Lipids Pages 16 18 Specification area 3 1














- Slides: 29

Lipids Pages 16 -18 Specification area 3. 1. 3

Objectives 1. Describe the roles of lipids 2. Describe the structure of triglycerides and how this relates to their function 3. Describe the structure of phospholipids and how this relates to their function 4. Describe the test for a lipid

Objective 1 Describe the roles of lipids • • List the 4 characteristics of lipids What are the 2 main groups of lipids? What is the role of a phospholipid? What other roles do lipids have? (4)

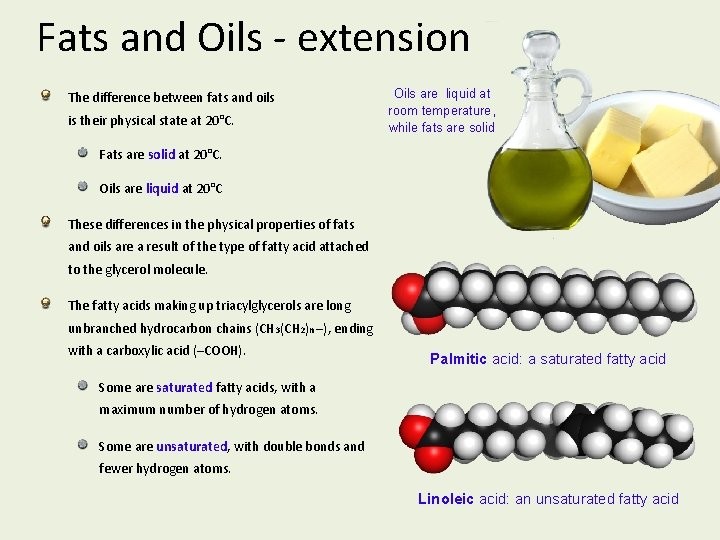
Fats and Oils - extension The difference between fats and oils is their physical state at 20°C. Oils are liquid at room temperature, while fats are solid Fats are solid at 20°C. Oils are liquid at 20°C These differences in the physical properties of fats and oils are a result of the type of fatty acid attached to the glycerol molecule. The fatty acids making up triacylglycerols are long unbranched hydrocarbon chains (CH 3(CH 2)n –), ending with a carboxylic acid (–COOH). Palmitic acid: a saturated fatty acid Some are saturated fatty acids, with a maximum number of hydrogen atoms. Some are unsaturated, with double bonds and fewer hydrogen atoms. Linoleic acid: an unsaturated fatty acid

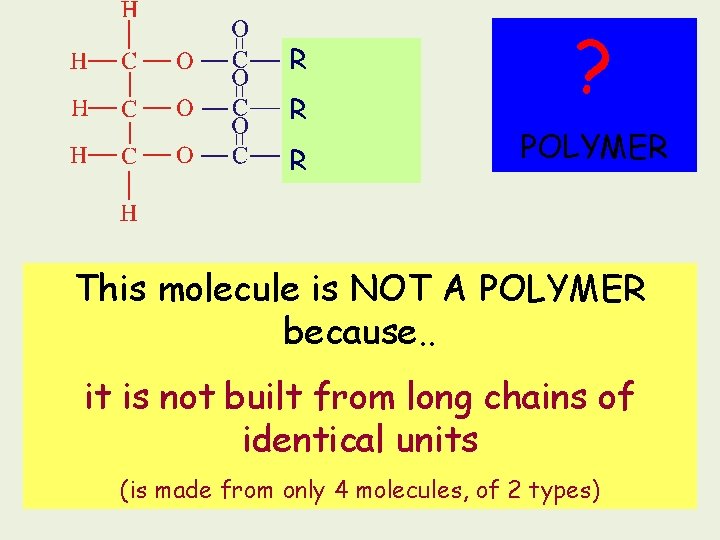
1. 2. 3. 4. 5. 6. 7. Objective 2 Describe the structure of triglycerides and how this relates to their function Why is it called a triglyceride? What are the 4 constituent molecules in a triglyceride? What reactions join these constituents together? And which reaction splits them up? What is the name of the bond formed each time? How many of these bonds can be found in a triglyceride? Draw the formation of a triglyceride and add the labels glycerol, condensation reaction, fatty acid, ester bond Why is a triglyceride NOT a polymer?

