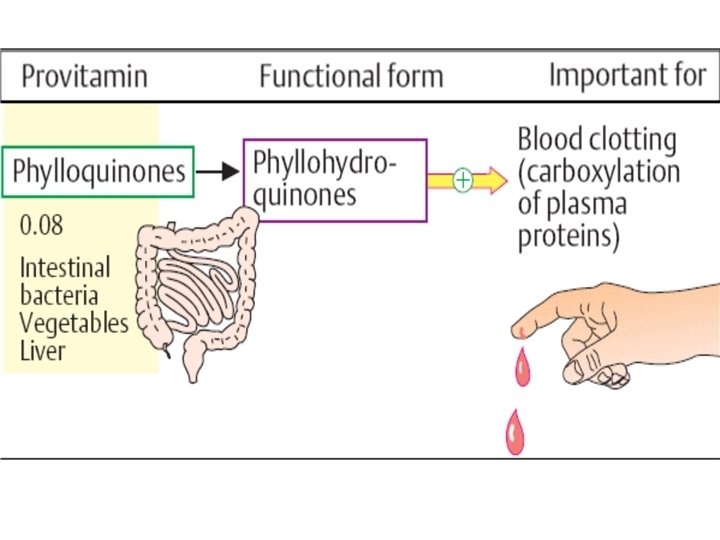
CHEMISTRY: • A group of polyisoprenoid-substituted naphthoquinones: • (1) K 1 = Phylloquinone (major form in plants). • (2) K 2 = Menaquinone (found in animal tissues) synthesized by intestinal bacteria. • (3) K 3 = Menadiol (synthetic) (water soluble).
Ain shams University- Faculity Of Medicine CHEMISTRY:

SOURCES: • (1) Vitamin K 1 (plant sources): is present in green leafy vegetable e. g. spinach, cabbage, cauliflower • (2) Vitamin K 2: egg yolk and liver (limited storage in liver) • It is synthesized by bacterial flora in large intestine.

DAILY REQUIREMENTS: As the vitamin is extensively synthesized by bacteria in the gut and is widely distributed in plant and animal foods, there is no RDA for vitamin K under physiological conditions. (70 -140 mg/day)

ABSORPTION: • It is absorbed in chylomicrons as other fat-soluble vitamins. • It is transferred to plasma in lipoproteins (VLDL, LDL).

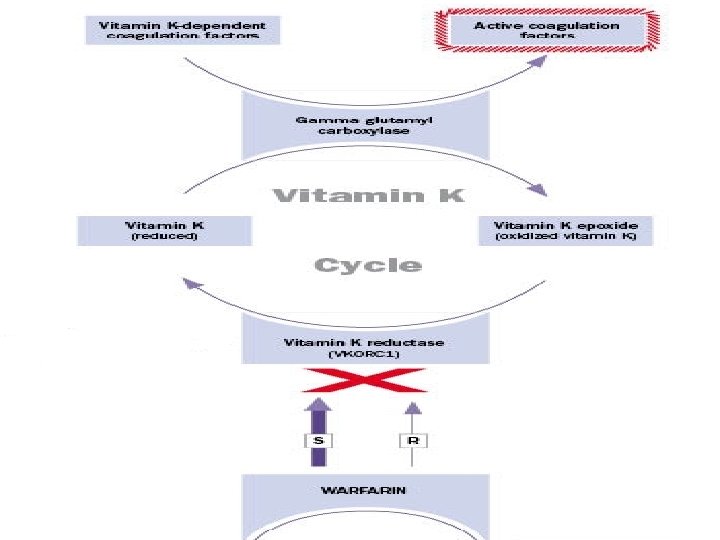
FUNCTIONS: • (1) The principal role of vitamin K is in the co-translational modification of the blood clotting factors II, VII, IX, and X and the anticoagulant proteins protein C and protein S.

