Lipids and Membranes What Are Lipid lipids are
Lipids and Membranes

What Are Lipid? lipids are a family of substances that are insoluble in water but soluble in nonpolar solvents and solvents of low polarity ( diethyl ether).

Lipid § Lipids: (Greek: lipos, fat) § § Are substances of biological origin Soluble in organic solvents such as chloroform and methanol § Sparingly soluble, if at all, in water. Fats, oils, § Certain vitamins and hormones, and most nonprotein membrane components are lipids.
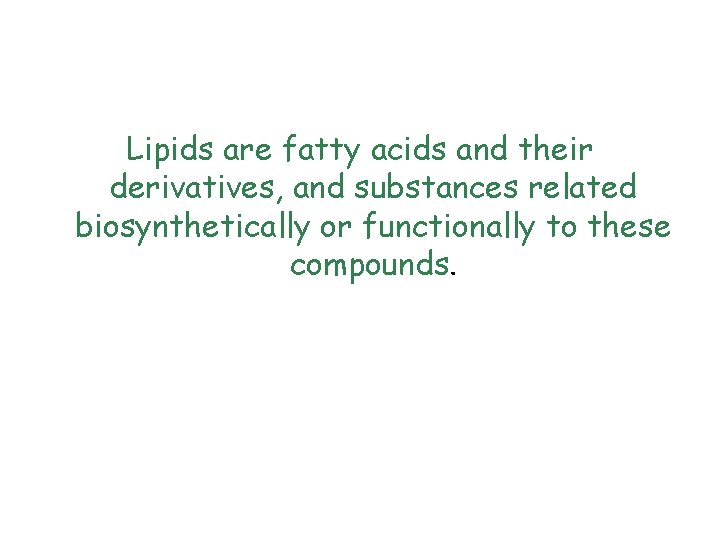
Lipids are fatty acids and their derivatives, and substances related biosynthetically or functionally to these compounds.

A. CLASSIFICATION BY FUNCTION Lipids play three major roles in human biochemistry (1) They store energy within fat cells (2) they are parts of membranes that separate compartments of aqueous solutions from each other. (3) they serve as chemical messengers.

I. Storage An important use for lipids especially in animals is the storage of energy. ● plants store energy in the form of starch. ● Animals (including humans) find it more economical to use fats instead. The reason is simple: The burning of fats produces more than twice as much energy (about 9 kcal/g) as the burning of an equal weight of carbohydrates (about 4 kcal/g).

II. Membrane Components The lack of water solubility of lipids is an important property because our body chemistry is so heavily based on water. Most body constituents, including carbohydrates and proteins, are soluble in water. However, the body also needs insoluble compounds for the membranes that separate compartments containing aqueous solutions, whether they are cells or organelles within the cells. Lipids provide these membranes. Their water insolubility derives from the fact that the polar groups they contain are much smaller than their alkane like (nonpolar) portions. These nonpolar portions provide the water repellent, or hydrophobic, property.

III. Messengers Primary messengers, such as steroid hormones, deliver signals from one part of the body to another part. Secondary messengers, such as prostaglandins and thromboxanes, mediate the hormonal response.

