Lipids Fats Oils AP Biology long term energy

Lipids: Fats & Oils AP Biology

long term energy storage concentrated energy AP Biology
Lipids • Lipids are composed of C, H, O – long hydrocarbon chains (H-C) • “Family groups” – fats – phospholipids – steroids • Do not form polymers – big molecules made of smaller subunits – not a continuing chain AP Biology
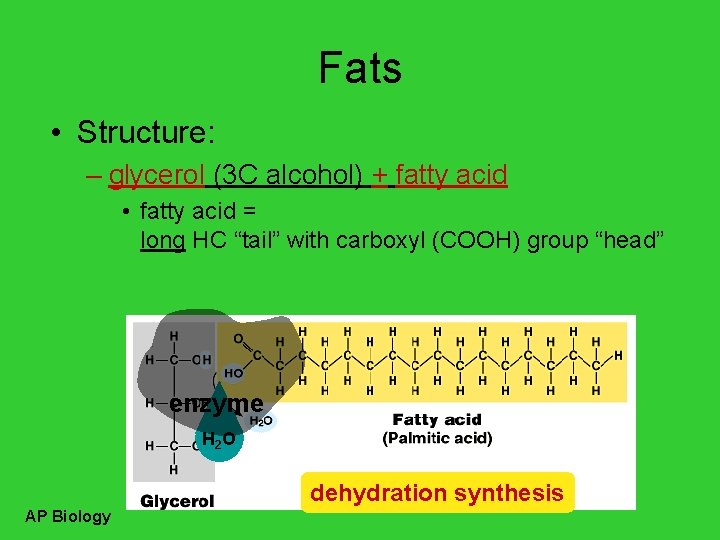
Fats • Structure: – glycerol (3 C alcohol) + fatty acid • fatty acid = long HC “tail” with carboxyl (COOH) group “head” enzyme H 2 O dehydration synthesis AP Biology

Building Fats • Triacylglycerol – 3 fatty acids linked to glycerol – ester linkage = between OH & COOH hydroxyl AP Biology carboxyl

Dehydration synthesis H 2 O dehydration synthesis enzyme H 2 O AP Biology enzyme HO
Fats store energy Why do humans like fatty foods? • Long HC chain – polar or non-polar? – hydrophilic or hydrophobic? • Function: – energy storage • concentrated – all H-C! • 2 x carbohydrates – cushion organs – insulates body AP Biology • think whale blubber!
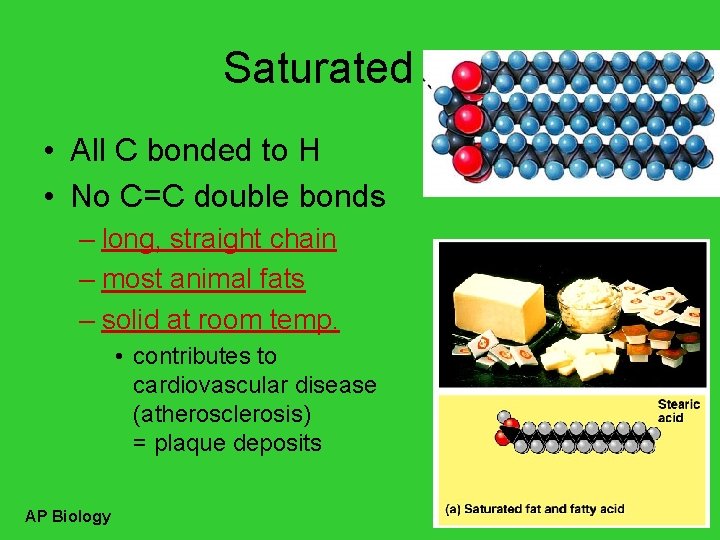
Saturated fats • All C bonded to H • No C=C double bonds – long, straight chain – most animal fats – solid at room temp. • contributes to cardiovascular disease (atherosclerosis) = plaque deposits AP Biology

Unsaturated fats • C=C double bonds in the fatty acids – plant & fish fats – vegetable oils – liquid at room temperature • the kinks made by double bonded C prevent the molecules from packing tightly together AP Biology
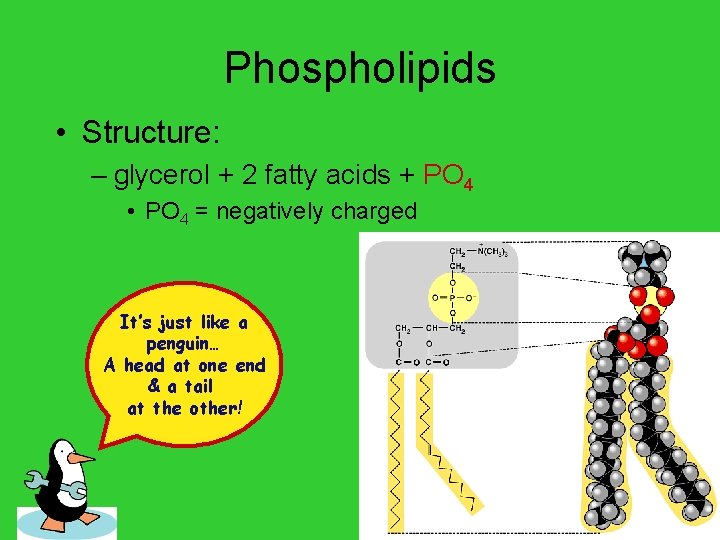
Phospholipids • Structure: – glycerol + 2 fatty acids + PO 4 • PO 4 = negatively charged It’s just like a penguin… A head at one end & a tail at the other! AP Biology
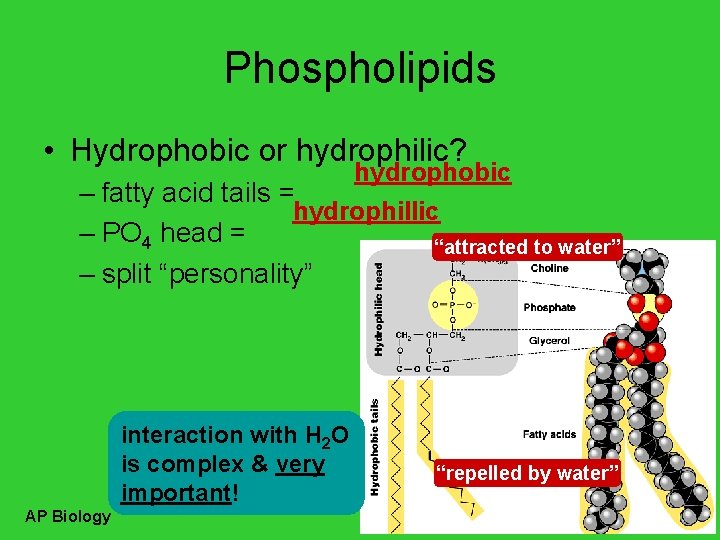
Phospholipids • Hydrophobic or hydrophilic? hydrophobic – fatty acid tails = hydrophillic – PO 4 head = “attracted to water” – split “personality” interaction with H 2 O is complex & very important! AP Biology “repelled by water”
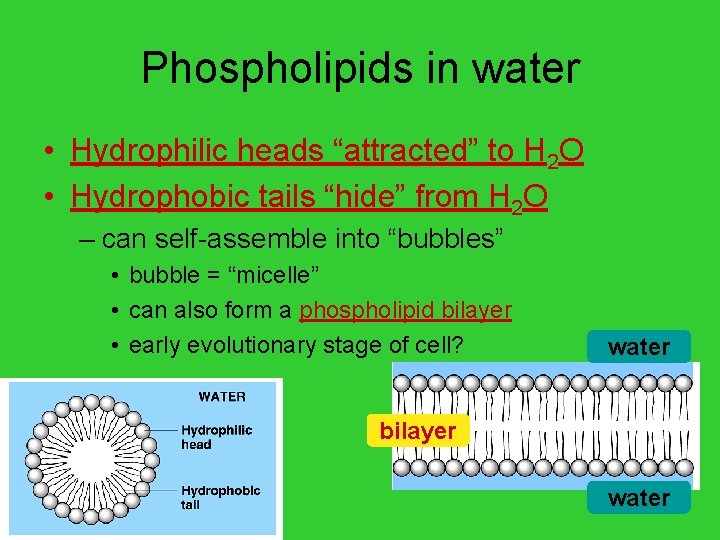
Phospholipids in water • Hydrophilic heads “attracted” to H 2 O • Hydrophobic tails “hide” from H 2 O – can self-assemble into “bubbles” • bubble = “micelle” • can also form a phospholipid bilayer • early evolutionary stage of cell? water bilayer AP Biology water

