Lipids phospholipids cholesterol steroids Biochem I lecture 5
Lipids: phospholipids, cholesterol, steroids Biochem I, lecture 5 Chapter 26. 0 -26. 1 (exclude sphingolipids, p. 736739), 26. 2, 26. 4 (exclude pregnenolone sections, p. 752 -754) Sept. 21 / 06
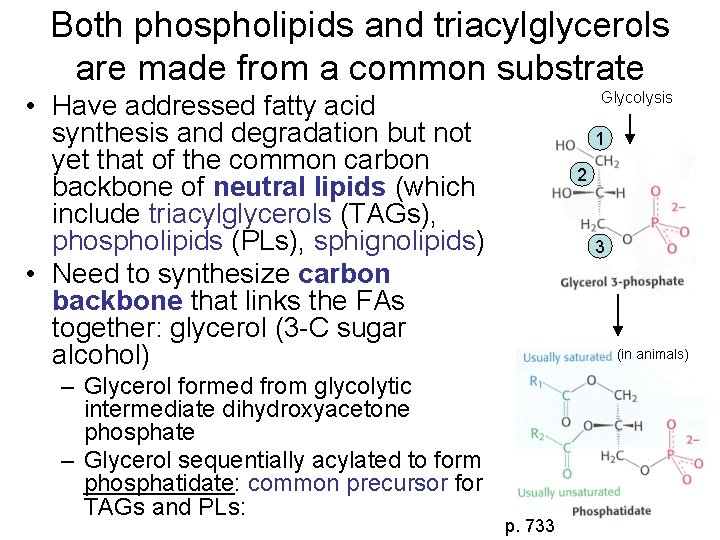
Both phospholipids and triacylglycerols are made from a common substrate Glycolysis • Have addressed fatty acid synthesis and degradation but not yet that of the common carbon backbone of neutral lipids (which include triacylglycerols (TAGs), phospholipids (PLs), sphignolipids) • Need to synthesize carbon backbone that links the FAs together: glycerol (3 -C sugar alcohol) – Glycerol formed from glycolytic intermediate dihydroxyacetone phosphate – Glycerol sequentially acylated to form phosphatidate: common precursor for TAGs and PLs: 1 2 3 (in animals) p. 733
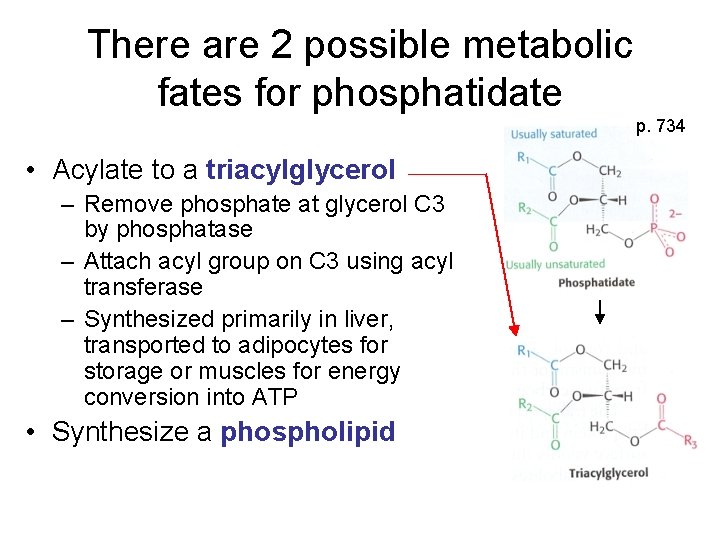
There are 2 possible metabolic fates for phosphatidate p. 734 • Acylate to a triacylglycerol – Remove phosphate at glycerol C 3 by phosphatase – Attach acyl group on C 3 using acyl transferase – Synthesized primarily in liver, transported to adipocytes for storage or muscles for energy conversion into ATP • Synthesize a phospholipid
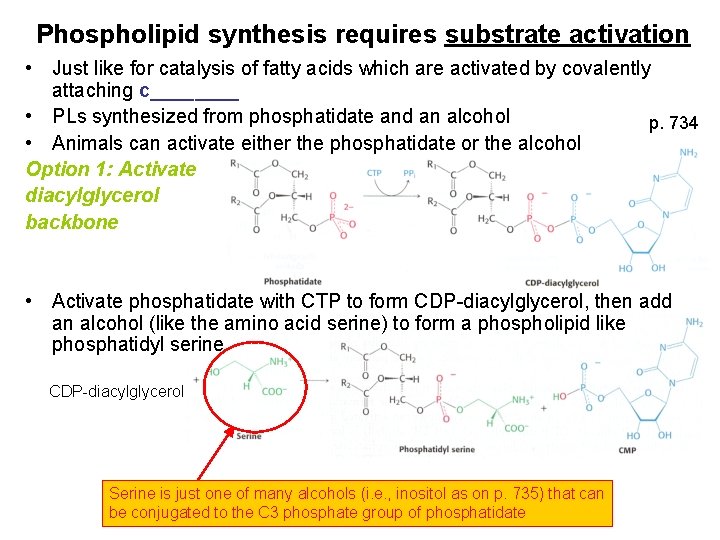
Phospholipid synthesis requires substrate activation • Just like for catalysis of fatty acids which are activated by covalently attaching c____ • PLs synthesized from phosphatidate and an alcohol p. 734 • Animals can activate either the phosphatidate or the alcohol Option 1: Activate diacylglycerol backbone • Activate phosphatidate with CTP to form CDP-diacylglycerol, then add an alcohol (like the amino acid serine) to form a phospholipid like phosphatidyl serine CDP-diacylglycerol Serine is just one of many alcohols (i. e. , inositol as on p. 735) that can be conjugated to the C 3 phosphate group of phosphatidate

