LIPIDS Lipids classification LIPIDS Insoluble in water Soluble

LIPIDS
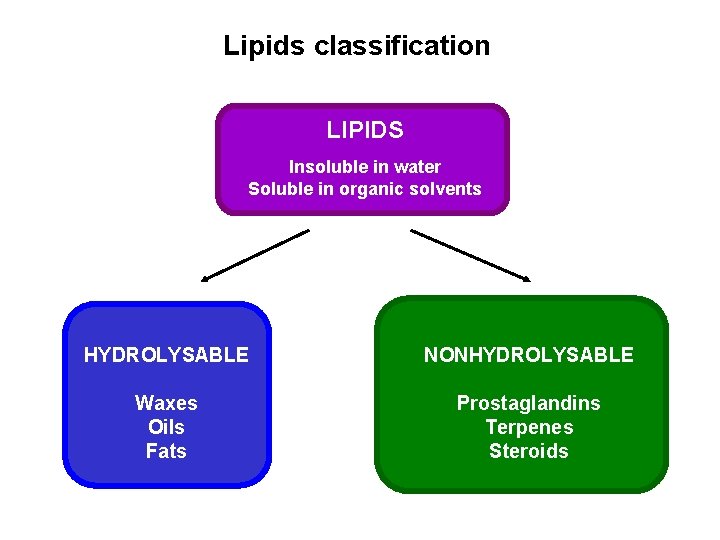
Lipids classification LIPIDS Insoluble in water Soluble in organic solvents HYDROLYSABLE NONHYDROLYSABLE Waxes Oils Fats Prostaglandins Terpenes Steroids
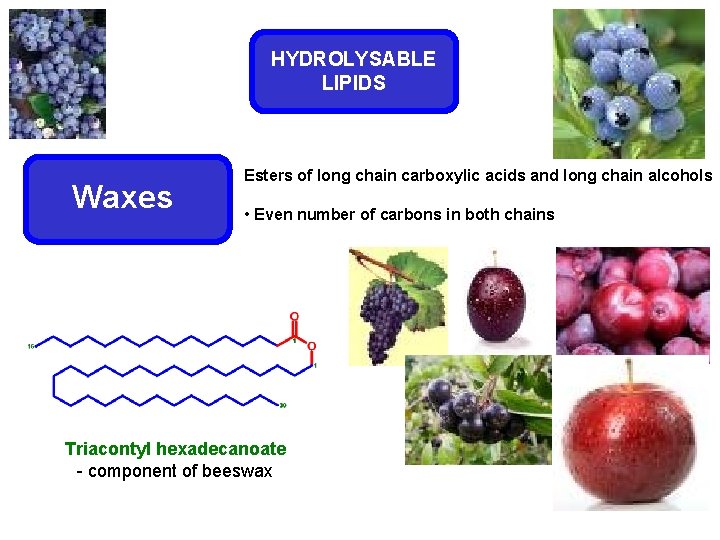
HYDROLYSABLE LIPIDS Waxes Esters of long chain carboxylic acids and long chain alcohols • Even number of carbons in both chains Triacontyl hexadecanoate - component of beeswax
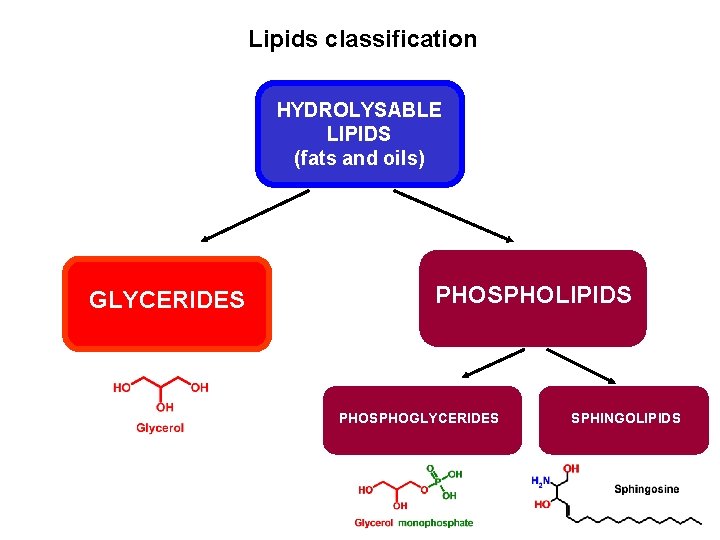
Lipids classification HYDROLYSABLE LIPIDS (fats and oils) GLYCERIDES PHOSPHOLIPIDS PHOSPHOGLYCERIDES SPHINGOLIPIDS
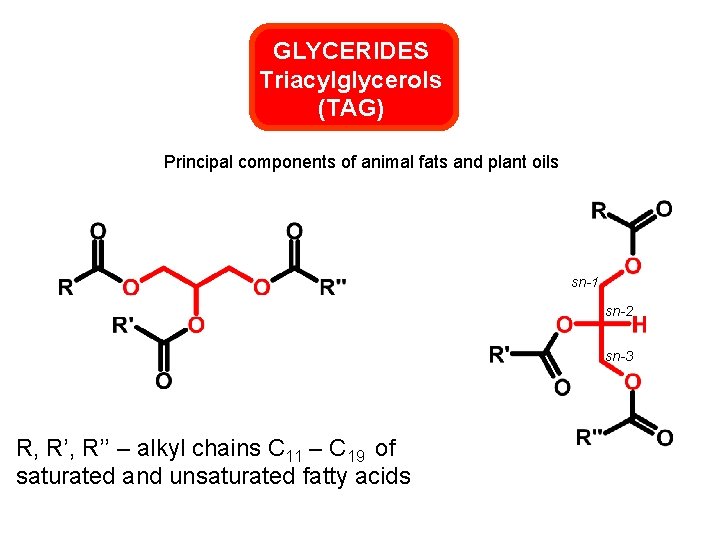
GLYCERIDES Triacylglycerols (TAG) Principal components of animal fats and plant oils sn-1 sn-2 sn-3 R, R’’ – alkyl chains C 11 – C 19 of saturated and unsaturated fatty acids