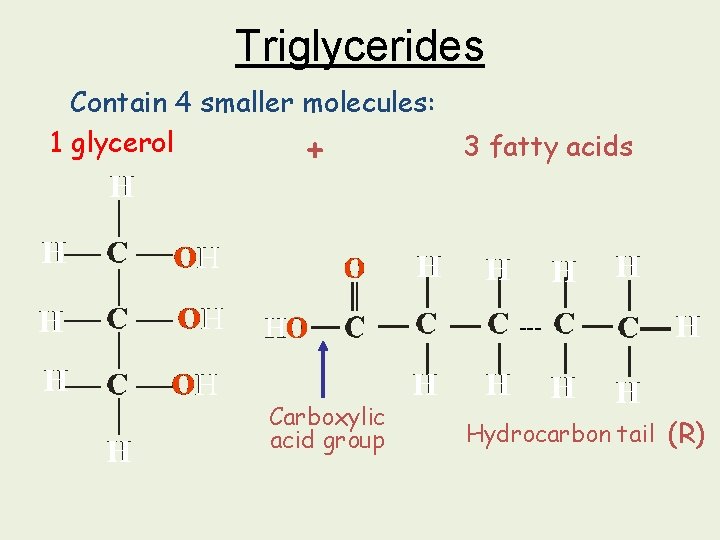
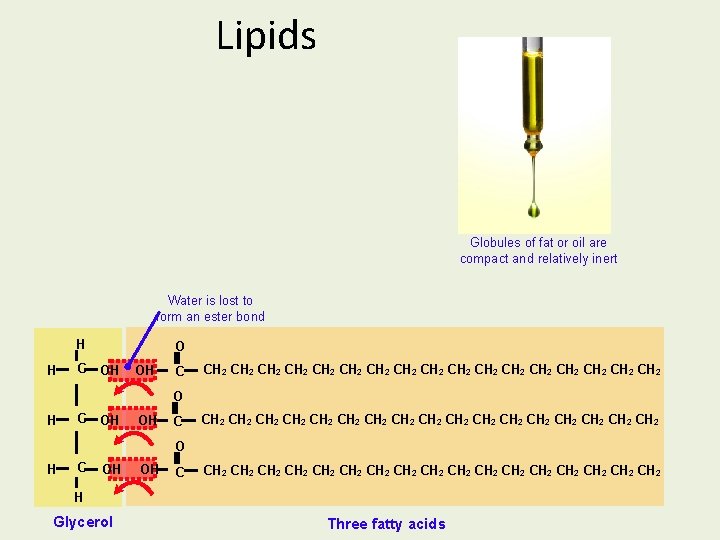
Triglycerides Contain 4 smaller molecules: 1 glycerol 3 fatty acids + --- Carboxylic acid group Hydrocarbon tail (R)

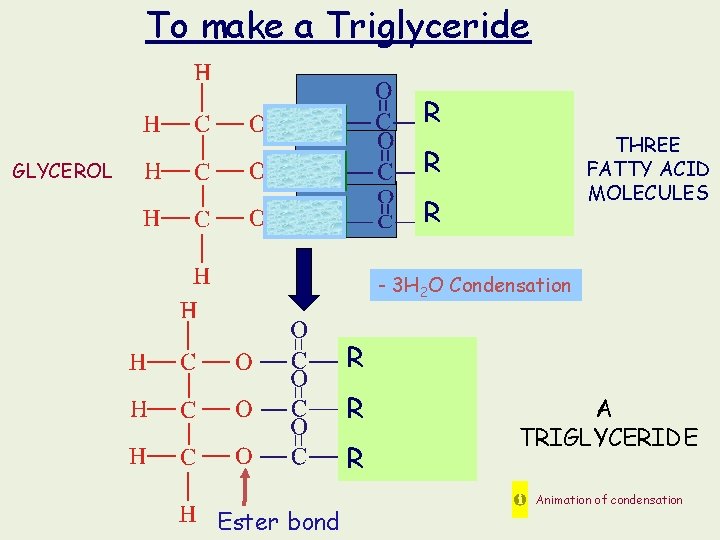
To make a Triglyceride R THREE FATTY ACID MOLECULES R GLYCEROL R - 3 H 2 O Condensation R R R Ester bond A TRIGLYCERIDE Animation of condensation