Why is this important? • Phospholipids create a barrier in water – define outside vs. inside – they make cell membranes! AP Biology
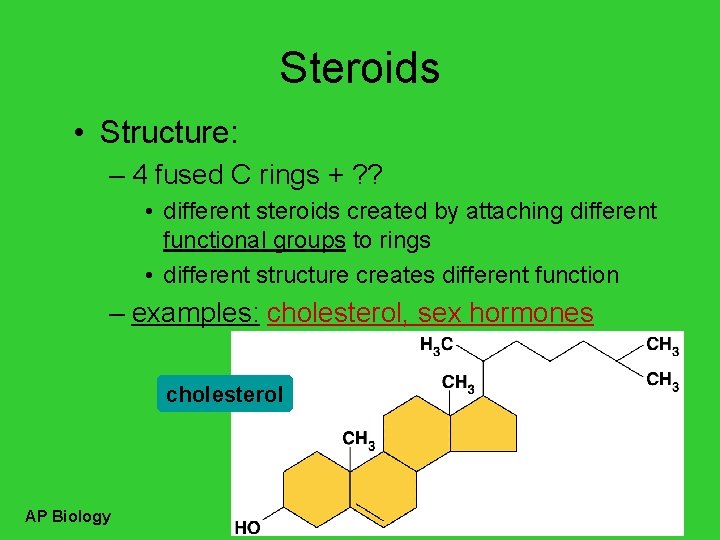
Steroids • Structure: – 4 fused C rings + ? ? • different steroids created by attaching different functional groups to rings • different structure creates different function – examples: cholesterol, sex hormones cholesterol AP Biology
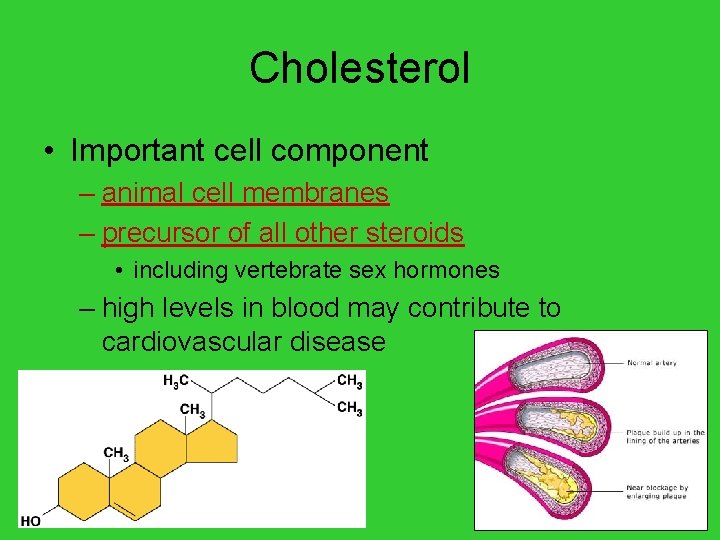
Cholesterol • Important cell component – animal cell membranes – precursor of all other steroids • including vertebrate sex hormones – high levels in blood may contribute to cardiovascular disease AP Biology
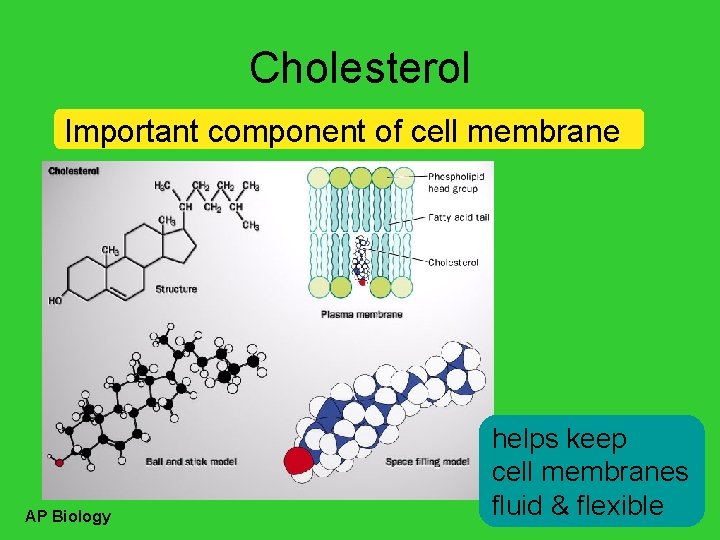
Cholesterol Important component of cell membrane AP Biology helps keep cell membranes fluid & flexible
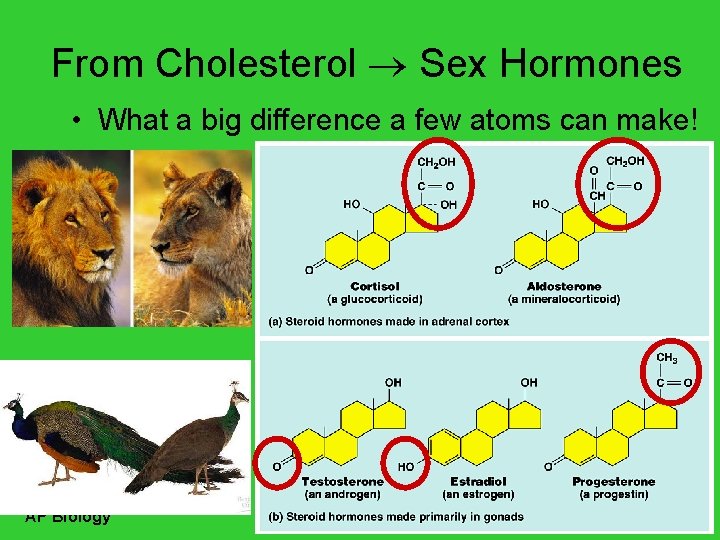
From Cholesterol Sex Hormones • What a big difference a few atoms can make! AP Biology

Phospholipids & cells • Phospholipids of cell membrane – double layer = bilayer – hydrophilic heads on outside • in contact with aqueous solution outside of cell and inside of cell – hydrophobic tails on inside • form core – forms barrier between cell & external environment AP Biology