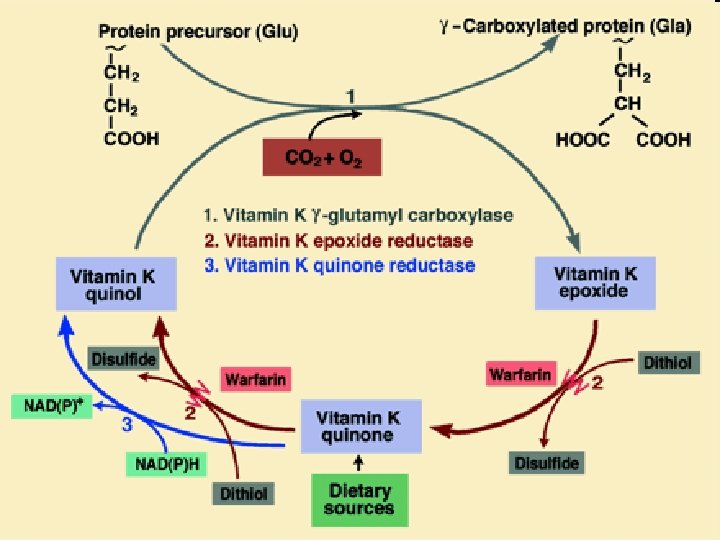
Vitamin K cycle • These vitamin K-dependent proteins are synthesized in the liver. • Vitamin K (the hydroquinone form) serves as a coenzyme for a carboxylase enzyme that adds a -carboxyl group to certain glutamate residues in these vitamin K-dependent proteins. • The reaction requires O 2, CO 2, and the hydroquinone form of vitamin K.
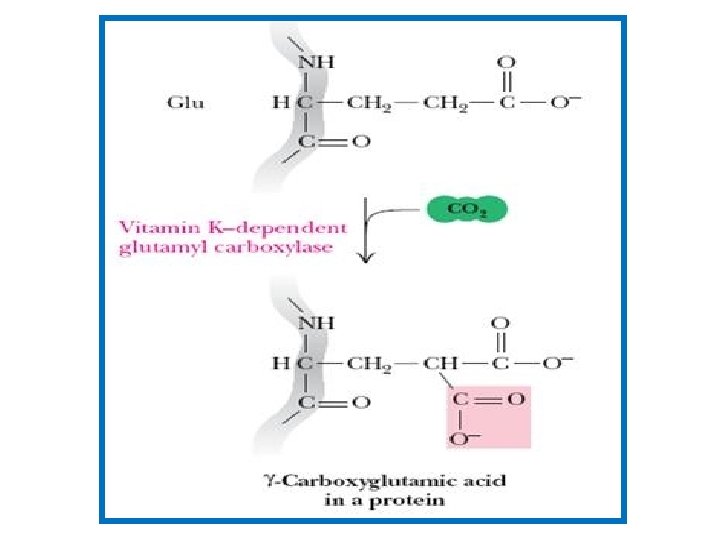
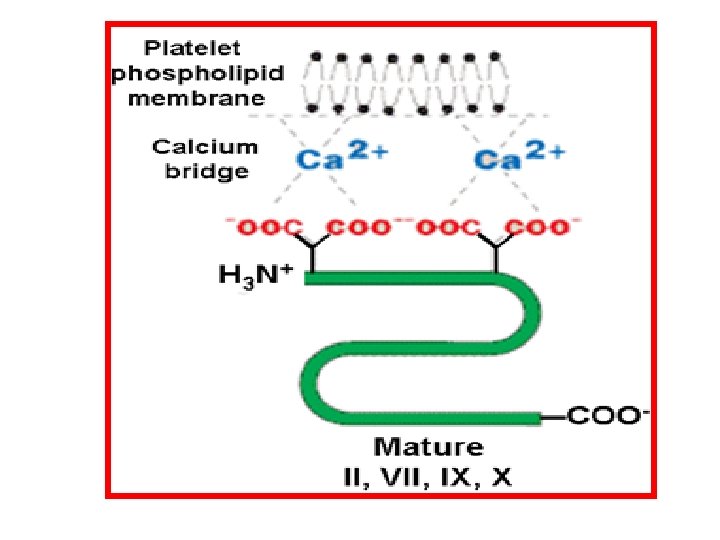

Vitamin K cycle • The Gla residues produced by this reaction, are essential for the assembly of the vitamin Kdependent proteins on the phospholipid membranes of platelets. Ca 2+ bridges the negatively charged Gla residues to the negatively charged membrane phospholipids of platelets (protein-Ca 2+-phospholipid interaction). • This assembly dramatically accelerates the activation of coagulation proteins as prothrombin.

Vitamin K cycle • The formation of -carboxyglutamate residues (Gla residues) is sensitive to inhibition by dicumarol, a natural anticoagulant in spoiled sweet clover, and by warfarin, a synthetic analog of vitamin K. Both act as competitive inhibitors for the two reductases which convert vitamin K epoxide to the hydroquinone active form. • A high dose of vitamin K is the antidote to an overdose of both anticoagulants
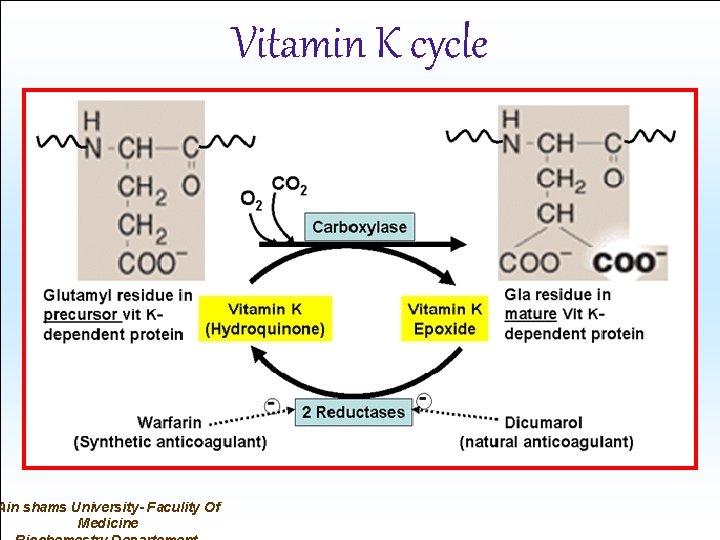
Ain shams University- Faculity Of Medicine Vitamin K cycle

FUNCTIONS: • (2) Recently, vitamin K has been shown to be essential for addition of Gla residues to other proteins in bones (osteocalcin) and in other tissues. • The physiological function of these vitamin K-dependent proteins is still unclear.

DEFICIENCY: • may occur in: • (1) Newborn infants as the gut is sterile and breast milk is very low in vitamin K. A postnatal injection of vitamin K may be given especially to premature babies. • (2) Adults, due to fat malabsorption or extensive use of antibiotics or prolonged dicumarol therapy.

Manifestations: • Haemorrhagic syndrome in newborn (especially in those born to mothers maintained on anticonvulsants that increase catabolism of vitamin K) • Prolonged prothrombin times, easy bruising and hemorrhage (internal and external). Prolonged prothrombin time (PT) without intake of anticoagulants and with normal liver functions should be interpreted as vitamin K deficiency especially in poorly nourished people.

TOXICITY OF VITAMIN K: • Prolonged administration of large doses of vitamin K produces hemolytic anemia and jaundice (hyperbilirubinemia) which may lead to brain damage in infants. • This is due to toxic effects of vitamin K on membrane of RBCs.
- Slides: 96