Lipids biochemistry Definition Lipids are hydrophobic or amphipathic

Lipids biochemistry

Definition

• Lipids are hydrophobic or amphipathic small molecules that may originate entirely or in part by carbanion-based condensations of thioesters (fatty acids, polyketides, etc. ) and/or by carbocation-based condensations of isoprene units (phenols, sterols, etc. ).


• The most common lipid classes in nature consist of fatty acids linked by an ester bond to the dihydric alcohol - glycerol, or to other alcohols such as cholesterol, or by amide bonds to sphingoid bases, or on occasion to other amines.

Functions of Lipids 1. Storage form of energy (triglycerides) 2. Structural components of bio-membranes phospholipids and cholesterol) 3. Metabolic regulators (steroid hormones and prostaglandins) 4. Act as surfactants, detergents and emulsifying agents (amphipathic lipids) 5. Act as electric insulators in neurons 6. Provide insulation against changes in external temperature (subcutaneous fat) 7. Give shape and contour to the body 8. Protect internal organs by providing a cushioning effect (pads of fat) 9. Help in absorption of fat soluble vitamins (A, D, E and K) 10. Improve taste and palatability to food.
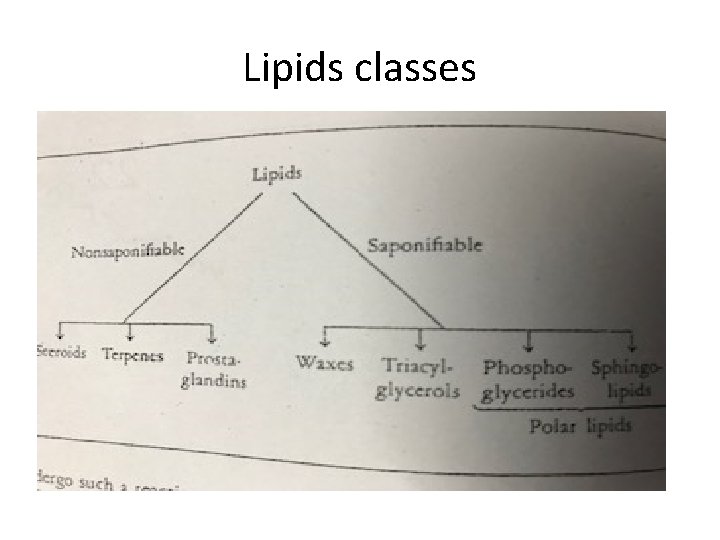
Lipids classes

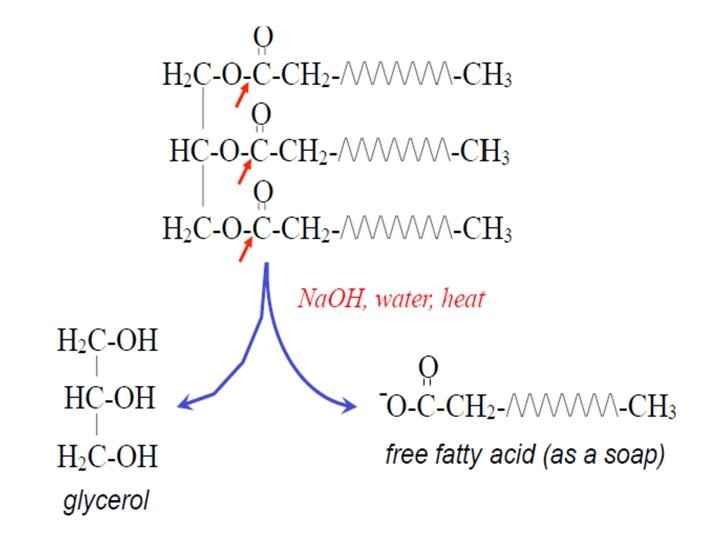
• Lipids that undergo alkaline hydrolysis are called saponifiable lipids. • Lipids that don’t undergo such reaction are called nonsaponifable lipids.

Saturated Vs. unsaturated fatty acids • saturated: the SFA’s of a lipid have no double bonds between carbons in chain • polyunsaturated: more than one double bond in the chain • most common polyunsaturated fats contain the polyunsaturated fatty acids (PUFAs) oleic, linoleic and linolenic acid • unsaturated fats have lower melting points • stearic (SFA) melts at 70 o. C, oleic (PUFA) at 26 o. C

Triacylglycerol • They consist of a glycerol moiety with each hydroxyl group esterified to a fatty acid. In nature, they are synthesised by enzyme systems, which determine that a center of asymmetry is created about carbon-2 of the glycerol backbone, so they exist in enantiomeric forms, i. e. with different fatty acids in each position.