Phospholipid synthesis requires substrate activation Option 2: Activate the alcohol – Take aminated ethanol – Phosphorylate it using ATP – Activate it by attaching cytidine diphosphate by CTP usage – Conjugate the activated alcohol to diacylglycerol – Voilà: the phospholipid phosphatidyl ethanolamine • Regardless of how they are formed, phospholipids can vary in structure and properties by varying Activated intermediate – FA chains attached to C 1 and C 2 – The alcohol connected to the phosphate • A single mammalian cell can contain thousands of different PL species, each possessing distinct properties p. 736

Cholesterol synthesis differs from that of neutral lipids • Cholesterol – is a lipid and a steroid – modulates the fluidity of animal cell membranes – is the precursor of steroid hormones: progesterone, testosterone, estradiol, cortisol • Recall that – cholesterol is a lipid but is not linear like FAs – it is synthesized by a distinct pathway from 5 C units (isoprene) versus FAs • 3 series of reactions to make cholesterol from isoprene 1. Synthesize isoprene in an activated form (isopentenyl pyrophosphate) 2. Condense 6 of these to make the branched hydrocarbon squalene 3. Cyclize squalene
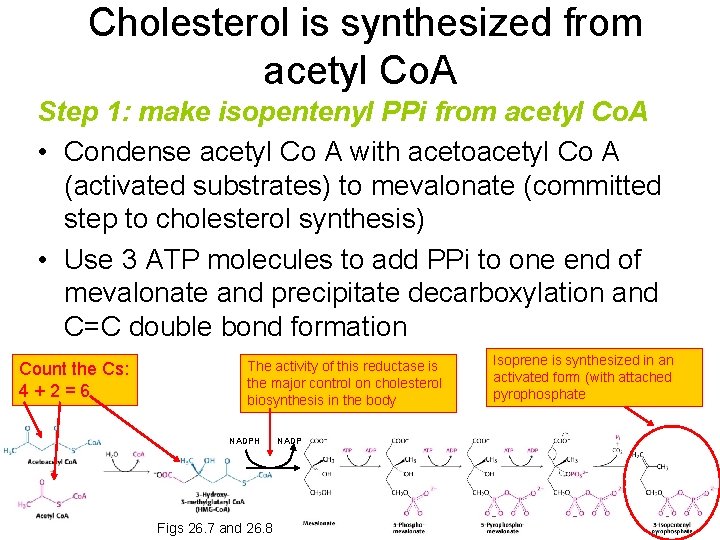
Cholesterol is synthesized from acetyl Co. A Step 1: make isopentenyl PPi from acetyl Co. A • Condense acetyl Co A with acetoacetyl Co A (activated substrates) to mevalonate (committed step to cholesterol synthesis) • Use 3 ATP molecules to add PPi to one end of mevalonate and precipitate decarboxylation and C=C double bond formation Count the Cs: 4+2=6 The activity of this reductase is the major control on cholesterol biosynthesis in the body NADPH Figs 26. 7 and 26. 8 NADP Isoprene is synthesized in an activated form (with attached pyrophosphate