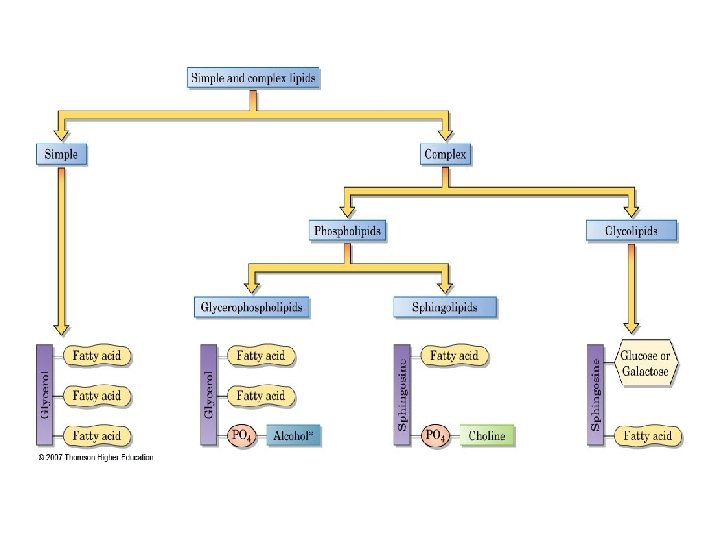
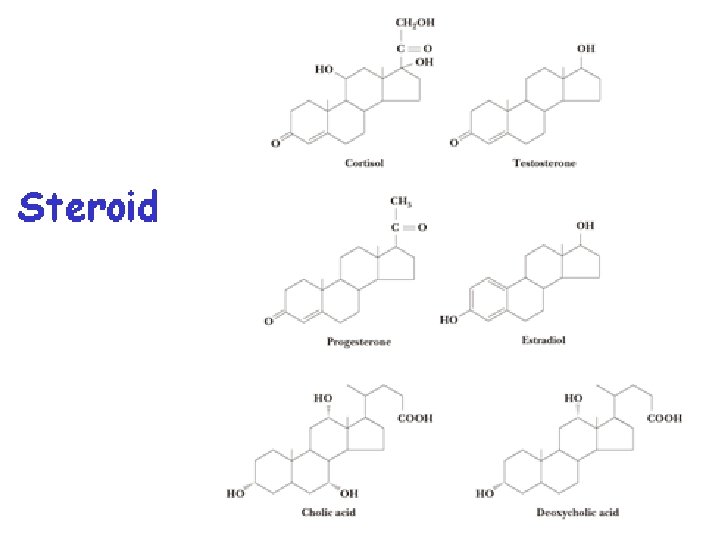
B. CLASSIFICATION BY STRUCTURE we can classify lipids into four groups: (1) simple lipids, such as fats and waxes; (2) complex lipids; (3) steroids; and (4) prostaglandins, thromboxanes, and leukotrienes.