Practice • P. 40 Q 11 -13, 16 -17 AP Biology

Proteins AP Biology

Proteins Multipurpose molecules AP Biology

Proteins • Most structurally & functionally diverse group • Function: involved in almost everything – – enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport (hemoglobin, aquaporin) cell communication • signals (insulin & other hormones) • receptors – defense (antibodies) – movement (actin & myosin) – storage (bean seed proteins) AP Biology
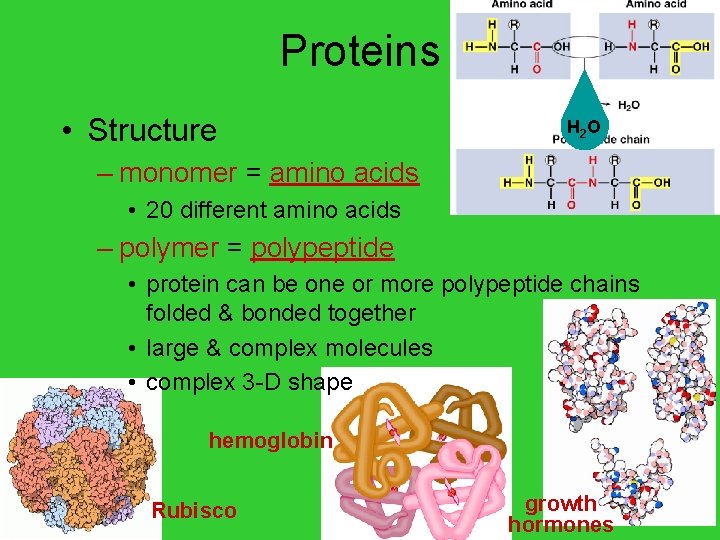
Proteins • Structure H 2 O – monomer = amino acids • 20 different amino acids – polymer = polypeptide • protein can be one or more polypeptide chains folded & bonded together • large & complex molecules • complex 3 -D shape hemoglobin AP Biology Rubisco growth hormones
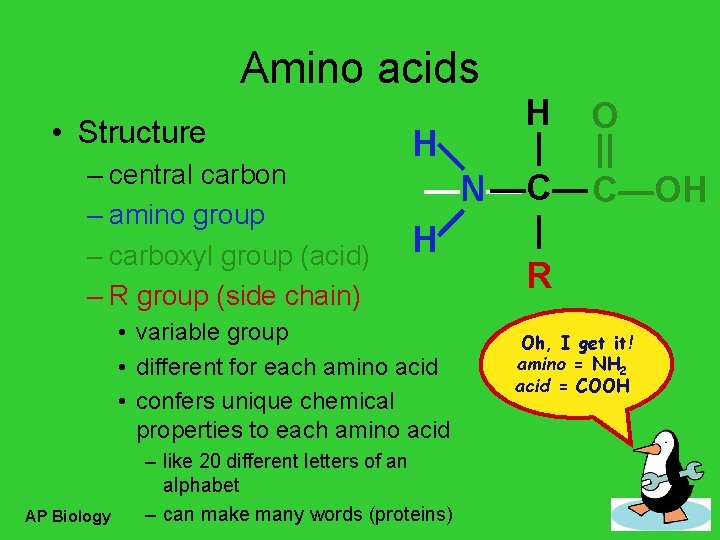
Amino acids • Structure – central carbon – amino group – carboxyl group (acid) – R group (side chain) H O H | || —C— C—OH —N— | H R • variable group • different for each amino acid • confers unique chemical properties to each amino acid AP Biology – like 20 different letters of an alphabet – can make many words (proteins) Oh, I get it! amino = NH 2 acid = COOH
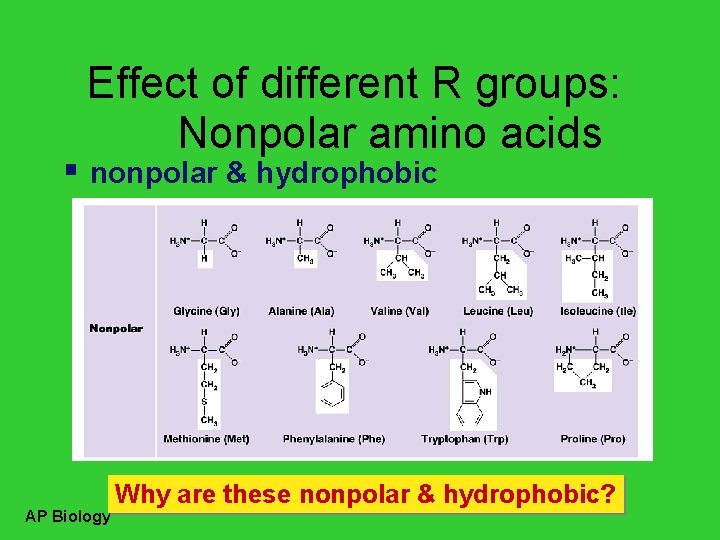
Effect of different R groups: Nonpolar amino acids nonpolar & hydrophobic AP Biology Why are these nonpolar & hydrophobic?