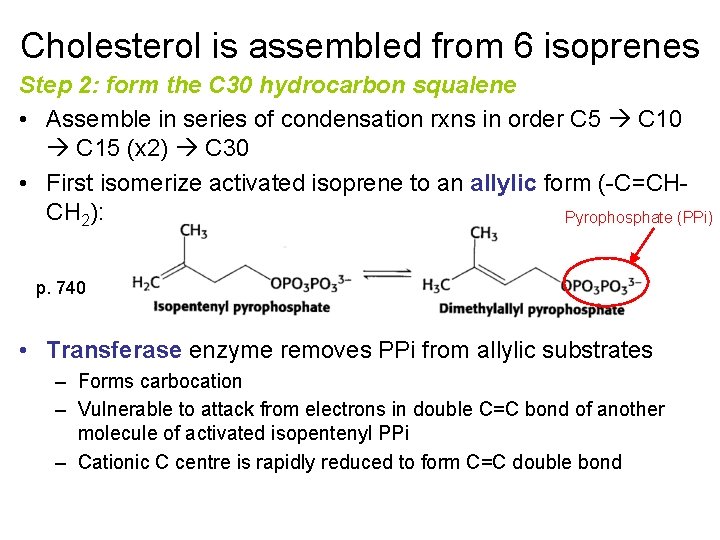
Cholesterol is assembled from 6 isoprenes Step 2: form the C 30 hydrocarbon squalene • Assemble in series of condensation rxns in order C 5 C 10 C 15 (x 2) C 30 • First isomerize activated isoprene to an allylic form (-C=CHCH 2): Pyrophosphate (PPi) p. 740 • Transferase enzyme removes PPi from allylic substrates – Forms carbocation – Vulnerable to attack from electrons in double C=C bond of another molecule of activated isopentenyl PPi – Cationic C centre is rapidly reduced to form C=C double bond
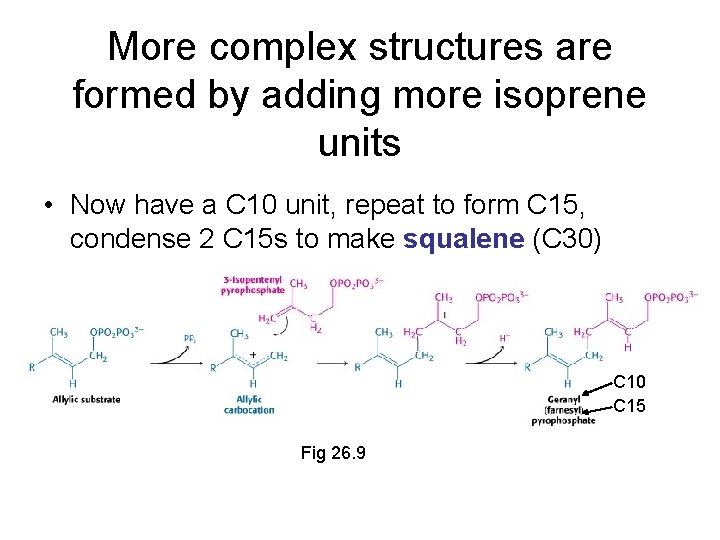
More complex structures are formed by adding more isoprene units • Now have a C 10 unit, repeat to form C 15, condense 2 C 15 s to make squalene (C 30) C 10 C 15 Fig 26. 9
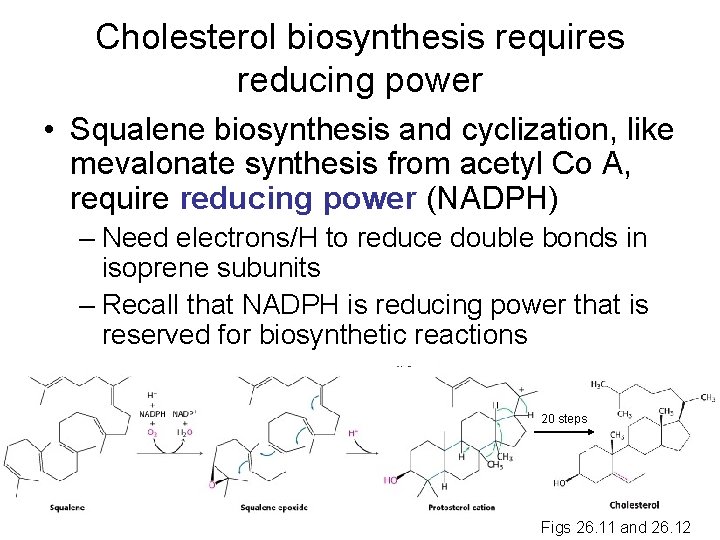
Cholesterol biosynthesis requires reducing power • Squalene biosynthesis and cyclization, like mevalonate synthesis from acetyl Co A, require reducing power (NADPH) – Need electrons/H to reduce double bonds in isoprene subunits – Recall that NADPH is reducing power that is reserved for biosynthetic reactions 20 steps Figs 26. 11 and 26. 12
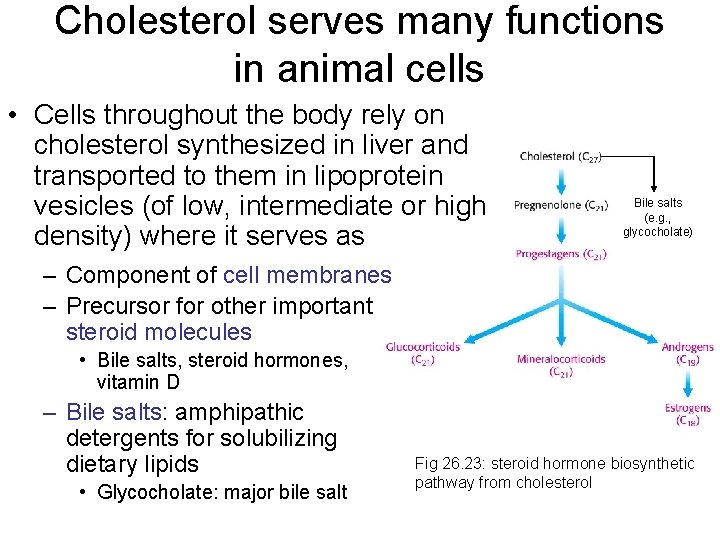
Cholesterol serves many functions in animal cells • Cells throughout the body rely on cholesterol synthesized in liver and transported to them in lipoprotein vesicles (of low, intermediate or high density) where it serves as Bile salts (e. g. , glycocholate) – Component of cell membranes – Precursor for other important steroid molecules • Bile salts, steroid hormones, vitamin D – Bile salts: amphipathic detergents for solubilizing dietary lipids • Glycocholate: major bile salt Fig 26. 23: steroid hormone biosynthetic pathway from cholesterol