R R R ? POLYMER This molecule is NOT A POLYMER because. . it is not built from long chains of identical units (is made from only 4 molecules, of 2 types)

Lipids Globules of fat or oil are compact and relatively inert Water is lost to form an ester bond H H C O OH OH C CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 O H C OH OH C CH 2 CH 2 CH 2 CH 2 CH 2 H Glycerol Three fatty acids

Objective 2 Describe the structure of triglycerides and how this relates to their function • How many different fatty acids are there? • What is common in all of their structures? • What is a saturated fatty acid? Draw one • What is an unsaturated fatty acid? Draw one • What is mono unsaturated? Polyunsaturated?

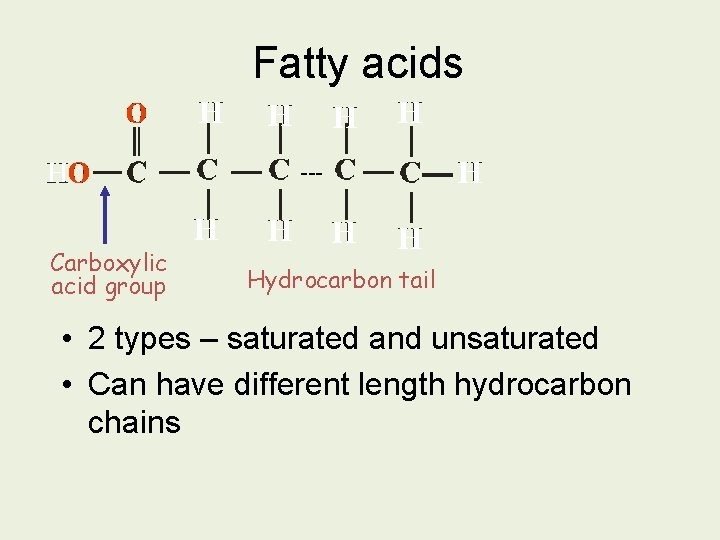
Fatty acids --- Carboxylic acid group Hydrocarbon tail • 2 types – saturated and unsaturated • Can have different length hydrocarbon chains

Saturated & Unsaturated Fatty Acids 1. Saturated fatty acids No double bonds in hydrocarbon tail, so maximum number of hydrogens ie. ’saturated’ Molecules can lie close to each other, so lipids containing mainly saturated fatty acids are SOLID at room temp. Typically ANIMAL fat

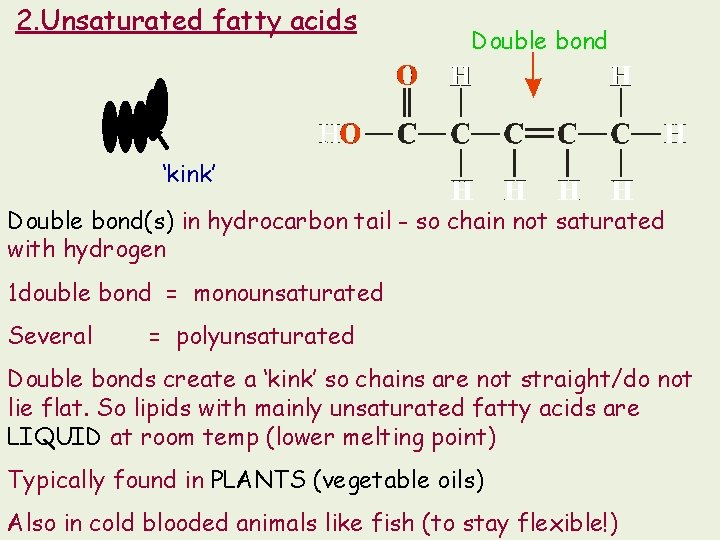
2. Unsaturated fatty acids Double bond ‘kink’ Double bond(s) in hydrocarbon tail - so chain not saturated with hydrogen 1 double bond = monounsaturated Several = polyunsaturated Double bonds create a ‘kink’ so chains are not straight/do not lie flat. So lipids with mainly unsaturated fatty acids are LIQUID at room temp (lower melting point) Typically found in PLANTS (vegetable oils) Also in cold blooded animals like fish (to stay flexible!)