Effect of different R groups: Polar amino acids polar or charged & hydrophilic AP Biology Why are these polar & hydrophillic?
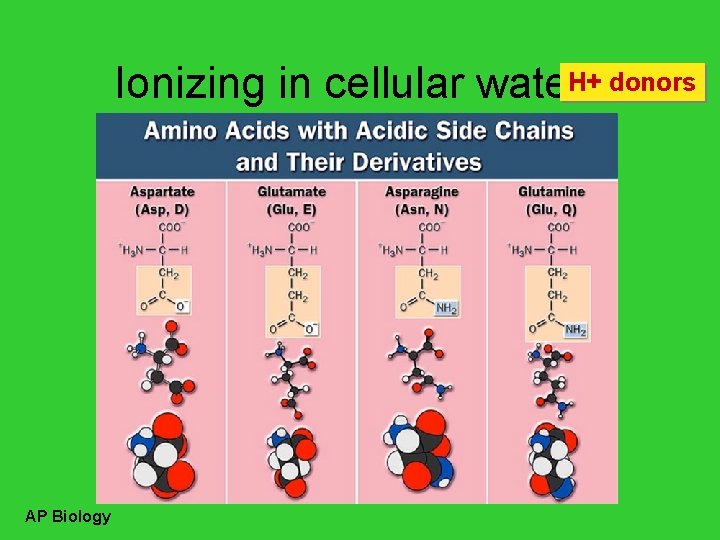
H+ donors Ionizing in cellular waters AP Biology
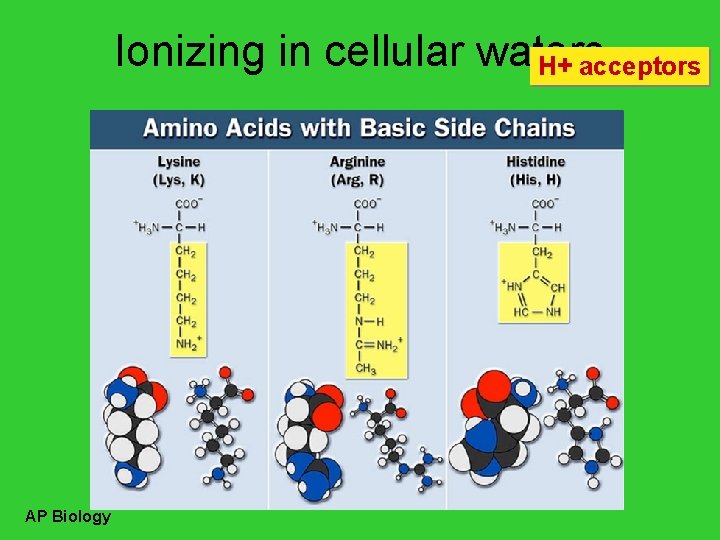
Ionizing in cellular waters H+ acceptors AP Biology
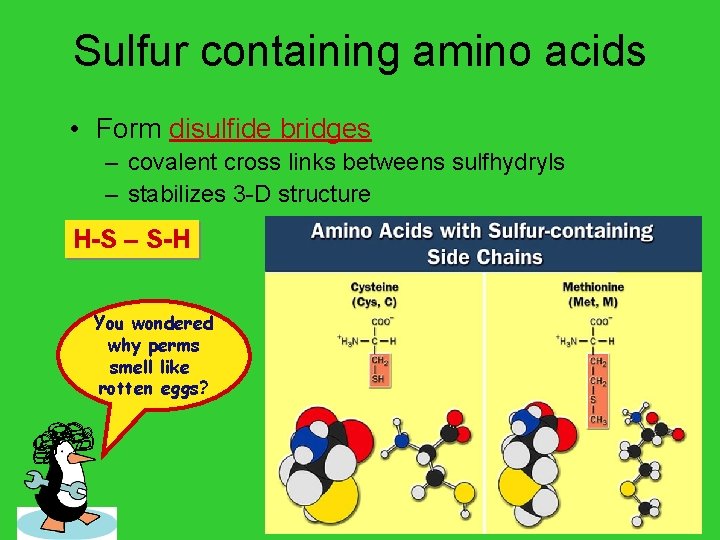
Sulfur containing amino acids • Form disulfide bridges – covalent cross links betweens sulfhydryls – stabilizes 3 -D structure H-S – S-H You wondered why perms smell like rotten eggs? AP Biology
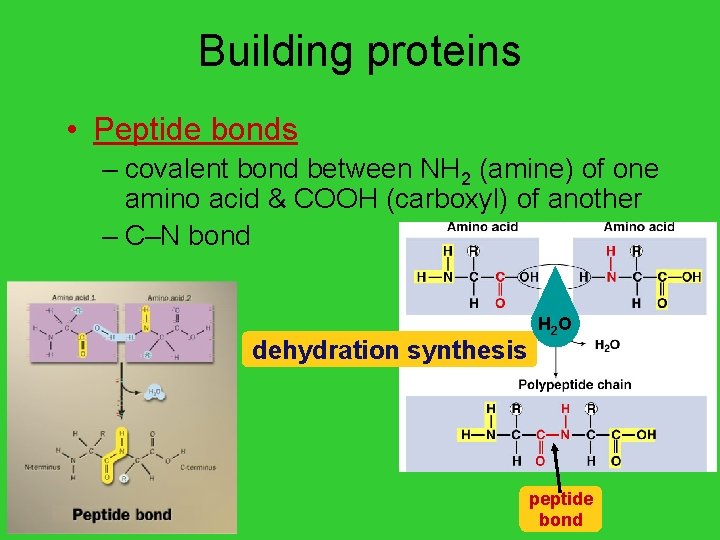
Building proteins • Peptide bonds – covalent bond between NH 2 (amine) of one amino acid & COOH (carboxyl) of another – C–N bond dehydration synthesis AP Biology H 2 O peptide bond
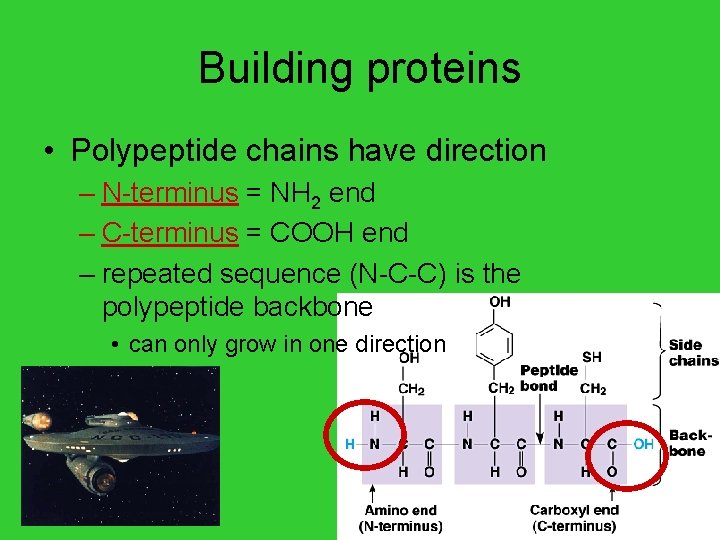
Building proteins • Polypeptide chains have direction – N-terminus = NH 2 end – C-terminus = COOH end – repeated sequence (N-C-C) is the polypeptide backbone • can only grow in one direction AP Biology
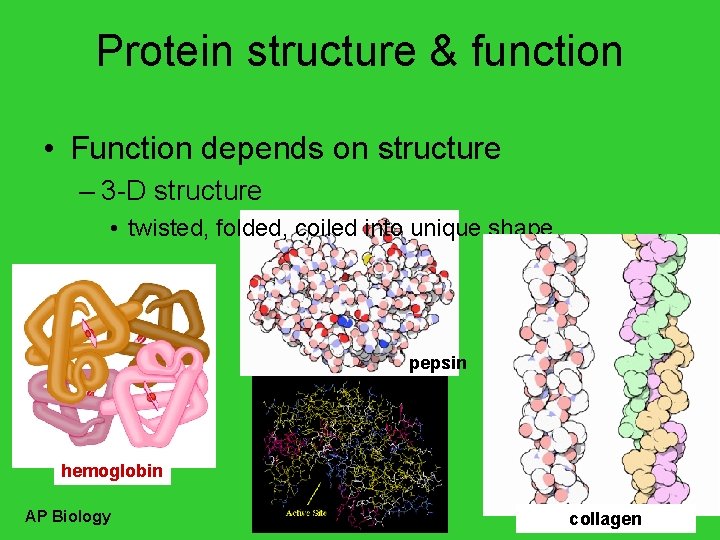
Protein structure & function • Function depends on structure – 3 -D structure • twisted, folded, coiled into unique shape pepsin hemoglobin AP Biology collagen
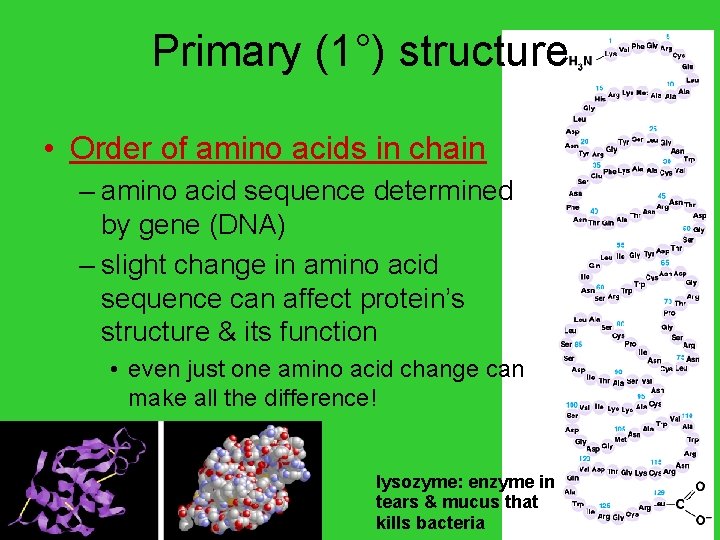