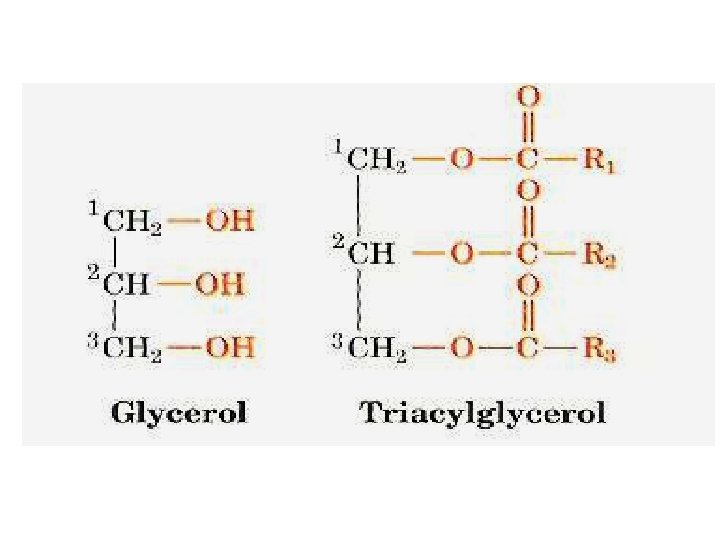
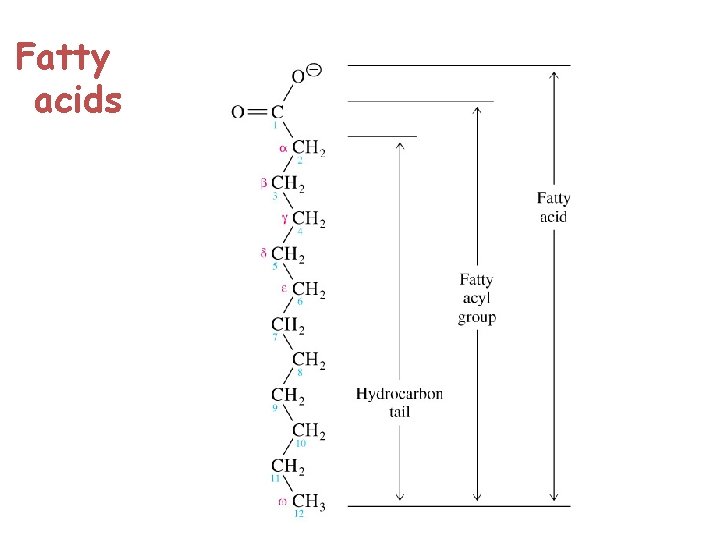
What Are the Structure of Triglycerides? Animal fats and plant oils are triglycerides. Triglycerides are triesters of glycerol and long chain carboxylic acids called fatty acids we saw that esters are made up of an alcohol part and an acid part.
Fatty acids
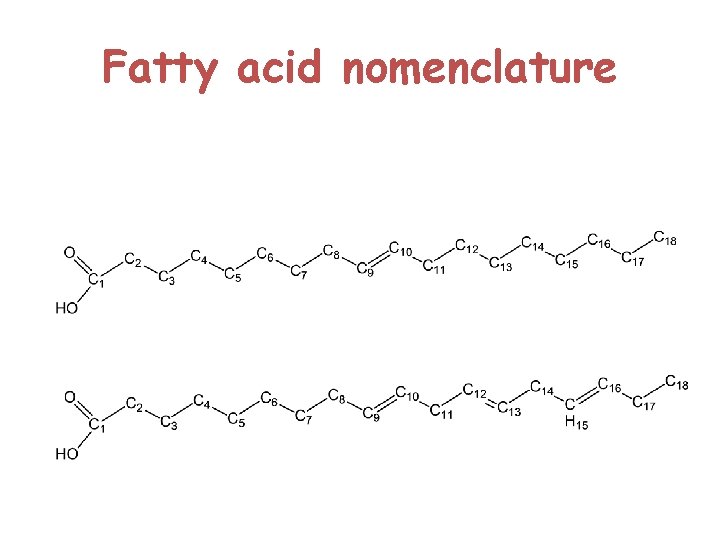
Fatty acid nomenclature
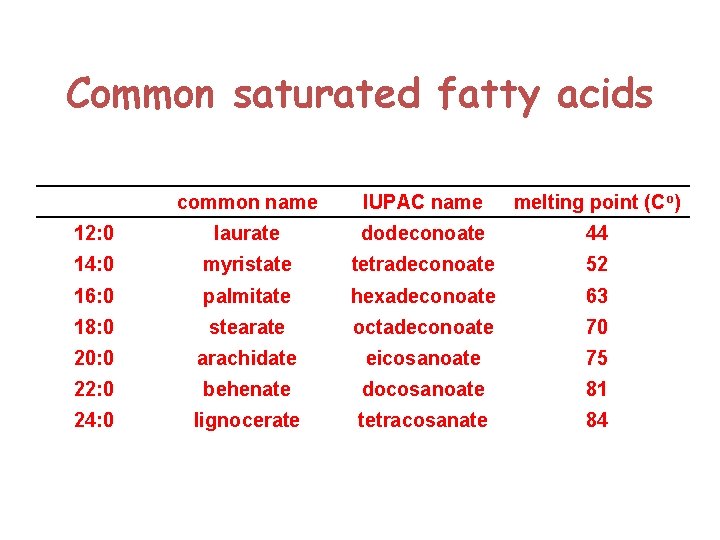
Common saturated fatty acids common name IUPAC name melting point (Co) 12: 0 laurate dodeconoate 44 14: 0 myristate tetradeconoate 52 16: 0 palmitate hexadeconoate 63 18: 0 stearate octadeconoate 70 20: 0 arachidate eicosanoate 75 22: 0 behenate docosanoate 81 24: 0 lignocerate tetracosanate 84