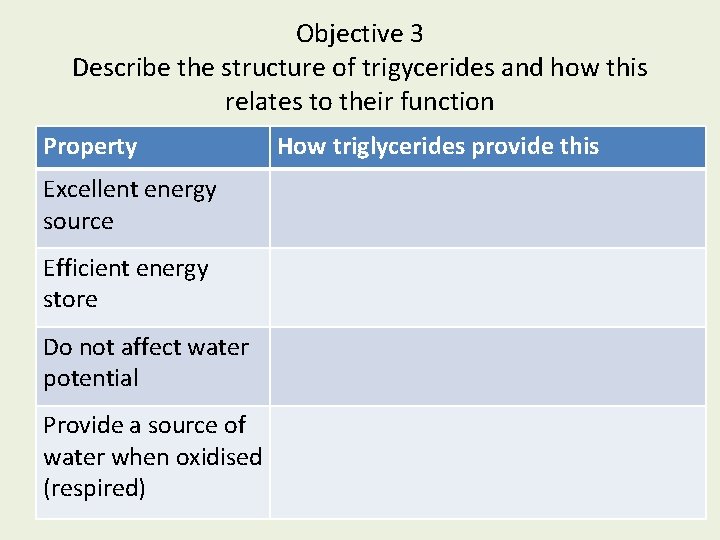
Objective 3 Describe the structure of trigycerides and how this relates to their function Property Excellent energy source Efficient energy store Do not affect water potential Provide a source of water when oxidised (respired) How triglycerides provide this

Objective 3 Describe the structure of phospholipids and how this relates to their function 1. Describe the difference between a triglyceride and a phospholipid 2. Which par of a phospholipid is hydrophilic? 3. Which part of a phospholipid is hydrophobic? 4. Phospholipids are polar molecules. What does polar mean?

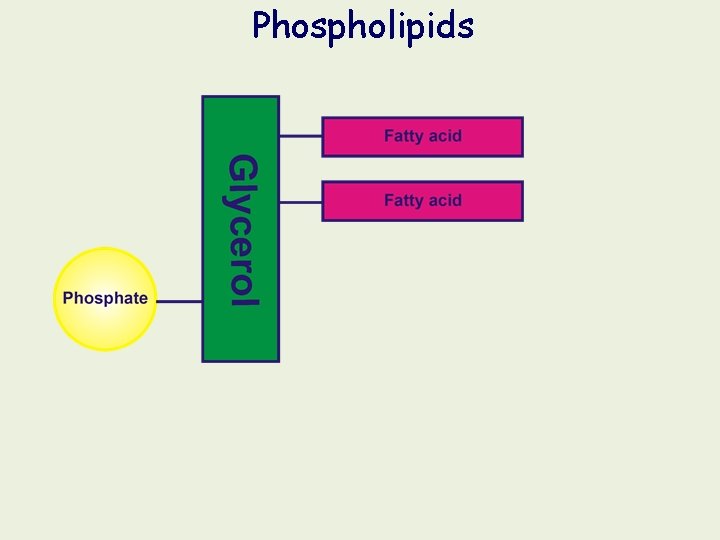
Phospholipids If one of the fatty acid groups of a triacylglyerol is replaced by a phosphate group, the molecule is known as a phospholipid. A phospholipid consists of: a glycerol molecule two fatty acid chains a phosphate (PO 43 -) group (ionised under the conditions in cells) H 2 C HC Nonpolar, hydrocarbon tails of two fatty acids condensed with glycerol COO O– H 2 C O P O– Fatty acid Phosphate group from phosphoric acid (HPO 4) condenses with the third -OH of glycerol Glycerol O PO 43 - Fatty acid Symbolic representation of a phospholipid