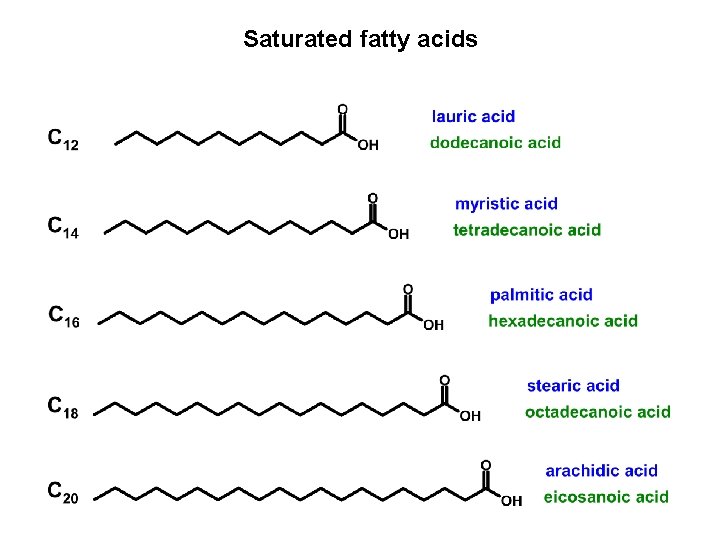
Saturated fatty acids
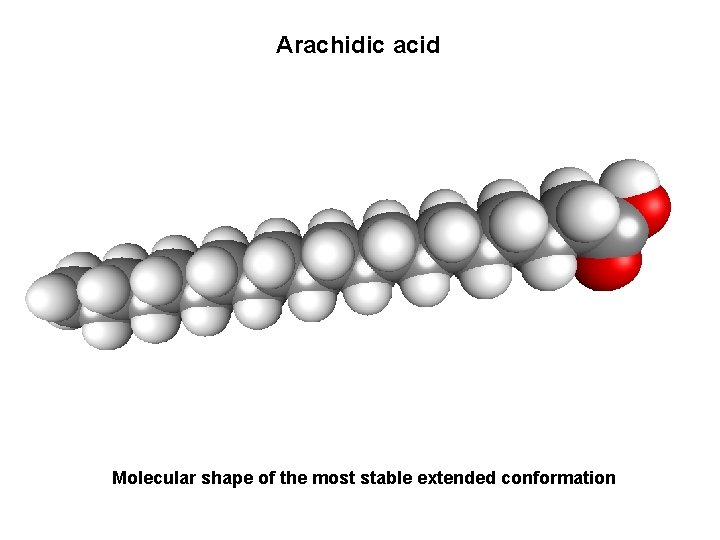
Arachidic acid Molecular shape of the most stable extended conformation
Fatty acids biosynthesis - Priming reactions

Fatty acids biosynthesis – Chain elongation cycle

Unsaturated fatty acids C 18
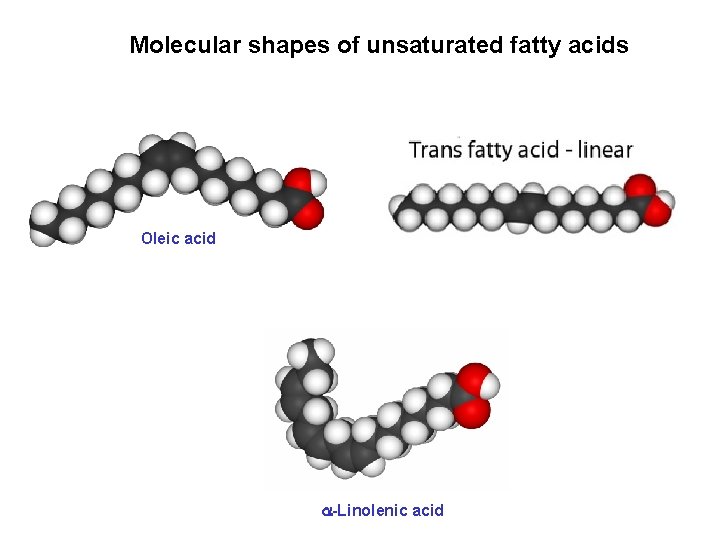
Molecular shapes of unsaturated fatty acids Oleic acid -Linolenic acid

Unsaturated fatty acid C 20
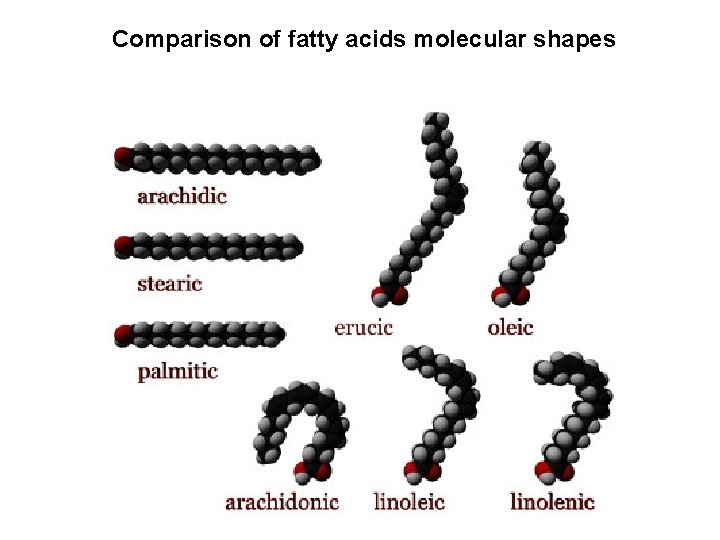
Comparison of fatty acids molecular shapes
![Melting points of fatty acids Saturated acid C 12 Lauric M. P. [ C] Melting points of fatty acids Saturated acid C 12 Lauric M. P. [ C]](http://slidetodoc.com/presentation_image/d5eda1fcbd7ed7344a5197a01b8fed46/image-14.jpg)