Common unsaturated fatty acids common name IUPAC name melting point (Co) 16: 0 palmitate hexadeconoate 63 16: 1 D 9 palmitoleate cis-D 9 -hexadeconoate -0. 5 18: 0 stearate octadeconoate 70 18: 1 D 9 oleate cis-D 9 - octadeconoate 13 18: 2 D 9, 12 linoleate cis-D 9, 12 - octadeconoate -9 18: 3 D 9, 12, 15 linolenate cis-D 9, 12, 15 - octadeconoate -17 20: 0 arachidate eicosanoate 75 20: 4 D 5, 8, 11, 14 arachindonate cis- D 5, 8, 11, 14 -eicosatetraenoate -49
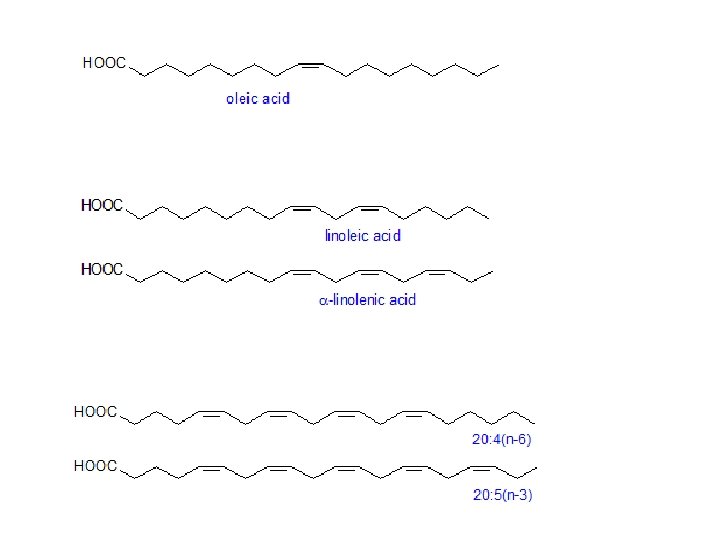

These fatty acids, have certain things in common: 1 Fatty acids are practically all unbranched carboxylic acids. 2 They range in size from about 10 to 20 carbons. 3 They contain an even number of carbon atoms. 4 Apart from the COOH group, they have no functional groups, except that some do have double bonds. 5 In most fatty acids that have double bonds, the cis isomers predominate.

q Only even numbered acids are found in triglycerides because the body builds these acids entirely from acetate units and therefore puts the carbons in two at a time. q In triglycerides (also called triacylglycerols), all three groups of glycerol are esterified.
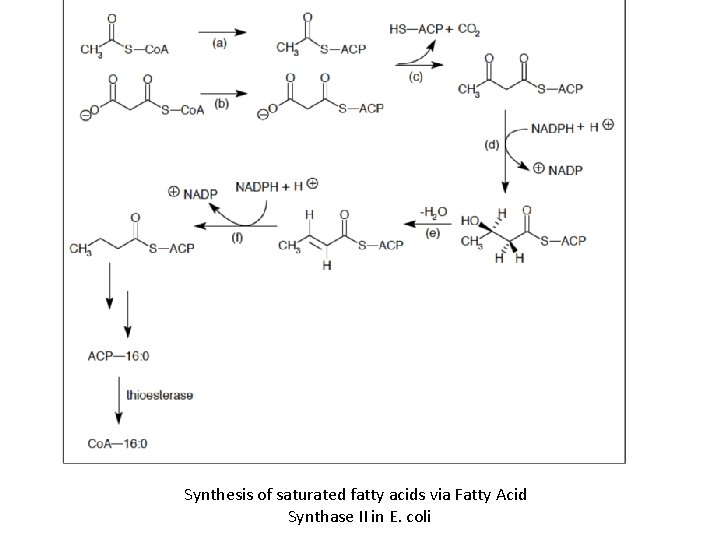
Synthesis of saturated fatty acids via Fatty Acid Synthase II in E. coli
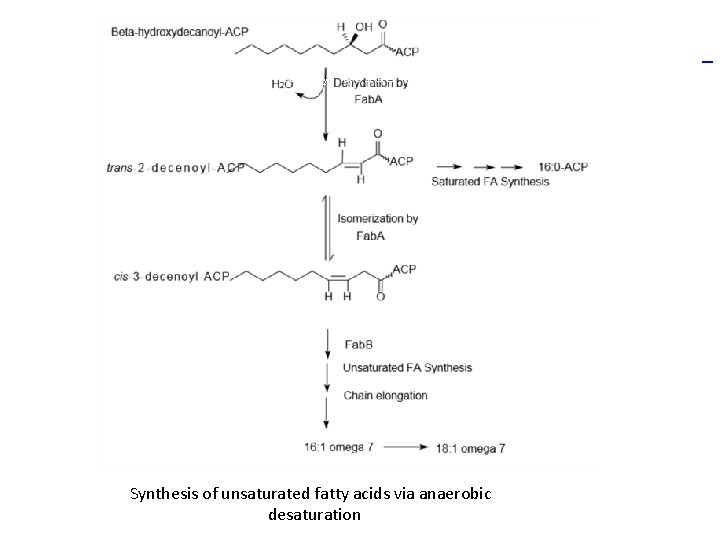
Synthesis of unsaturated fatty acids via anaerobic desaturation