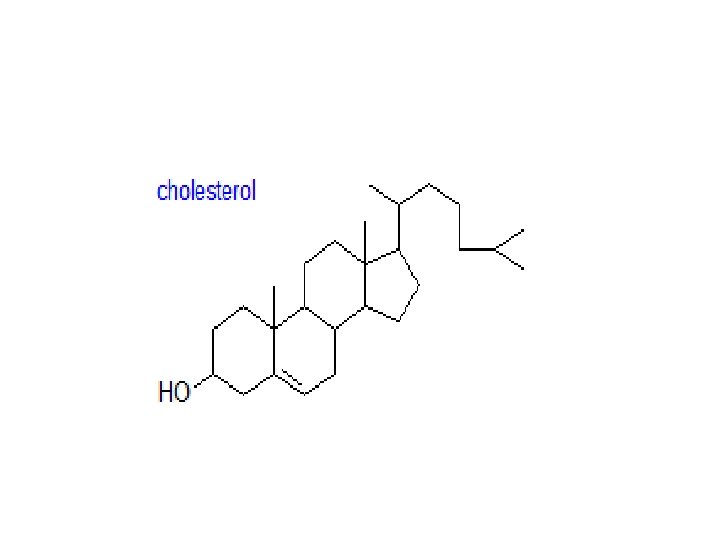
Cholesterol • Sterols and sterol esters: Cholesterol is by far the most common member of a group of steroids in animal tissues; it has a tetracyclic ring system with a double bond in one of the rings and one free hydroxyl group. It is found both in the free state, where it has an essential role in maintaining membrane fluidity, and in esterified form, i. e. as cholesterol esters. Other sterols are present in free and esterified form in animal tissues, but at trace levels only. Cholesterol is the precursor of the bile acids and steroidal hormones.

Waxes In their most common form, wax esters consist of fatty acids esterified to long-chain alcohols with similar chain-lengths. (even number of carbone 36 -34 ) The latter tend to be saturated or have one double bond only. Such compounds are found in animal, plant and microbial tissues and they have a variety of functions, such as acting as energy stores, waterproofing , lubrication, protect plant from lose moisture, harmful insects and protective coating.

• Carnauba wax obtain from plant its widely used as floor and automobile wax. • Sometimes the alcohol part of a wax is steroids , for example lanolin used in cosmetic, cream and ointments. • Waxes are soft and pliable when warm, yet hard when cold.

Phosphoglycerides phosphoglycerides are ester of glycerol. They contain two acyl groups. One of the primary hydroxyl groups of glycerol forms an ester with phosphoric acid. Which in turn joined by phosphate ester bonded to alcohol. Its characteristic important in cell membrane because of polar head (phosphate ) and non polar hydrocarbones tail.
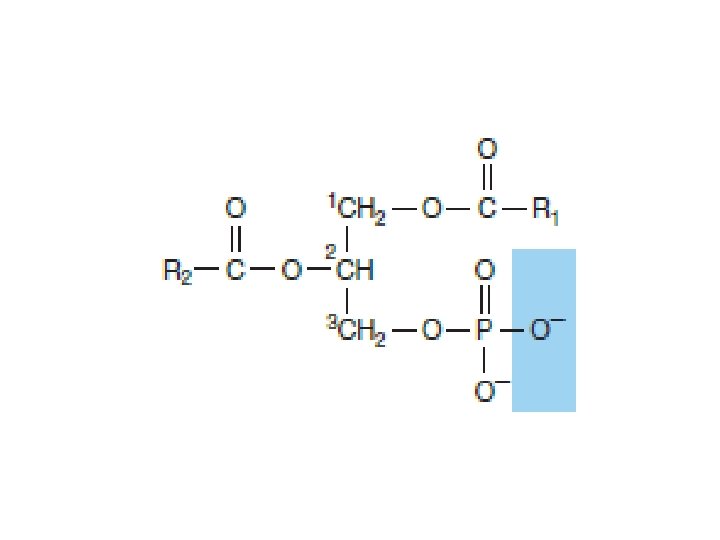