Melting points of fatty acids Saturated acid C 12 Lauric M. P. [ C] 44 Unsaturated acid M. P. [ C] C 14 Myristic 58 C 18 (18: 1) Oleic 4 C 16 Palmitic 63 C 18 (18: 2) Linoleic -5 C 18 Stearic 70 C 18 (18: 3) Linolenic -11 C 20 Arachidic 75 C 20 (20: 4) Arachidonic -50
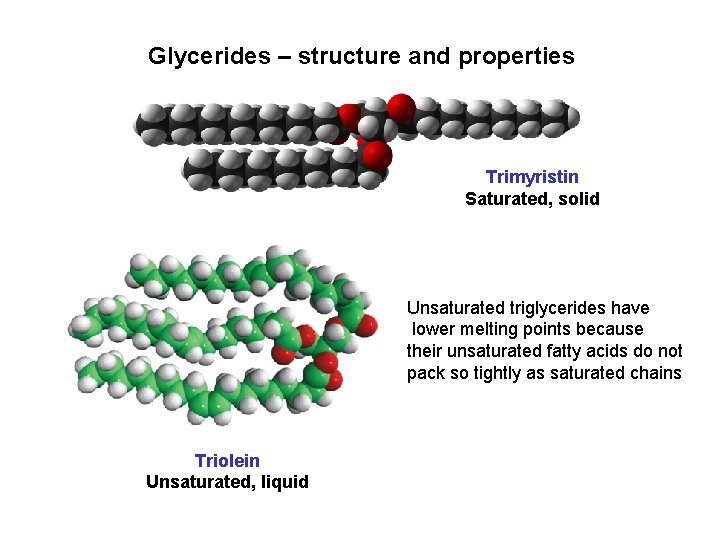
Glycerides – structure and properties Trimyristin Saturated, solid Unsaturated triglycerides have lower melting points because their unsaturated fatty acids do not pack so tightly as saturated chains Triolein Unsaturated, liquid
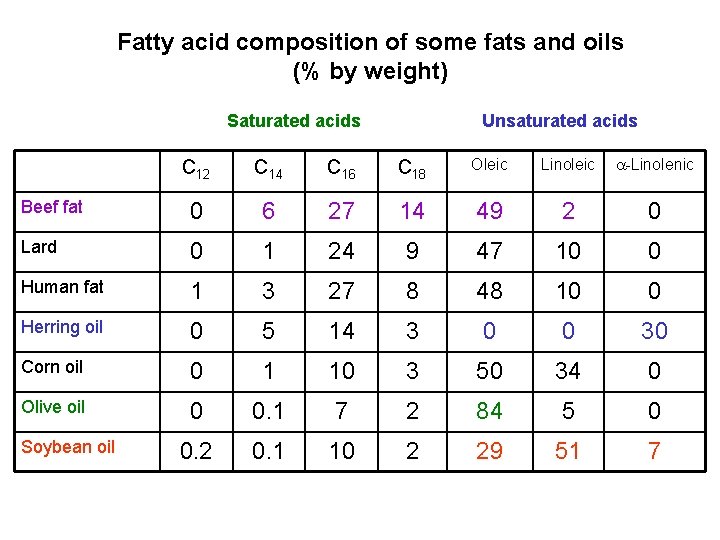
Fatty acid composition of some fats and oils (% by weight) Saturated acids Unsaturated acids C 12 C 14 C 16 C 18 Oleic Linoleic -Linolenic Beef fat 0 6 27 14 49 2 0 Lard 0 1 24 9 47 10 0 Human fat 1 3 27 8 48 10 0 Herring oil 0 5 14 3 0 0 30 Corn oil 0 1 10 3 50 34 0 Olive oil 0 0. 1 7 2 84 5 0 0. 2 0. 1 10 2 29 51 7 Soybean oil