• Triglycerides are the most common lipid materials, although mono and diglycerides are not infrequent. In the latter two types, only one or two OH groups of the glycerol are esterified by fatty acids. • Triglycerides are complex mixtures. Although some of the molecules contain three identical fatty acids, in most cases two or three different acids are present. • The hydrophobic character of triglycerides is caused by the long hydrocarbon chains. The ester groups ( C O ), although polar them selves, are buried in a nonpolar environment, which makes the triglycerides insoluble in water.

What Are Some Properties of Triglycerides? A. Physical State Fat A mixture of triglycerides containing a high proportion of long chain, saturated fatty acids oil A mixture of triglycerides containing a high proportion of long chain, unsaturated fatty acids or short chain, saturated fatty acids. Ø With some exceptions, fats that come from animals are generally ●solids at room temperature, ●liquids. from plants or fish. Ø Liquid fats are often called oils What is the structural difference between solid fats and liquid oils? In most cases, it is the degree of unsaturation.

• Solid animal fats contain mainly saturated fatty acids, Note that even solid fats contain some unsaturated acids • Vegetable oils contain high amounts of unsaturated fatty acids and that liquid fats contain some saturated acids. Table 21. 1 shows the average fatty acid content of some common fats and oils.
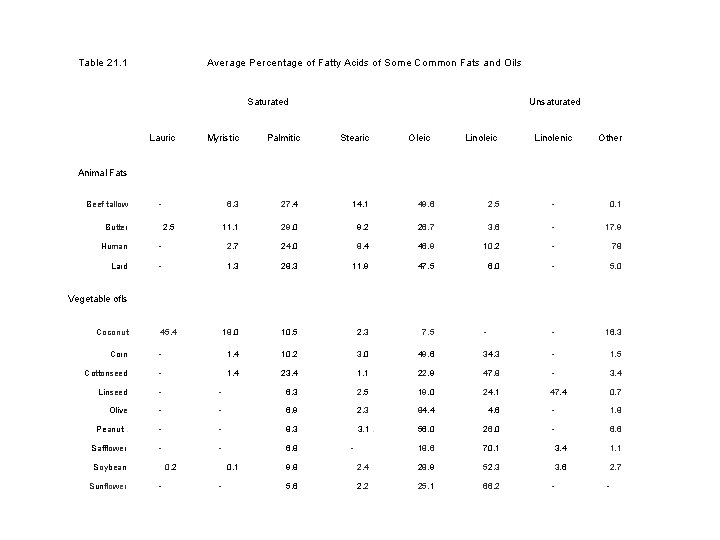
Table 21. 1 Average Percentage of Fatty Acids of Some Common Fats and Oils Saturated Lauric Unsaturated Myristic Palmitic Stearic Oleic Linolenic Other 6. 3 27. 4 14. 1 49. 6 2. 5 - 0. 1 11. 1 29. 0 9. 2 26. 7 3. 6 - 17. 9 Animal Fats Beef tallow - Butter 2. 5 Human - 2. 7 24. 0 8. 4 46. 9 10. 2 - 78 Lard - 1. 3 28. 3 11. 9 47. 5 6. 0 - 5. 0 18. 0 10. 5 2. 3 7. 5 - - 16. 3 Vegetable ofls Coconut 45. 4 Corn - 1. 4 10. 2 3. 0 49. 6 34. 3 - 1. 5 Cottonseed - 1. 4 23. 4 1. 1 22. 9 47. 8 - 3. 4 Linseed - - 6. 3 2. 5 19. 0 24. 1 47. 4 0. 7 Olive - - 6. 9 2. 3 84. 4 4. 6 - 1. 8 Peanut. - - 8. 3 3. 1. 56. 0 26. 0 - 6. 6 Safflower - - 6. 8 18. 6 70. 1 3. 4 1. 1 3. 6 2. 7 Soybean Sunflower 0. 2 - 0. 1 - - 9. 8 2. 4 28. 9 52. 3 5. 6 2. 2 25. 1 66. 2 - -