Primary (1°) structure • Order of amino acids in chain – amino acid sequence determined by gene (DNA) – slight change in amino acid sequence can affect protein’s structure & its function • even just one amino acid change can make all the difference! AP Biology lysozyme: enzyme in tears & mucus that kills bacteria
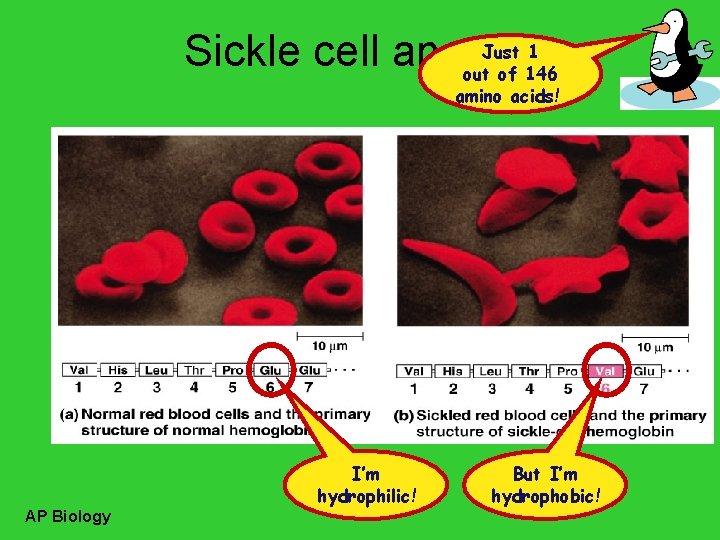
Just 1 Sickle cell anemia out of 146 amino acids! I’m hydrophilic! AP Biology But I’m hydrophobic!
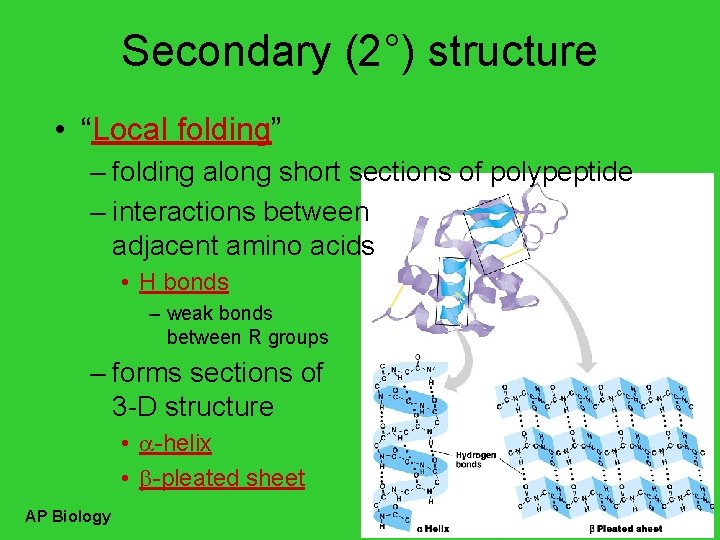
Secondary (2°) structure • “Local folding” – folding along short sections of polypeptide – interactions between adjacent amino acids • H bonds – weak bonds between R groups – forms sections of 3 -D structure • -helix • -pleated sheet AP Biology
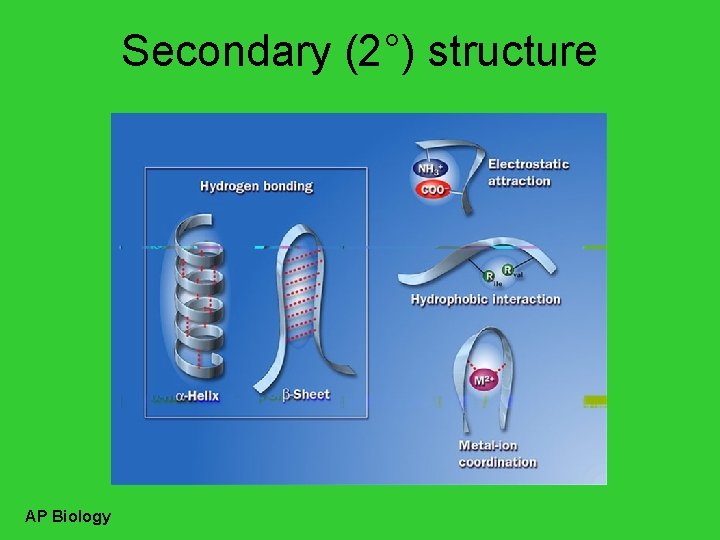
Secondary (2°) structure AP Biology
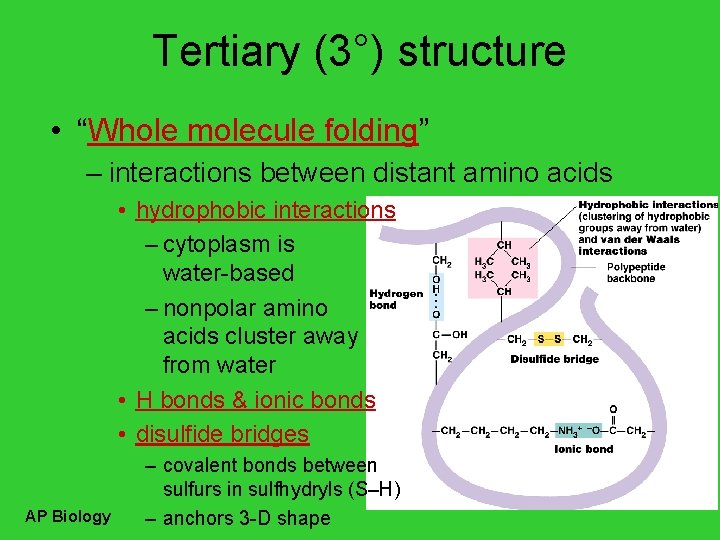
Tertiary (3°) structure • “Whole molecule folding” – interactions between distant amino acids • hydrophobic interactions – cytoplasm is water-based – nonpolar amino acids cluster away from water • H bonds & ionic bonds • disulfide bridges AP Biology – covalent bonds between sulfurs in sulfhydryls (S–H) – anchors 3 -D shape
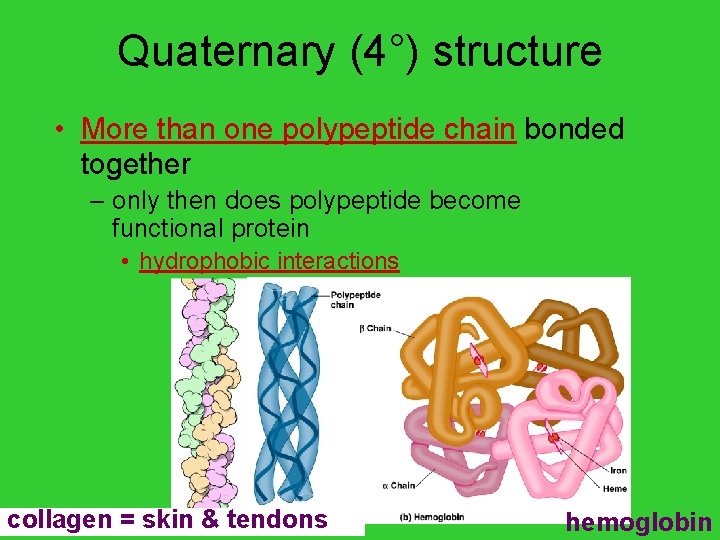