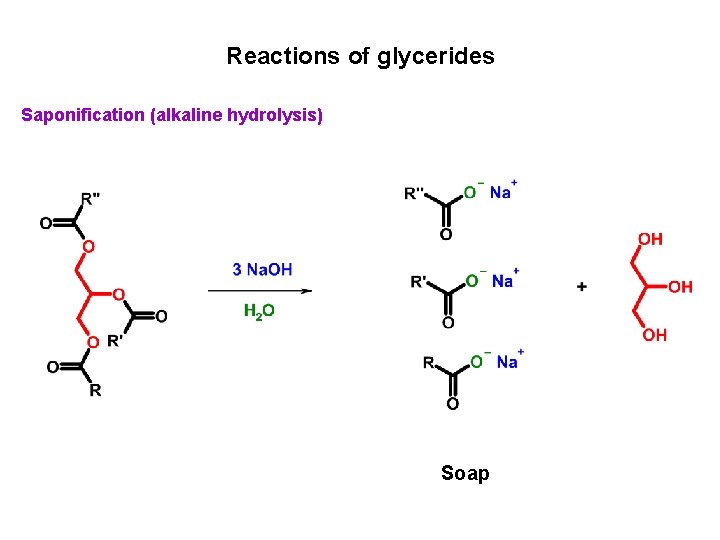
Reactions of glycerides Saponification (alkaline hydrolysis) Soap
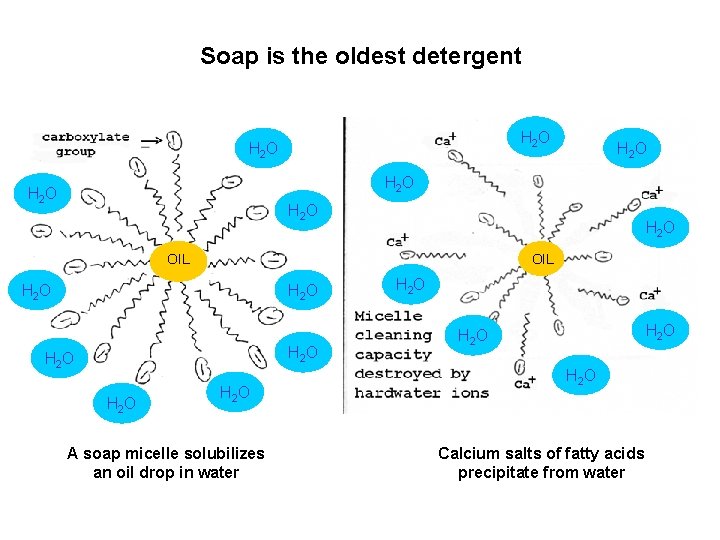
Soap is the oldest detergent H 2 O H 2 O OIL H 2 O H 2 O A soap micelle solubilizes an oil drop in water H 2 O Calcium salts of fatty acids precipitate from water
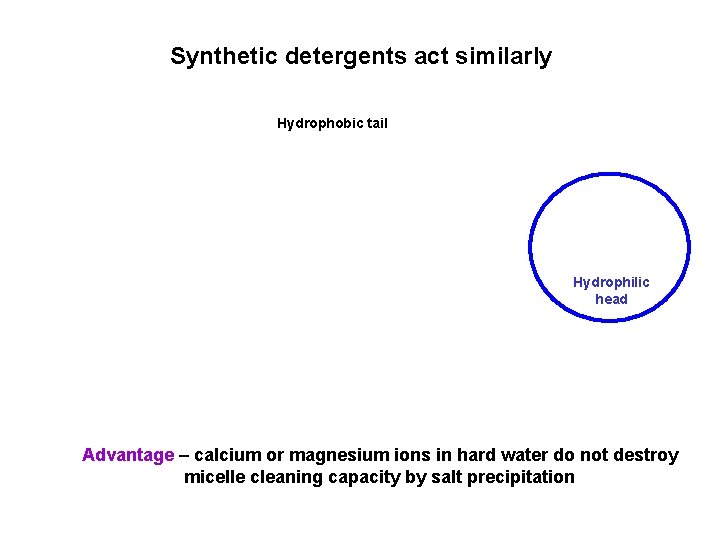
Synthetic detergents act similarly Hydrophobic tail Hydrophilic head Advantage – calcium or magnesium ions in hard water do not destroy micelle cleaning capacity by salt precipitation

Reactions of glycerides Transesterification (alcoholysis) Mixture of fatty acids methyl esters Reaction used for: • analysis of fatty acids composition in fats (GC), • „biodiesel” manufacturing

Reactions of glycerides Hydrogenation Triolein Margarine production from plant oils 2 -oleoyl-1, 3 -distearoylglycerol