●Some unsaturated fatty acids (linoleic and linolenic acids) are called essential fatty acids because the body cannot synthesize them from precursors; they must, therefore, be consumed as part of the diet. ●Although most vegetable oils contain high amounts of unsaturated fatty acids, there are exceptions. q. Coconut oil, for example, has only a small amount of unsaturated acids. This oil is a liquid not because it contains many double bonds, but rather because it is rich in low molecular weight fatty acids (chiefly lauric acid). • Oils with an average of more than one double bond per fatty acid chain are called polyunsaturated.

Pure fats and oils are colorless, odorless, and tasteless. This statement may seem surprising because we all know the tastes and colors of such fats and oils as butter. and olive oil. The tastes, odors, and colors are caused by small amounts of other substances dissolved in the fat or oil

B. Hydrogenation. We learned that we can reduce carbon double bonds to single bonds by treating them with hydrogen and a catalyst. It is, therefore, not difficult to convert unsaturated liquid oils to solids.

◊ This hydrogenation is carried out on a large scale to produce the solid shortening solid in stores under such brand names as Crisco, Spry, and Dexo. ◊ In making such products, manufacturers must be careful not to hydrogenate all of the double bonds, because a fat with no double bonds would be too solid. Partial, but not complete, hydrogenation results in a product with just the right consistency for cooking. ü Margarine is also made by partial hydrogenation of vegetable oils. Because less hydrogen is used, margarine contains more unsaturation than fully hydrogenated shortenings.

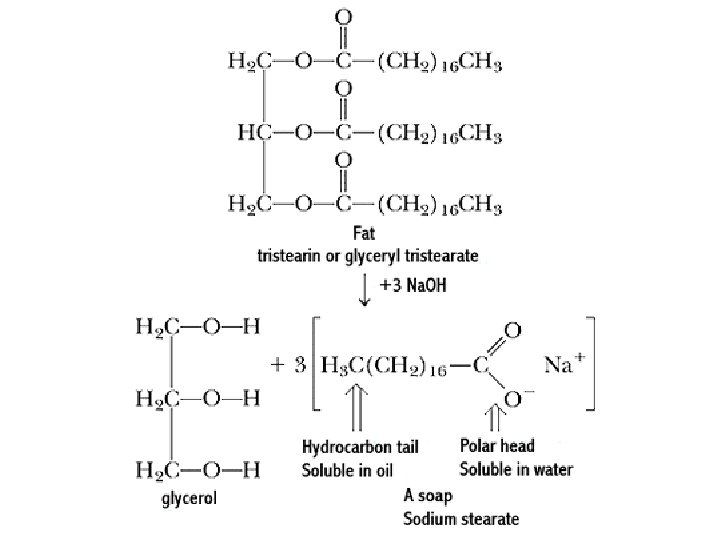
C. Saponification • Soap is the base promoted hydrolysis of fats and oils producing glycerol and a mixture of fatty acid salts • it has been used for thousands of years, and saponification is one of the oldest known chemical reactions

soaps are soluble sodium or potassium salts of carboxylic acids Hydrolysis of esters under basic conditions to form an alcohol and the salt of a carboxyli acid (carboxylates).


