Lipids fats Main Function longterm energy storage 2









- Slides: 15

Lipids (fats) ü Main Function: long-term energy storage 2 Lipids Which are made of Glycerol & 3 Fatty Acids which contain Carbon, hydrogen, oxygen main function ENERGY STORAGE long-term ü Special Feature: contain more energy per gram than any 3 molecule 4 other biological Nucleic acids ü Groupings: Mostly C and H Proteins atoms(hydrocarbons) (e. g. , DNA/RNA) Which 1. are made of ü Types: Triglycerides Which are made of 3. Steroids 2. Phospholipids Nucleotides which contain Carbon, hydrogen oxygen, nitrogen, phosphorus main function Amino Acids which contain Carbon, hydrogen, oxygen, nitrogen, main function CATALYSIS ENCODING & HEREDITARY Plant oils (liquid @ room temp) STRUCTURE INFORMATION /SUPPORT Animal fat (solid @ room temp)

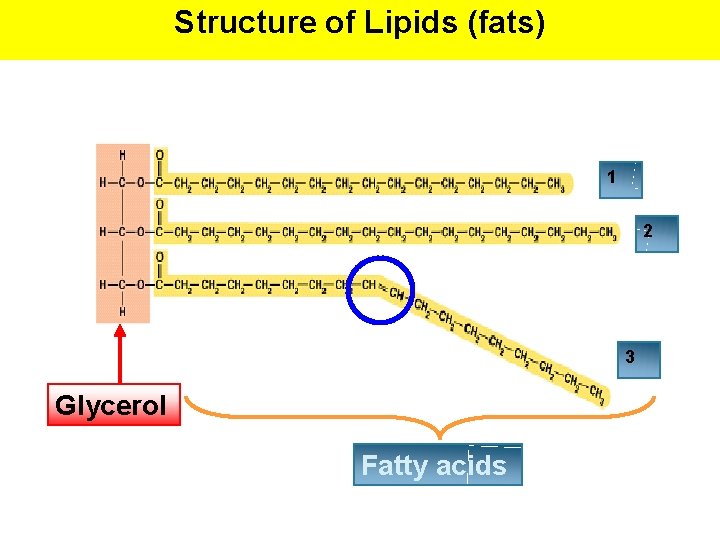
Structure of Lipids (fats) 1 2 3 Glycerol Fatty acids

1. Triglycerides Glycerol FA FA FA = TG (Triglyceride) Structure • Triglycerides are composed of 1 glycerol + 3 fatty acids (16 -18 Carbons long) Formation and Breakdown • Triglycerides are formed through a condensation/dehydration reaction between the glycerol and each of the fatty acids. • Triglycerides are broken down into 1 glycerol and 3 fatty acids through hydrolysis 2 Types of Fatty Acids 1. Saturated 2. Unsaturated

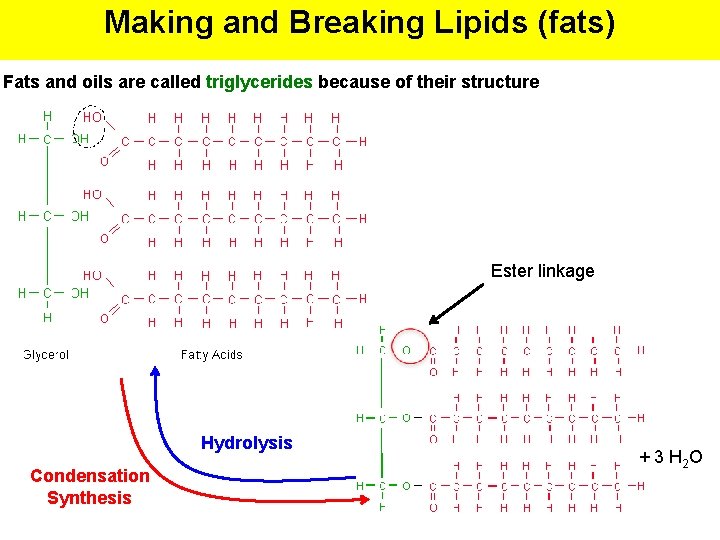
Making and Breaking Lipids (fats) Fats and oils are called triglycerides because of their structure Ester linkage Hydrolysis Condensation Synthesis + 3 H 2 O