Fatty acids • The common fatty acids of plant and animal tissues. They are carboxylic acid in which long straight hydrocarbons chain in attached to carboxyl group. Fatty acids differ mainly in chain length and number and position of double bond in hydrocarbon chain. Fatty acids with one or more double bond are called unsaturated fatty acid while , with no double bond is called saturated fatty acids.
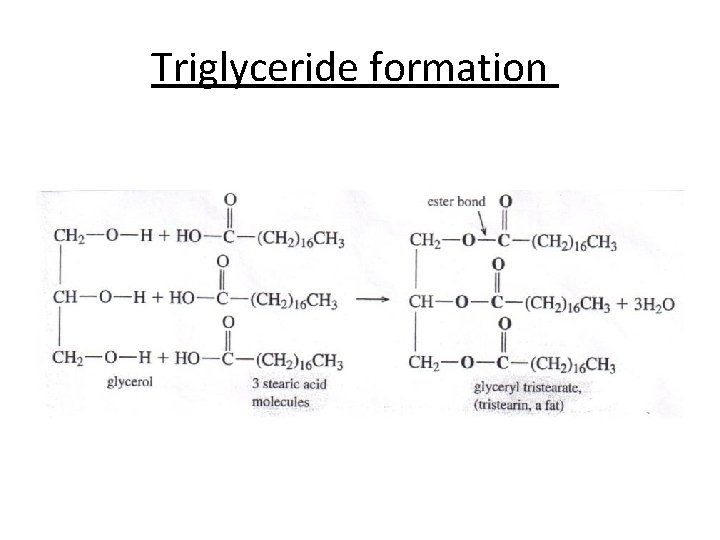
Triglyceride formation

Hexadecanoic palmitic 16: 0
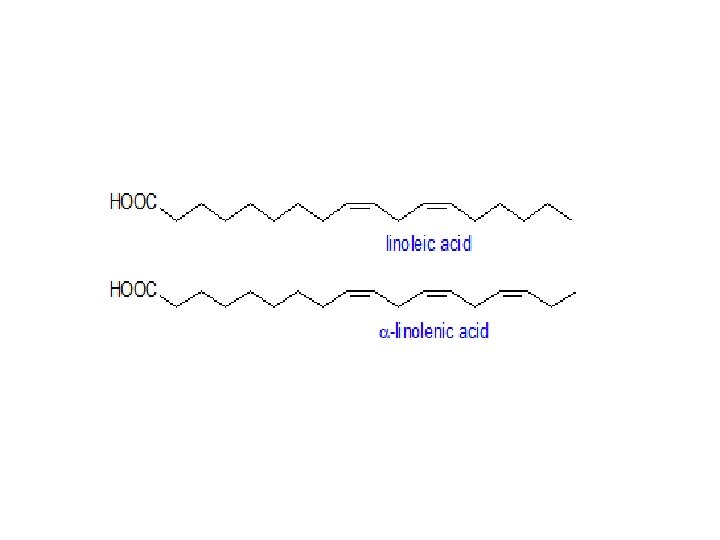

• Most fatty acids are straight chain compounds and have even number of carbon atoms between 14 -22, palmitic (C 16 H 32 O 2) and stearic (C 18 H 36 O 2) are the most common saturated fatty acids. • Mammals can made almost all fatty acids they need • The major exceptions are linolenic and linoleic Mammals required in diet they called essentials fatty acids they obtain them from plant

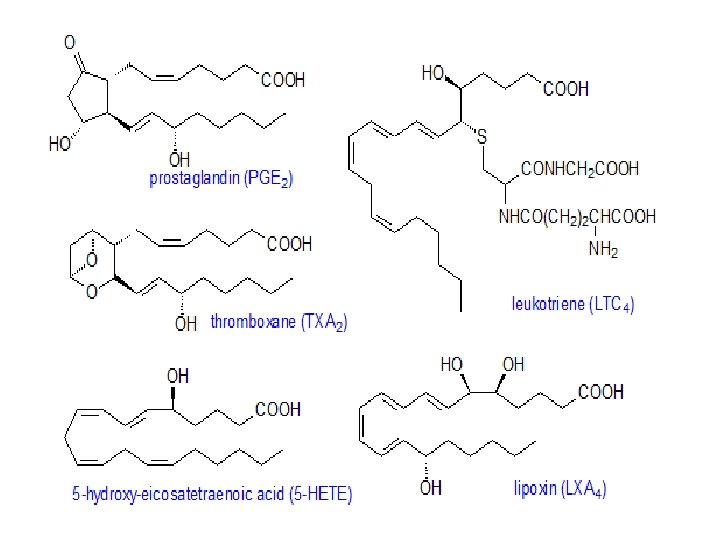
Eicosanoid and prostaglandins • The term eicosanoid is used to embrace biologically active lipid mediators (C 20 fatty acids and their metabolites), including prostaglandins, thromboxanes, leukotrienes and other oxygenated derivatives, which exert their effects at very low concentrations.
- Slides: 25