Quaternary (4°) structure • More than one polypeptide chain bonded together – only then does polypeptide become functional protein • hydrophobic interactions AP Biology = skin & tendons collagen hemoglobin
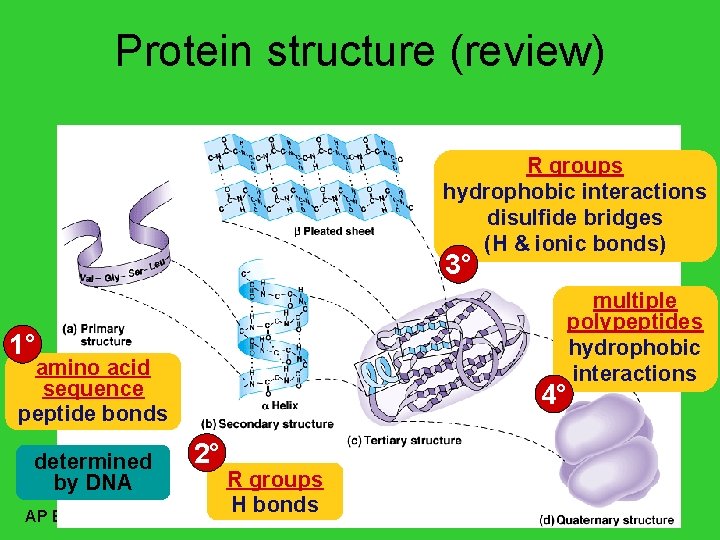
Protein structure (review) R groups hydrophobic interactions disulfide bridges (H & ionic bonds) 3° multiple polypeptides hydrophobic interactions 1° amino acid sequence peptide bonds determined by DNA AP Biology 4° 2° R groups H bonds
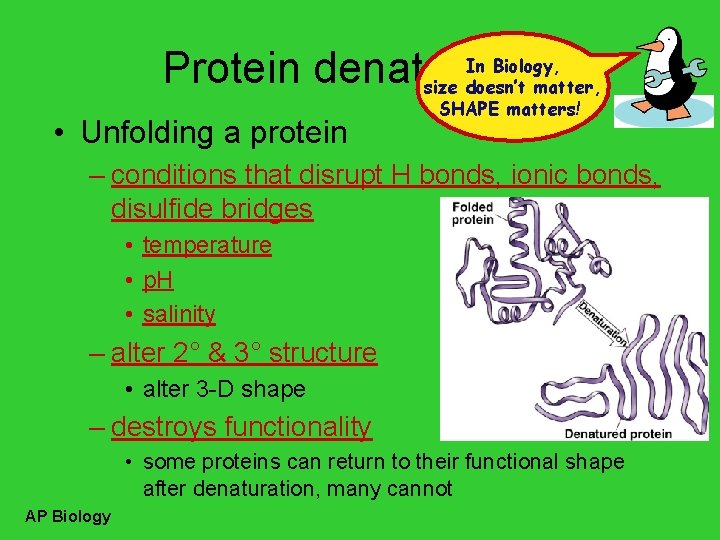
In Biology, Protein denaturation size doesn’t matter, • Unfolding a protein SHAPE matters! – conditions that disrupt H bonds, ionic bonds, disulfide bridges • temperature • p. H • salinity – alter 2° & 3° structure • alter 3 -D shape – destroys functionality • some proteins can return to their functional shape after denaturation, many cannot AP Biology
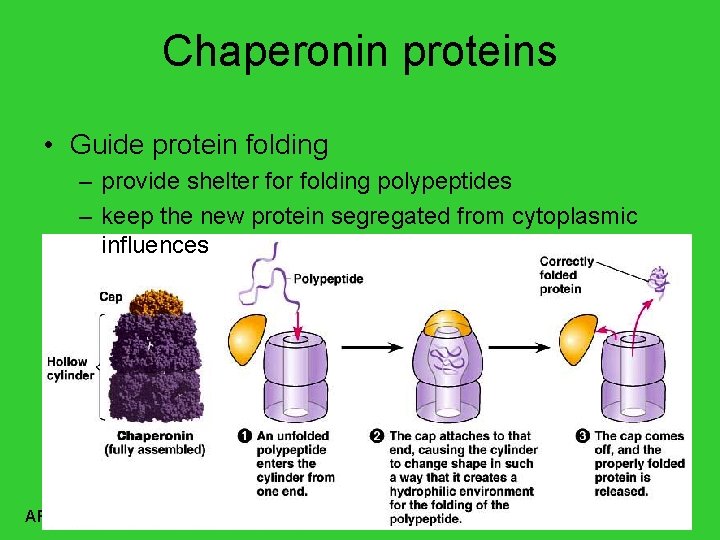
Chaperonin proteins • Guide protein folding – provide shelter folding polypeptides – keep the new protein segregated from cytoplasmic influences AP Biology
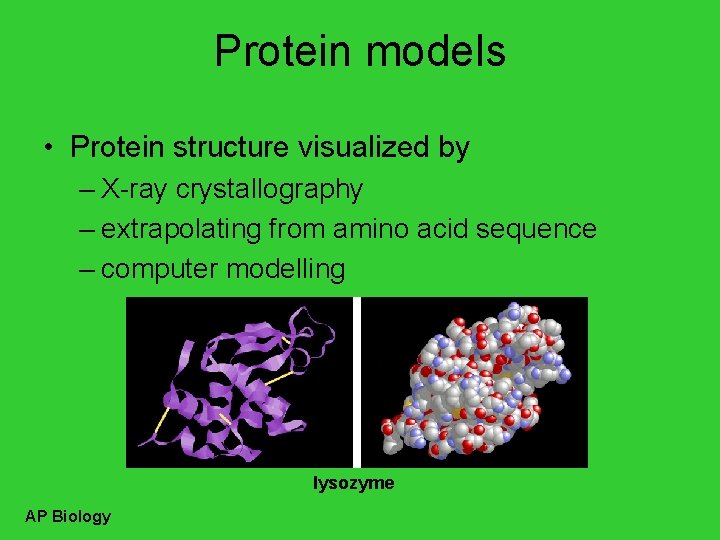
Protein models • Protein structure visualized by – X-ray crystallography – extrapolating from amino acid sequence – computer modelling lysozyme AP Biology