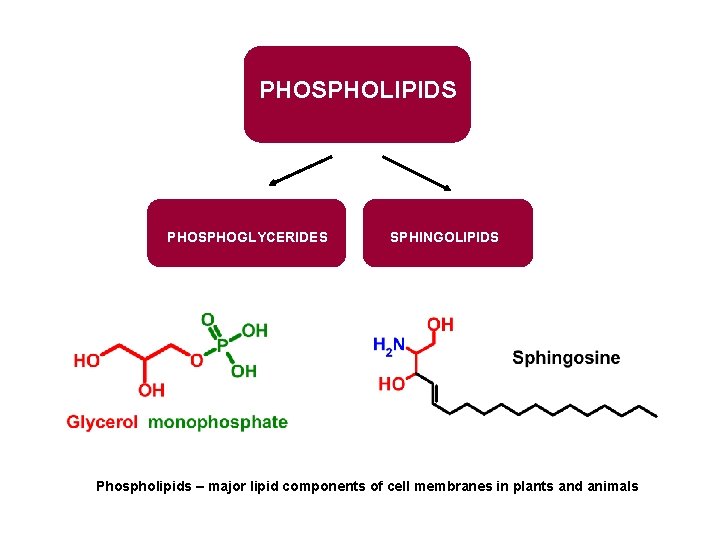
PHOSPHOLIPIDS PHOSPHOGLYCERIDES SPHINGOLIPIDS Phospholipids – major lipid components of cell membranes in plants and animals

PHOSPHOGLYCERIDES LECITHINS (choline derivatives) CEPHALINS (ethanolamine derivatives) R – usually saturated chain R’ – usually unsaturated chain
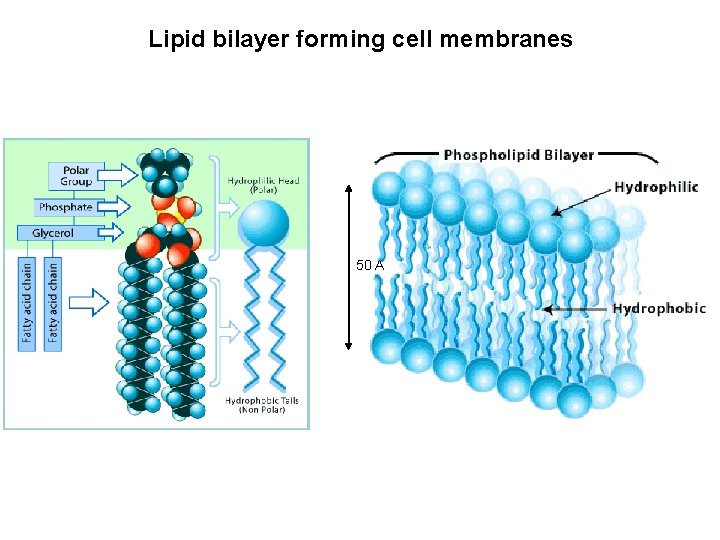
Lipid bilayer forming cell membranes 50 A
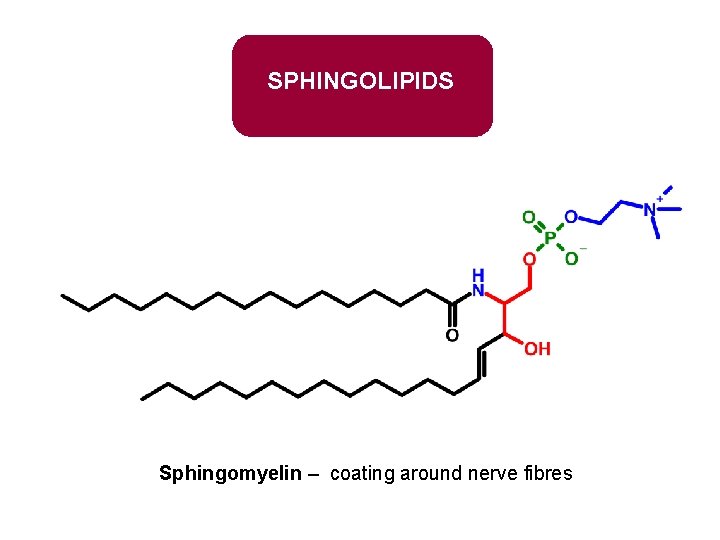
SPHINGOLIPIDS Sphingomyelin – coating around nerve fibres
NONHYDROLYSABLE LIPIDS PROSTAGLANDINS STEROIDS TERPENES
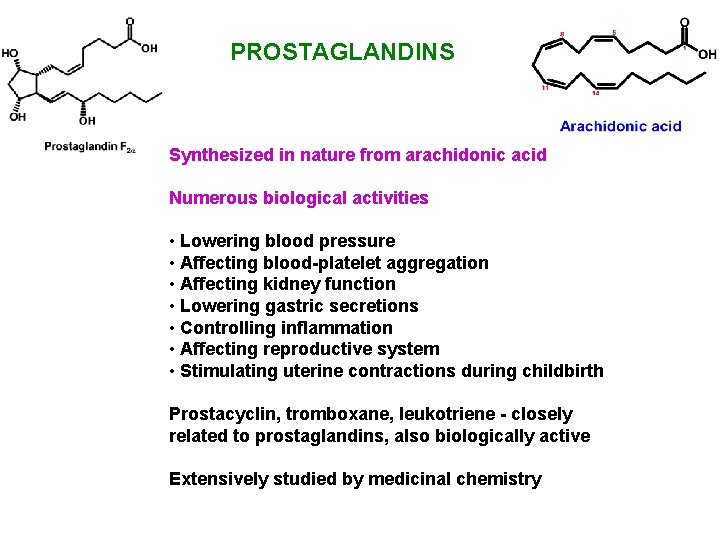
PROSTAGLANDINS Synthesized in nature from arachidonic acid Numerous biological activities • Lowering blood pressure • Affecting blood-platelet aggregation • Affecting kidney function • Lowering gastric secretions • Controlling inflammation • Affecting reproductive system • Stimulating uterine contractions during childbirth Prostacyclin, tromboxane, leukotriene - closely related to prostaglandins, also biologically active Extensively studied by medicinal chemistry
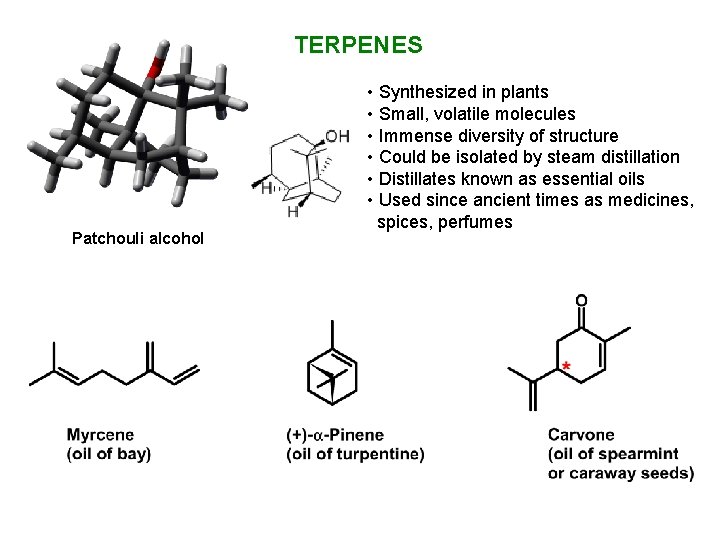
TERPENES Patchouli alcohol • Synthesized in plants • Small, volatile molecules • Immense diversity of structure • Could be isolated by steam distillation • Distillates known as essential oils • Used since ancient times as medicines, spices, perfumes
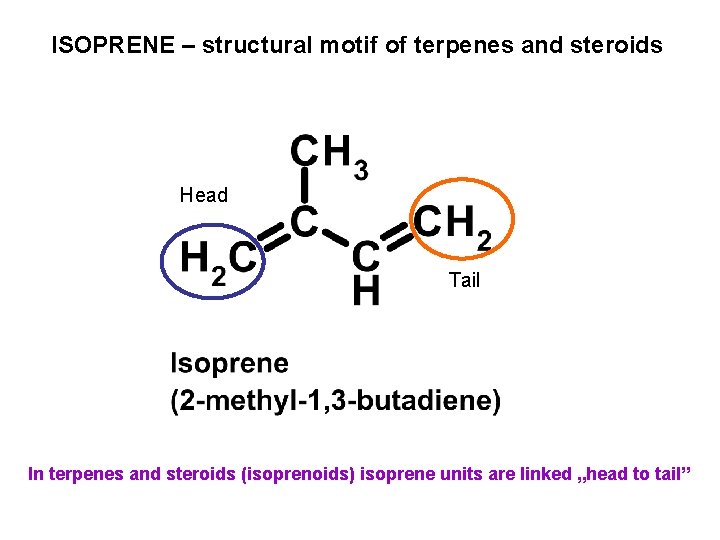
ISOPRENE – structural motif of terpenes and steroids Head Tail In terpenes and steroids (isoprenoids) isoprene units are linked „head to tail”
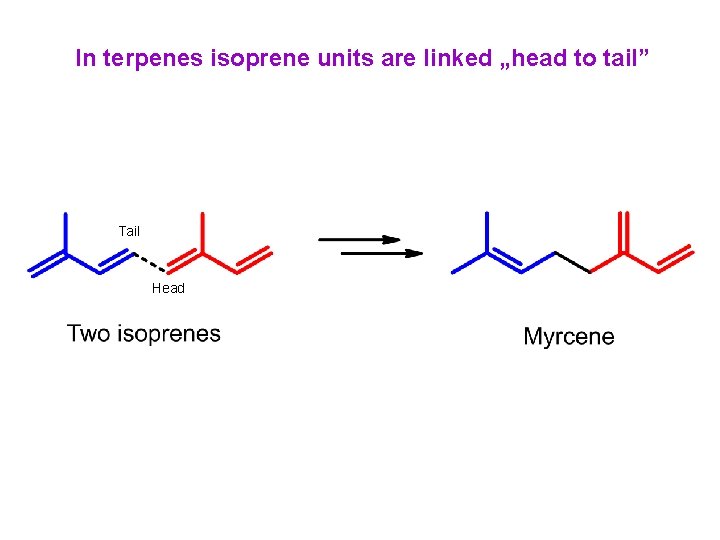
In terpenes isoprene units are linked „head to tail” Tail Head

In terpenes isoprene units are linked „head to tail” Head Tail