Phospholipids

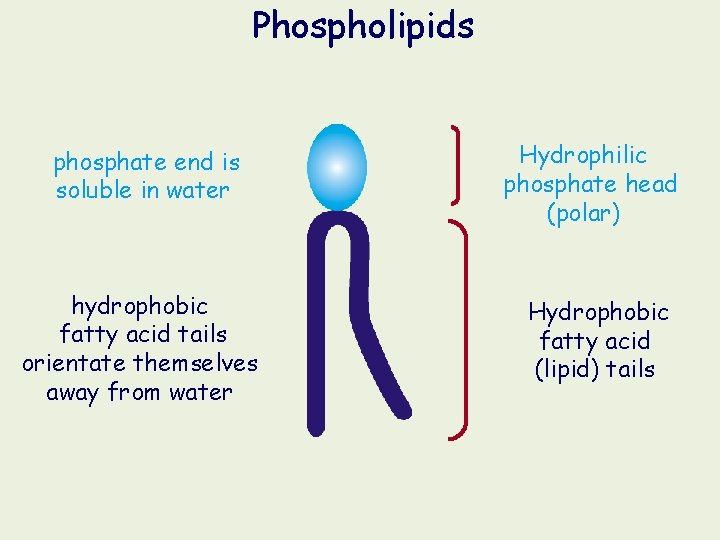
Phospholipids phosphate end is soluble in water hydrophobic fatty acid tails orientate themselves away from water Hydrophilic phosphate head (polar) Hydrophobic fatty acid (lipid) tails

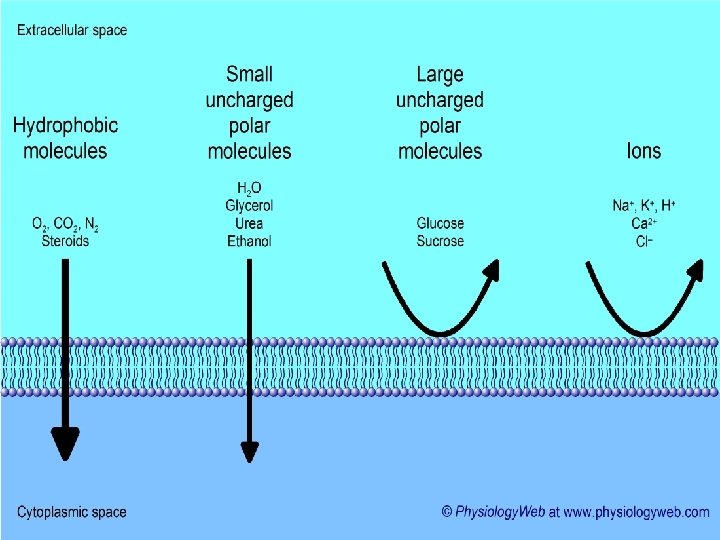
Phospholipids The phosphate end of the molecule is polar and attracted to water (hydrophilic) while the fatty acid end is non-polar and is repelled (hydrophobic). As a result, phospholipids naturally form a bilayer with the hydrophobic ends orientated inwards. The phospholipid bilayer forms the main component of cellular membranes. Glycerol and phosphate ‘head’: the hydrophilic part of the molecule Hydrocarbon tail: hydrophobic part of the molecule.