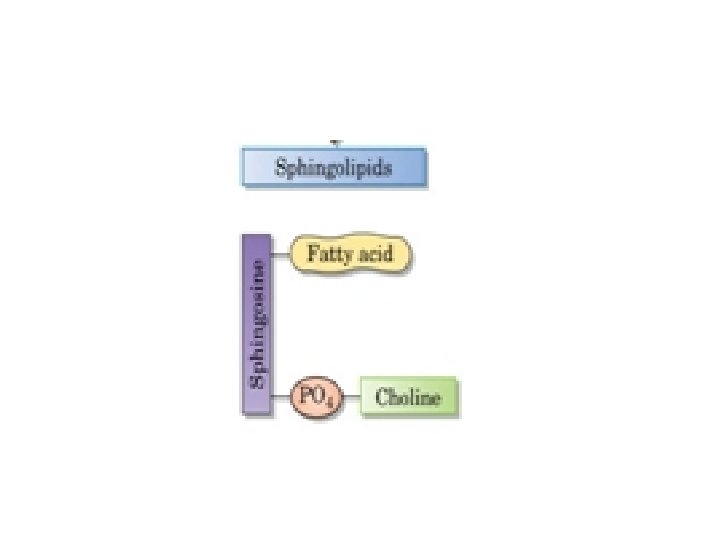
What Are the Structures of Complex Lipids? Complex lipids can be classified into two groups: . Ø Phospholipids contain an alcohol, two fatty acids, and a phosphate group. There are two types: o glycerophospholipids alcohol is glycerol o sphingolipids, the alcohol is sphingosine. Ø Glycolipids are complex lipids that contain carbohydrates.

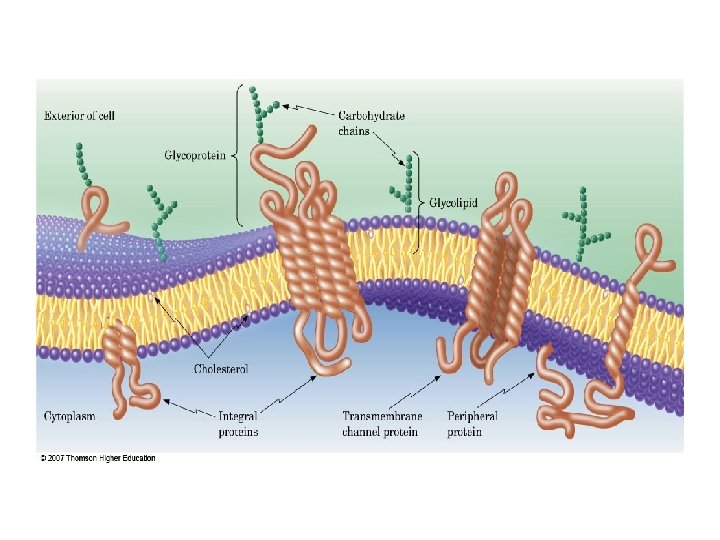
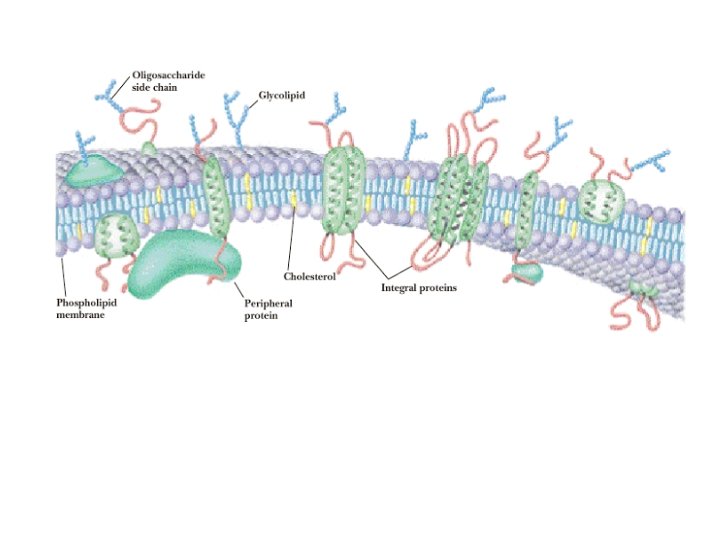
What Role Do Lipids Play in the Structure of Membrane? The complex lipids form the membranes around body cells and around small structures inside the cell's. (organelles. ) Unsaturated fatty acids are important components of these lipids. Most lipid molecules in the bilayer contain at least one unsaturated fatty acid. The cell membranes separate cells from the external environment and provide selective transport for nutrients and waste products into and out of cells.