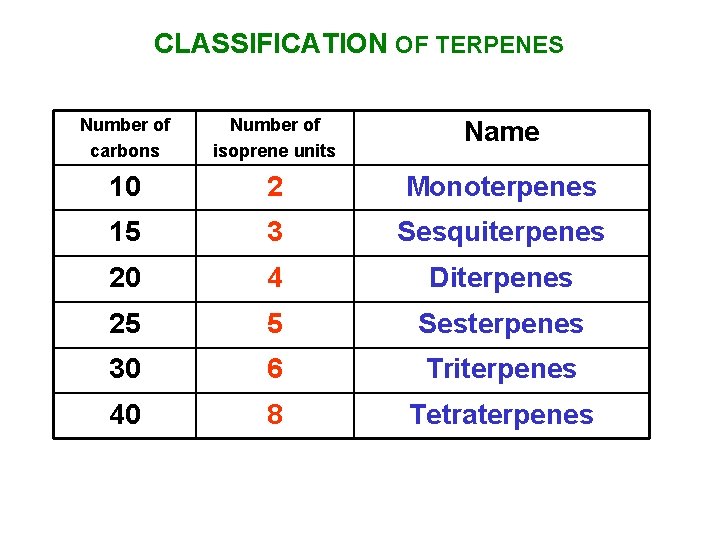
CLASSIFICATION OF TERPENES Number of carbons Number of isoprene units Name 10 2 Monoterpenes 15 3 Sesquiterpenes 20 4 Diterpenes 25 5 Sesterpenes 30 6 Triterpenes 40 8 Tetraterpenes
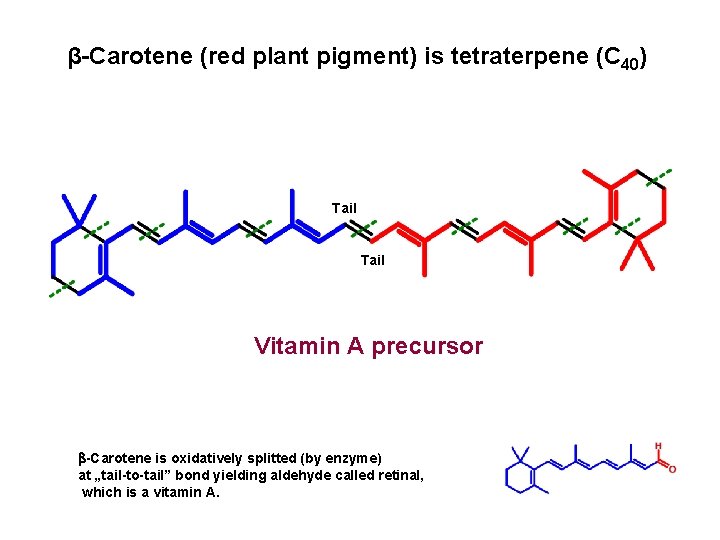
β-Carotene (red plant pigment) is tetraterpene (C 40) Tail Vitamin A precursor -Carotene is oxidatively splitted (by enzyme) at „tail-to-tail” bond yielding aldehyde called retinal, which is a vitamin A.
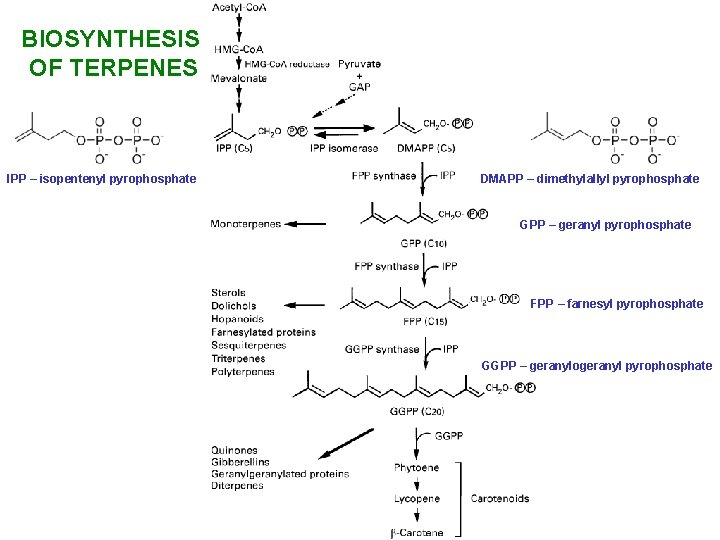
BIOSYNTHESIS OF TERPENES IPP – isopentenyl pyrophosphate DMAPP – dimethylallyl pyrophosphate GPP – geranyl pyrophosphate FPP – farnesyl pyrophosphate GGPP – geranylogeranyl pyrophosphate

Formation of cyclic monoterpenes from geranyl pyrophosphate
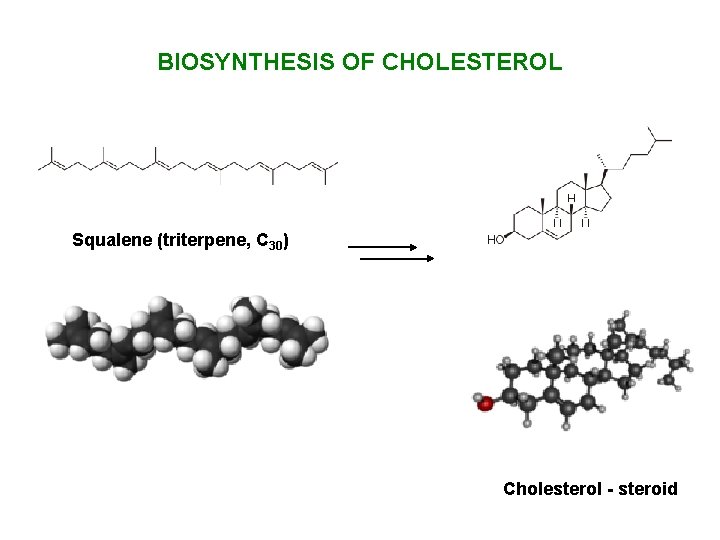