Practice • P. 50 Q 19 -26 AP Biology

Nucleic Acids Information storage AP Biology
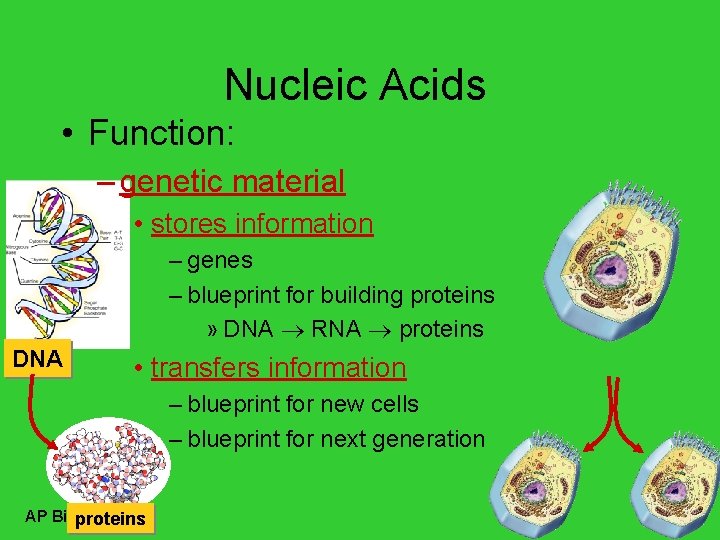
Nucleic Acids • Function: – genetic material • stores information – genes – blueprint for building proteins » DNA RNA proteins DNA • transfers information – blueprint for new cells – blueprint for next generation AP Biology proteins
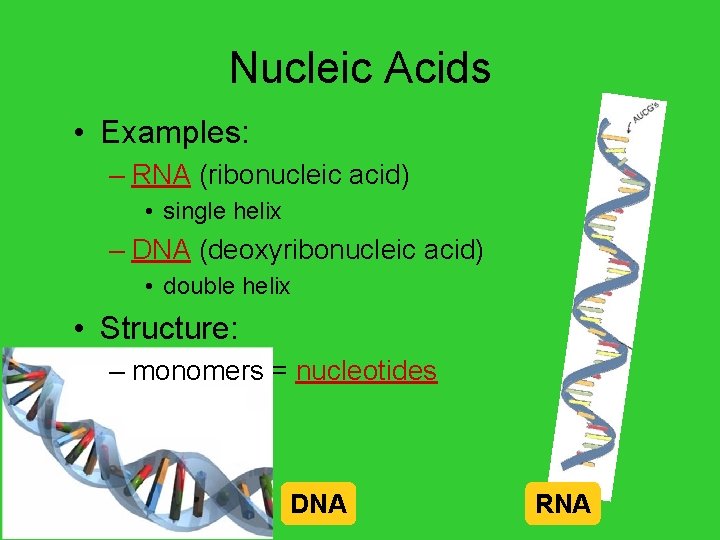
Nucleic Acids • Examples: – RNA (ribonucleic acid) • single helix – DNA (deoxyribonucleic acid) • double helix • Structure: – monomers = nucleotides AP Biology DNA RNA
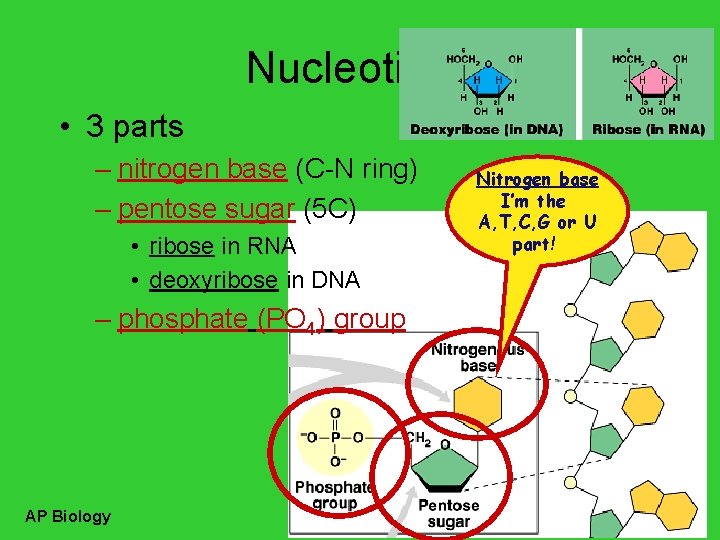
Nucleotides • 3 parts – nitrogen base (C-N ring) – pentose sugar (5 C) • ribose in RNA • deoxyribose in DNA – phosphate (PO 4) group AP Biology Nitrogen base I’m the A, T, C, G or U part!
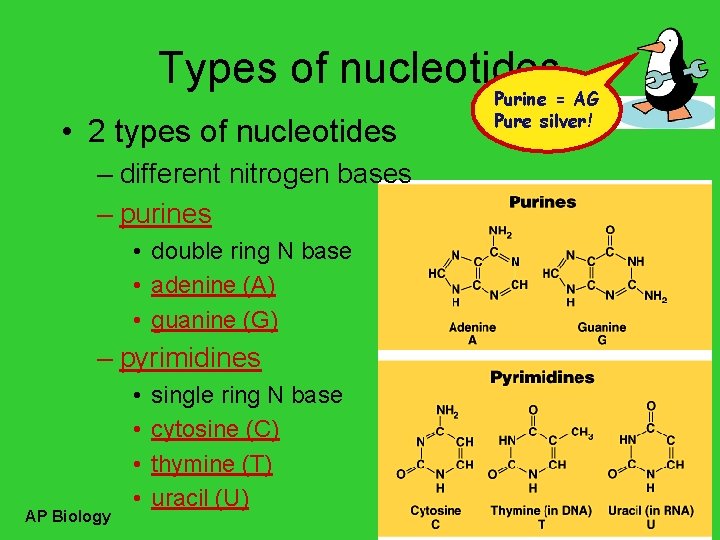
Types of nucleotides • 2 types of nucleotides – different nitrogen bases – purines • double ring N base • adenine (A) • guanine (G) – pyrimidines AP Biology • • single ring N base cytosine (C) thymine (T) uracil (U) Purine = AG Pure silver!
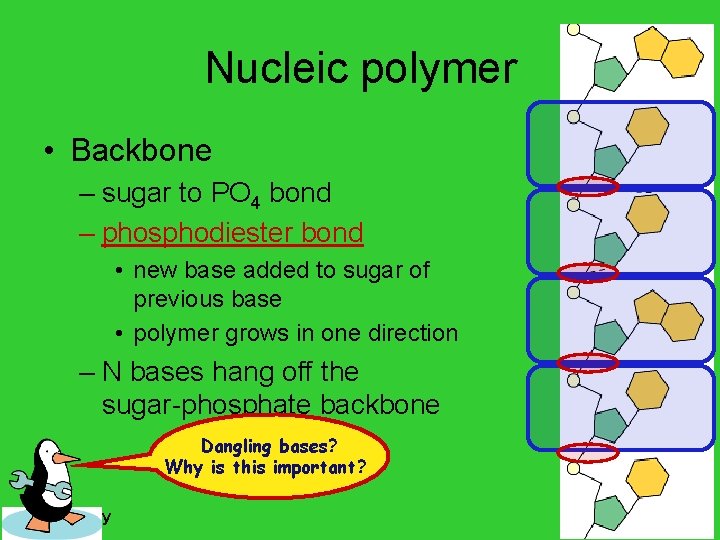
Nucleic polymer • Backbone – sugar to PO 4 bond – phosphodiester bond • new base added to sugar of previous base • polymer grows in one direction – N bases hang off the sugar-phosphate backbone Dangling bases? Why is this important? AP Biology
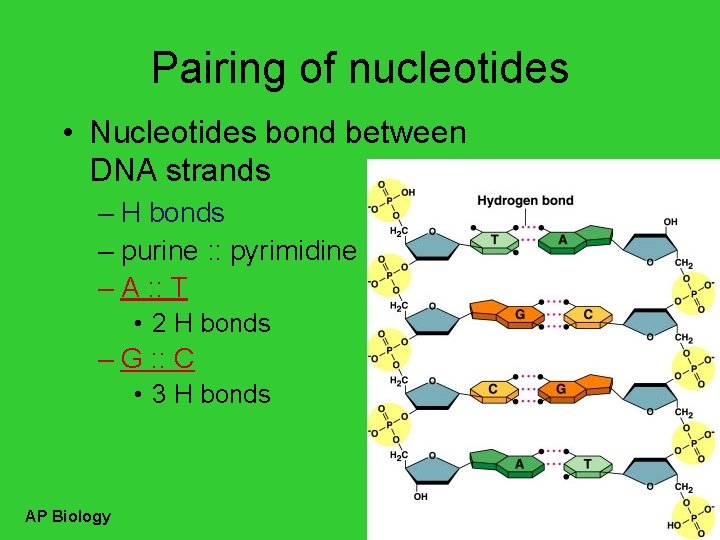
Pairing of nucleotides • Nucleotides bond between DNA strands – H bonds – purine : : pyrimidine – A : : T • 2 H bonds – G : : C • 3 H bonds AP Biology
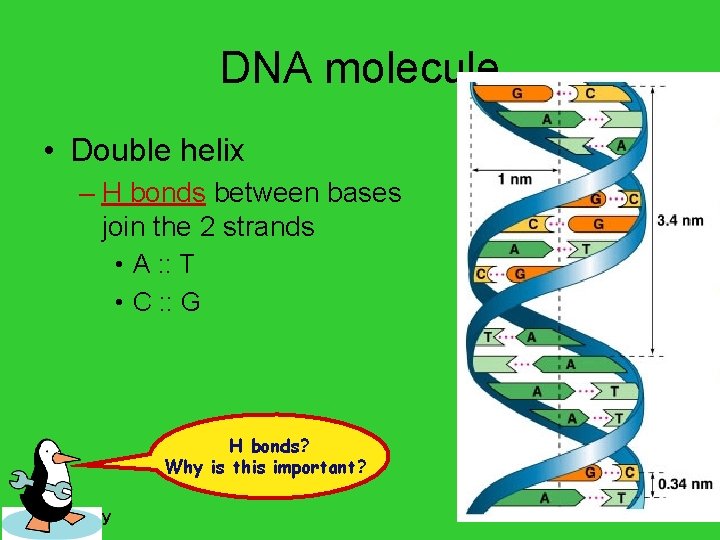
DNA molecule • Double helix – H bonds between bases join the 2 strands • A : : T • C : : G H bonds? Why is this important? AP Biology
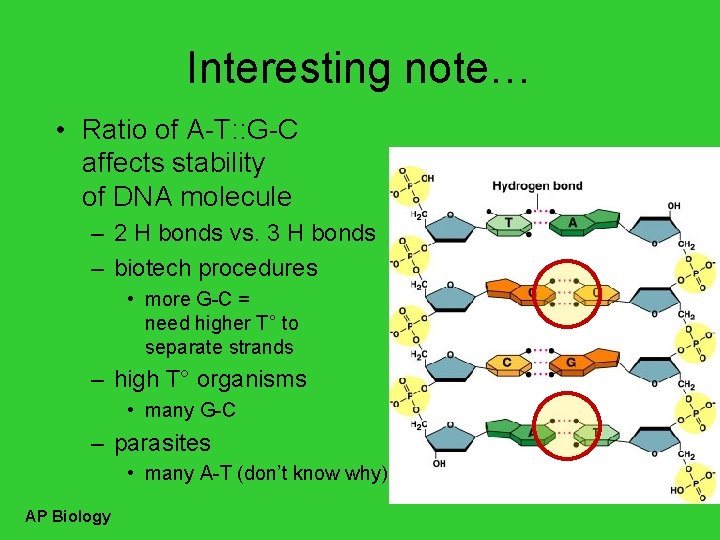
Interesting note… • Ratio of A-T: : G-C affects stability of DNA molecule – 2 H bonds vs. 3 H bonds – biotech procedures • more G-C = need higher T° to separate strands – high T° organisms • many G-C – parasites • many A-T (don’t know why) AP Biology
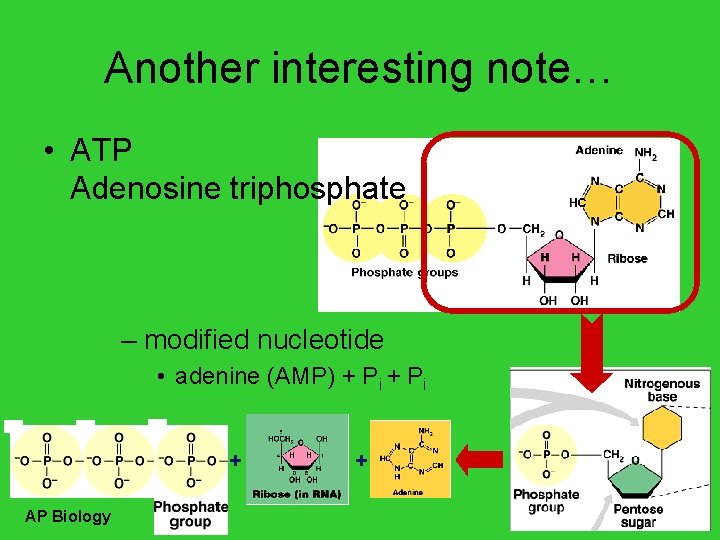
Another interesting note… • ATP Adenosine triphosphate – modified nucleotide • adenine (AMP) + Pi + AP Biology +