Cholesterol is the precursor of all steroid hormones • Remember that hormones initiate signal transduction cascades and thus change metabolism in target cells (e. g. , to start breaking down stored triacylglycerols to free FAs in adipocytes and export them to muscle cells) • Many different slight variations in hormone structure (though they are all similar: derived from cholesterol!) enable binding to different cell-surface receptors and thus activation of distinct cell targets
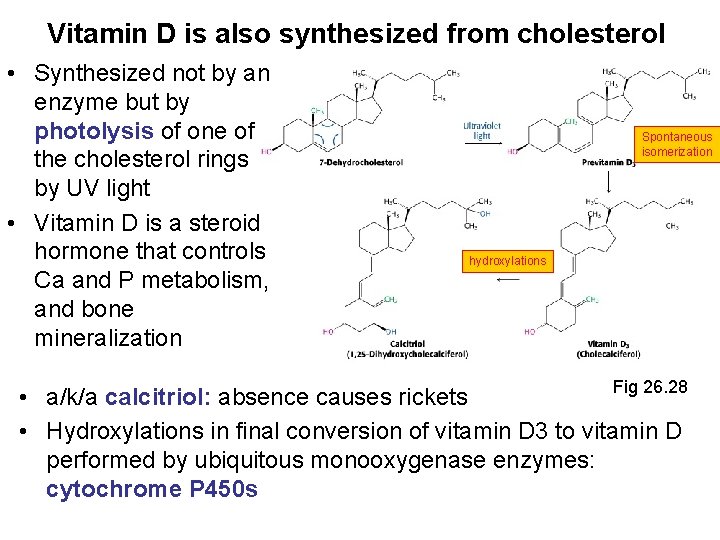
Vitamin D is also synthesized from cholesterol • Synthesized not by an enzyme but by photolysis of one of the cholesterol rings by UV light • Vitamin D is a steroid hormone that controls Ca and P metabolism, and bone mineralization Spontaneous isomerization hydroxylations Fig 26. 28 • a/k/a calcitriol: absence causes rickets • Hydroxylations in final conversion of vitamin D 3 to vitamin D performed by ubiquitous monooxygenase enzymes: cytochrome P 450 s

Cytochrome P 450 enzymes are oxygenases • Very interesting class of enzymes • Bind molecular oxygen (O 2) with the help of a reductant, NADPH • One O molecule used to hydroxylate (add OH) substrate (“R”) and the other to produce H 2 O • Generic reaction: • Contain a heme group, just like hemoglobin, and can thus bind O 2 • NADPH contributes 1 e- to reduce the iron centre of hemoglobin from Fe 3+ to Fe 2+ • Acceptance of second e- cleaves bonds between O atoms in O 2 and releases H 2 O • Cytochrome P 450 s are important in detoxifying foreign (xenobiotic) substances in body • Drugs are inactivated by these enzymes! Can also generate very carcinogenic compounds by metabolic activation (e. g. , plant toxins, pigments)
- Slides: 14