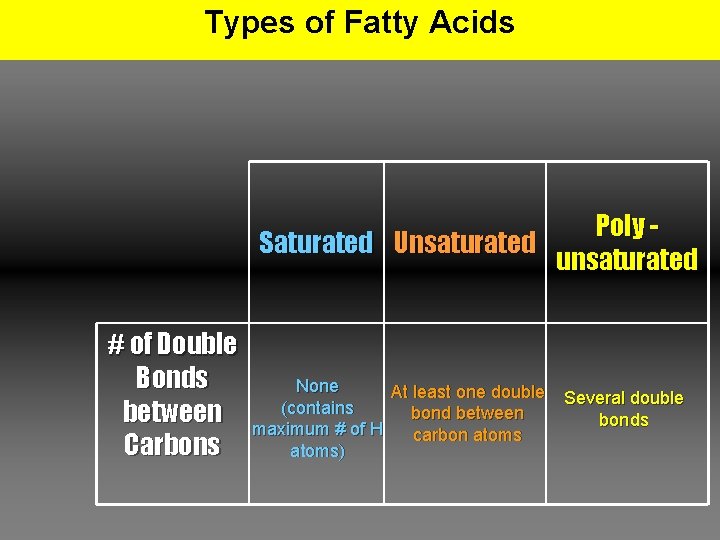
Types of Fatty Acids Poly Saturated Unsaturated unsaturated # of Double Bonds between Carbons None At least one double (contains bond between maximum # of H carbon atoms) Several double bonds

Types of Fatty Acids: Saturated Fats • Solid at room temperature • Contain only single C-C bonds (resulting in straight chains) • During digestion these are converted into “bad cholesterol” which can clog arteries • Ex. animal fats, butter

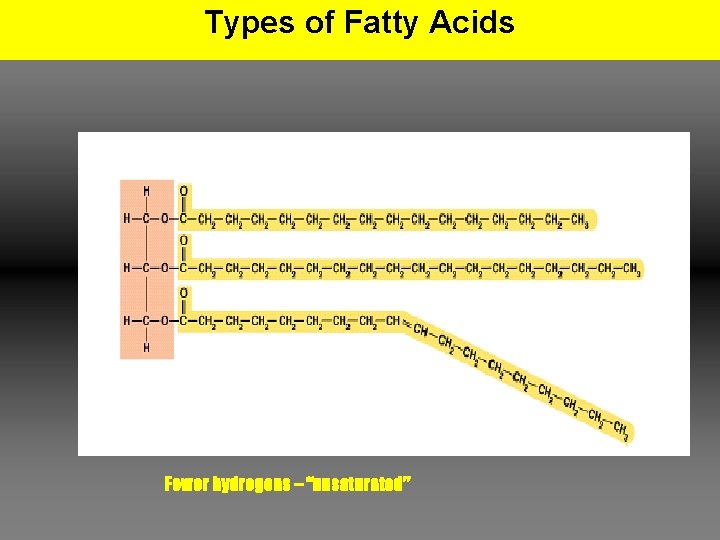
Types of Fatty Acids Fewer hydrogens – “unsaturated”

Types of Fatty Acids CH 2 -CH =C H BEND DUE TO DOUBLE BOND How do you think this bend affects density? ? ?