Macromolecule Review AP Biology
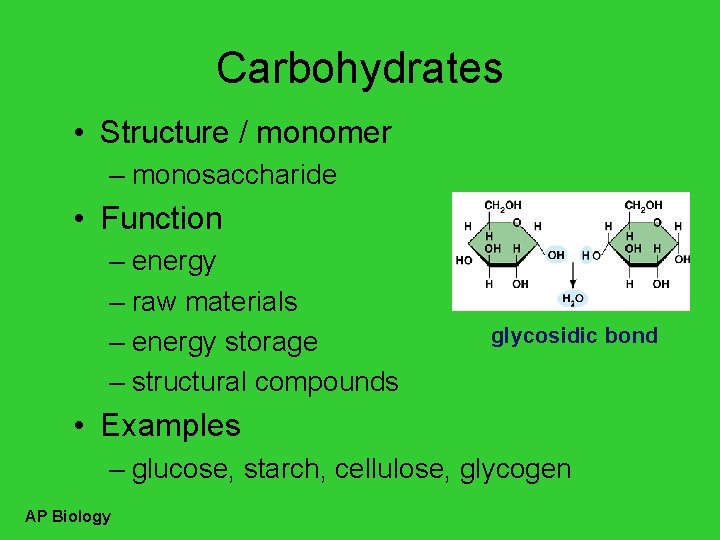
Carbohydrates • Structure / monomer – monosaccharide • Function – energy – raw materials – energy storage – structural compounds glycosidic bond • Examples – glucose, starch, cellulose, glycogen AP Biology
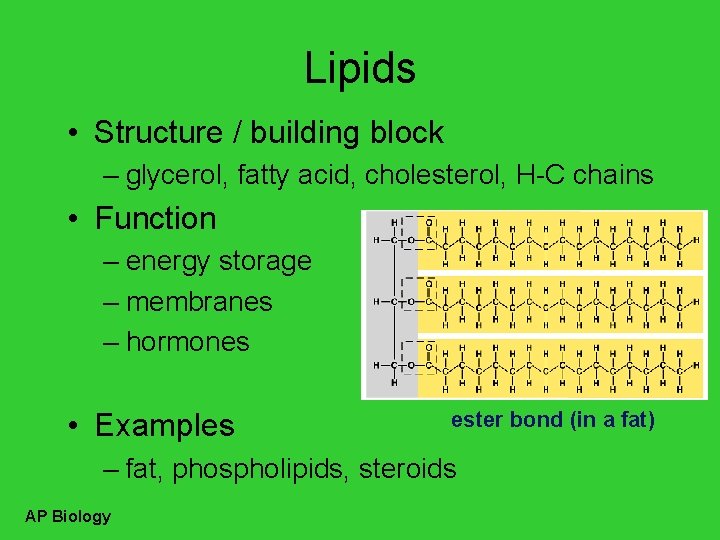
Lipids • Structure / building block – glycerol, fatty acid, cholesterol, H-C chains • Function – energy storage – membranes – hormones • Examples ester bond (in a fat) – fat, phospholipids, steroids AP Biology
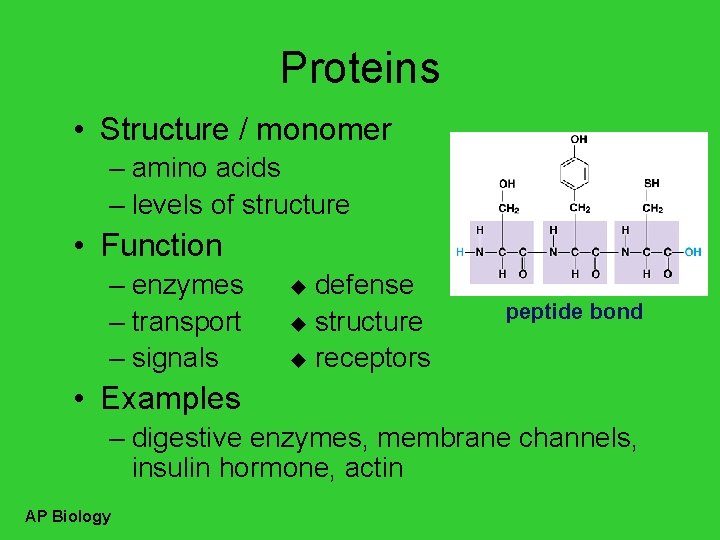
Proteins • Structure / monomer – amino acids – levels of structure • Function – enzymes – transport – signals defense u structure u receptors u peptide bond • Examples – digestive enzymes, membrane channels, insulin hormone, actin AP Biology
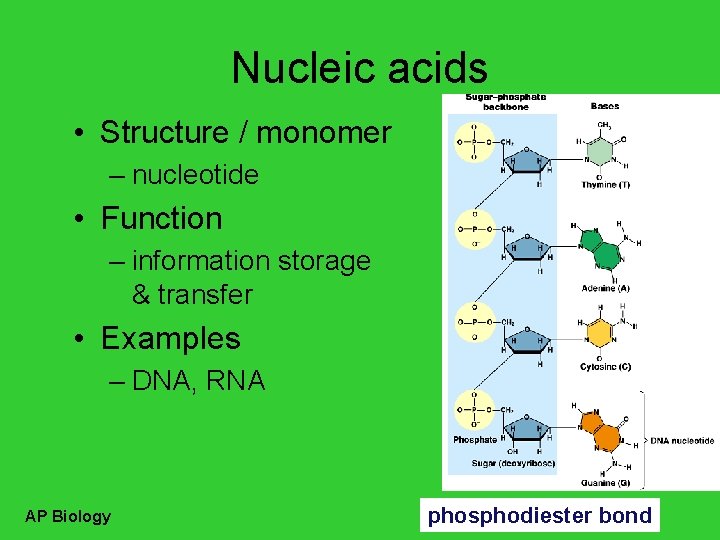
Nucleic acids • Structure / monomer – nucleotide • Function – information storage & transfer • Examples – DNA, RNA AP Biology phosphodiester bond
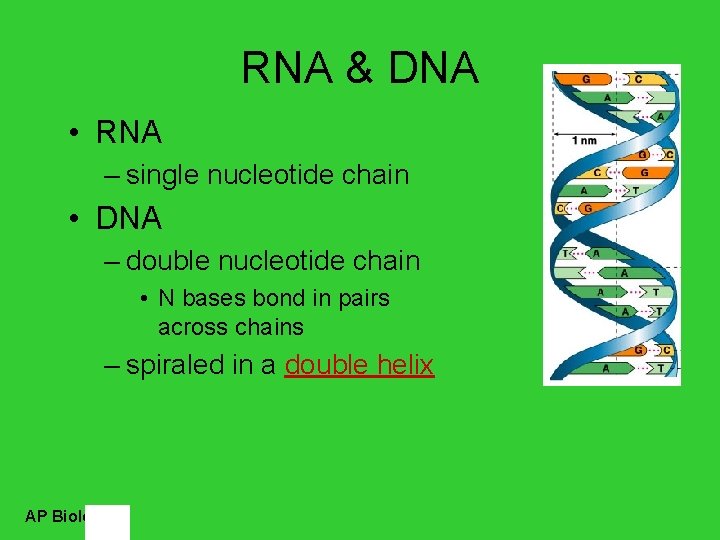
RNA & DNA • RNA – single nucleotide chain • DNA – double nucleotide chain • N bases bond in pairs across chains – spiraled in a double helix AP Biology
- Slides: 59