BIOSYNTHESIS OF CHOLESTEROL Squalene (triterpene, C 30) Cholesterol - steroid
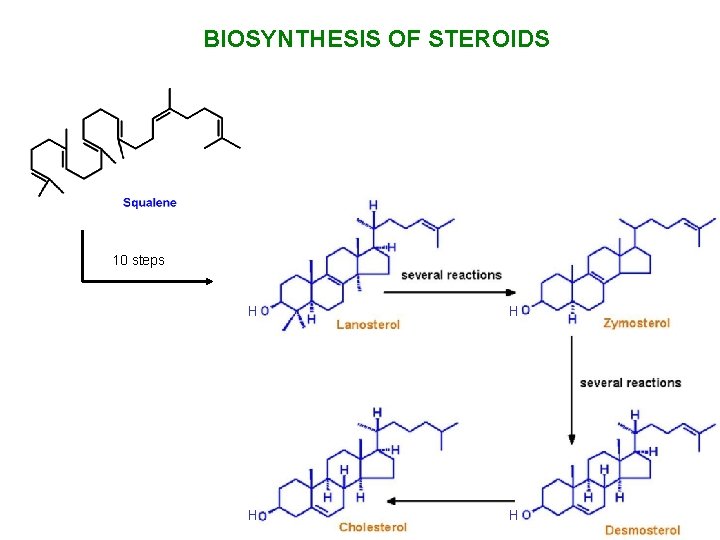
BIOSYNTHESIS OF STEROIDS 10 steps H H
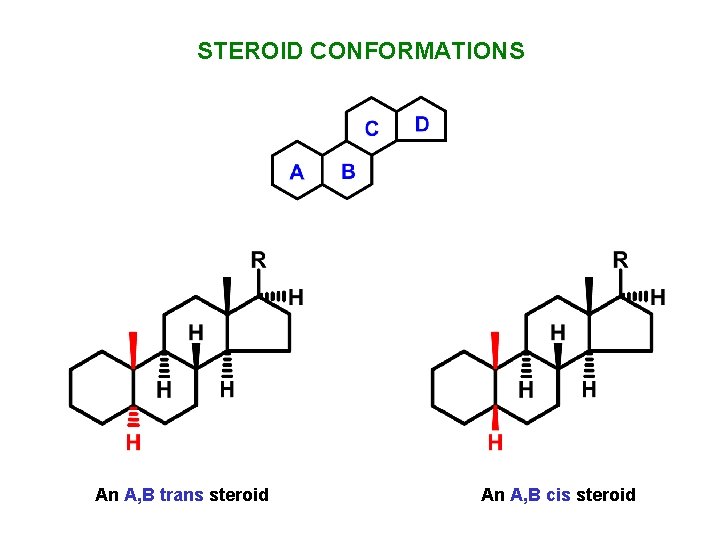
STEROID CONFORMATIONS An A, B trans steroid An A, B cis steroid
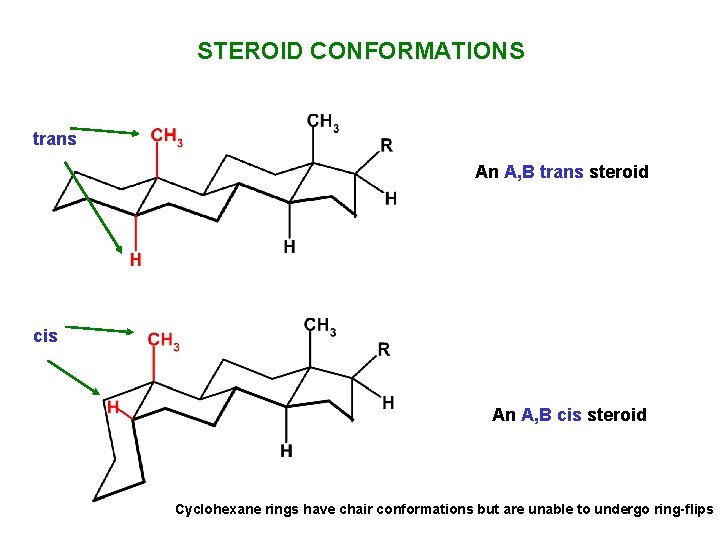
STEROID CONFORMATIONS trans An A, B trans steroid cis An A, B cis steroid Cyclohexane rings have chair conformations but are unable to undergo ring-flips
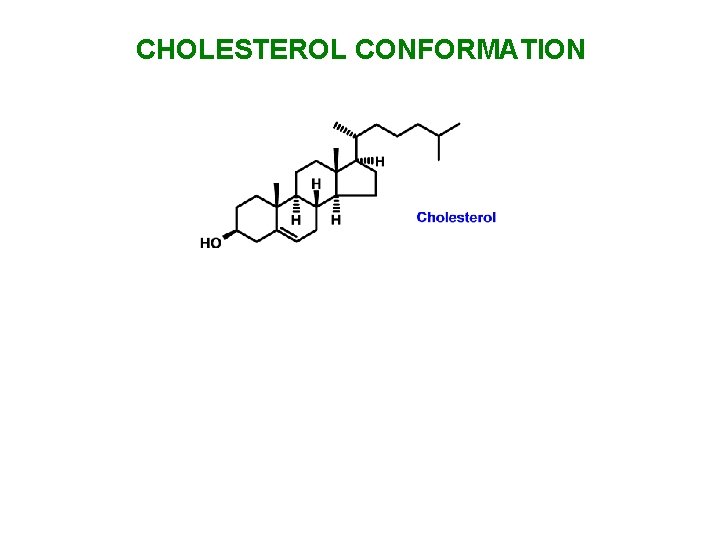
CHOLESTEROL CONFORMATION

STEROID HORMONES Male sex hormones

STEROID HORMONES Female sex hormones

STEROID HORMONES Adrenocortical hormones A mineralocorticoid A glucocorticoid

SYNTHETIC STEROIDS A synthetic estrogen A synthetic progestin Components of oral contraceptive pills

ANABOLIC STEROIDS Both steroids have strong tissue-building effect
- Slides: 45