Types of Fatty Acids: Unsaturated Fats • Liquid at room temperature • Contain double C-C bonds (resulting in kinks in their chains) • Unsaturated fats with more than one double bond are called polyunsaturated fats • Ex. vegetable oils

Types of Fatty Acids Trans Fat Taking a perfectly good fat and making it bad! üUnsaturated fat (contains double bonded carbons) that has the properties of a saturated fat ü Often solid at room temperature ü Bad for you! Also causes an increase in “bad cholesterol”

How Trans Fats Became out Enemy… b Fill out the question sheet while you watch this short You. Tube clip b http: //www. youtube. com/watch? v=2 S 1 m. QY O 0 g 68

Phospholipids v Fat derivatives in which one fatty acid has been replaced by a phosphate group and one of several nitrogencontaining molecules. v an important part of the cell membrane (phospholipid bilayer)

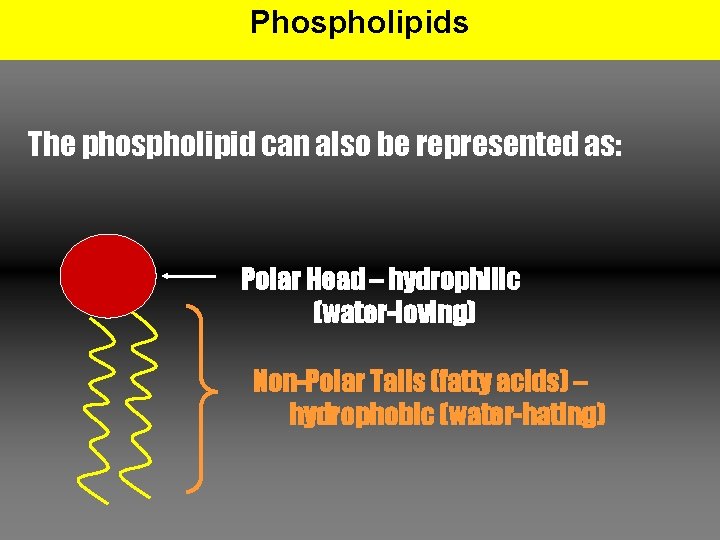
Phospholipids Structure: • Composed of glycerol, 2 fatty acids, and a phosphatecontaining functional group • Phosphate head is hydrophillic (water-loving) and the fatty acid tails are hydrophobic (water-hating) Why do you think the fatty acids are hydrophobic? Why is the phosphate portion hydrophillic?

Phospholipids The phospholipid can also be represented as: Polar Head – hydrophilic (water-loving) Non-Polar Tails (fatty acids) – hydrophobic (water-hating)

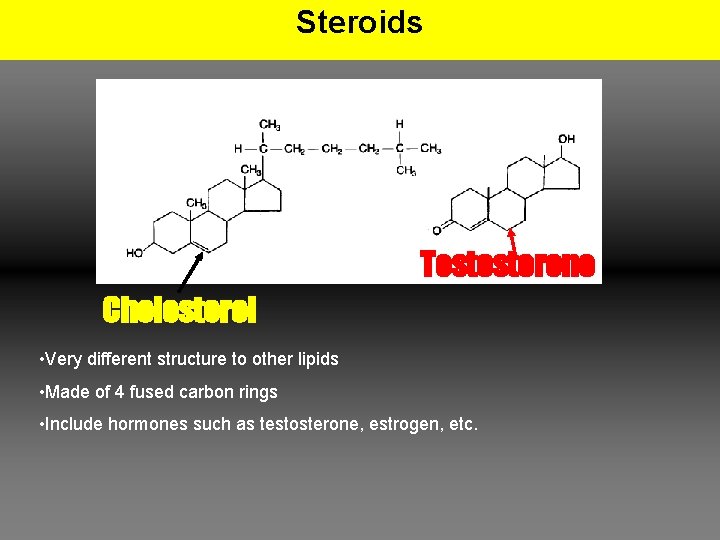
Steroids Testosterone Cholesterol • Very different structure to other lipids • Made of 4 fused carbon rings • Include hormones such as testosterone, estrogen, etc.