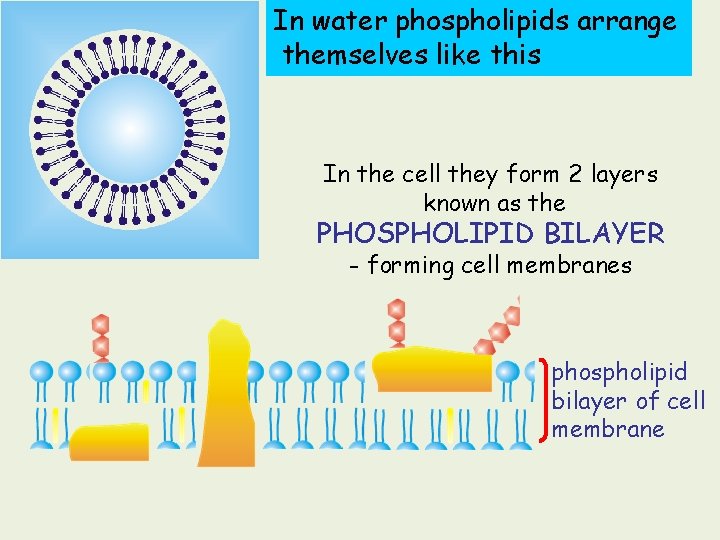
In water phospholipids arrange themselves like this In the cell they form 2 layers known as the PHOSPHOLIPID BILAYER - forming cell membranes phospholipid bilayer of cell membrane


Objective 3 Describe of phospholipids and how Property the structure How phospholipids provide this relates to their function Form a bilayer which is the cell surface membrane. Provides a partially permeable barrier at the cell surface membrane Are important in cell recognition

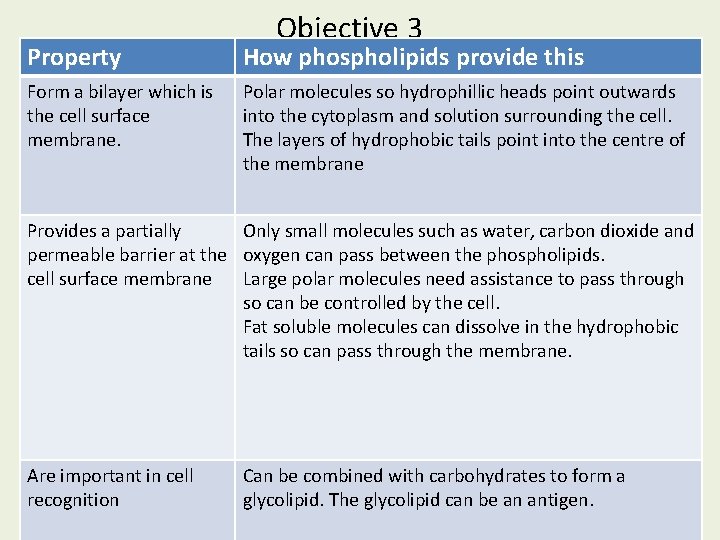
Objective 3 Property How phospholipids provide this Describe the structure of phospholipids and how Form a bilayer which is Polar molecules so hydrophillic heads point outwards to their the cell surface this relates into the cytoplasm andfunction solution surrounding the cell. membrane. The layers of hydrophobic tails point into the centre of the membrane Provides a partially Only small molecules such as water, carbon dioxide and permeable barrier at the oxygen can pass between the phospholipids. cell surface membrane Large polar molecules need assistance to pass through so can be controlled by the cell. Fat soluble molecules can dissolve in the hydrophobic tails so can pass through the membrane. Are important in cell recognition Can be combined with carbohydrates to form a glycolipid. The glycolipid can be an antigen.

Objective 4 Describe the test for a lipid • Summarise the method in 3 essential points

Homework • Summary questions p 18 MAHAC

Assessment • Self

Extension activity • Oils are usually liquid at room temperature. Do you think the fatty acids making up oil triglycerides are saturated or unsaturated? Why? • Ans purple box p 16 • Are triglycerides (and phospholipids) polymers? Explain your answer. • Why is the degree of saturation of the fatty acid chains important?

Keywords • Triglyceride, phospholipid, saturated, unsaturated, condensation, hydrolysis, polar, hydrophillic, hydrophobic, bilayer,

• Past exam paper material: BIOL 1 Jan 2011 – Q 4. • • Exampro – BYB 1 June 04 Q 2 • Phospholipids • Past exam paper material: BIOL 1 Jan 2012 – Q 1 b.