• These membranes are made of lipid bilayers. • The hydrophobic tails point toward each other, which enables them to get as far away from the water as possible. • the hydrophilic heads projecting to the inner and outer surfaces of the membrane.

q. Cholesterol , another membrane component, Ø positions the hydrophilic portion of the molecule on the surface of the membranes and the hydrophobic portion inside the bilayer. Ø The unsaturated fatty acids prevent the tight packing of the hydrophobic chains in the lipid bilayer, thereby providing a liquid like character to the membranes. This property of membrane fluidity is of extreme importance because many products of the body's biochemical processes must cross the membrane, and the liquid nature of the lipid bilayer allows such transport.

q. Protein molecules , (in the lipid bilayer) Ø are either suspended on the surface (peripheral proteins) or Ø partly or fully embedded in the bilayer (integral proteins). Others are thoroughly embedded, going through the bilayer and projecting from both sides.

q. The model, fluid mosaic model of membranes, allows the passage of v nonpolar compounds by diffusion, as these compounds are soluble in the lipid membranes. v polar compounds are transported either via Ø specific channels through the protein regions or Ø active transport mechanism.

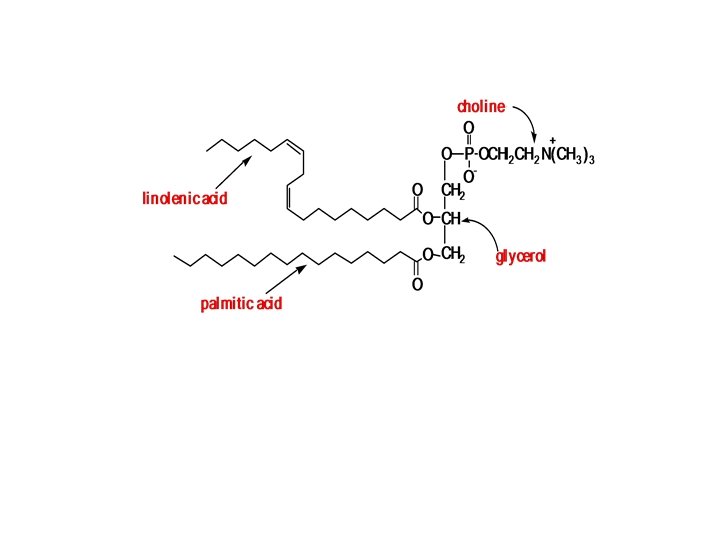
What Are Glycerophospholipids? The structure of glycerophospholipids (phosphoglycerides) is very similar to that of fats. q Glycerophospholipids are membrane components of cells throughout the body. q The alcohol is glycerol. q Two of the three groups are esterified by fatty acids, these fatty acids may be any long chain carboxylic cids, with or without double bonds. q In all glycerophospholipids, lecithins, cephalins, and phosphatidylinositols, the fatty acid on carbon 2 of glycerol is always unsaturated.
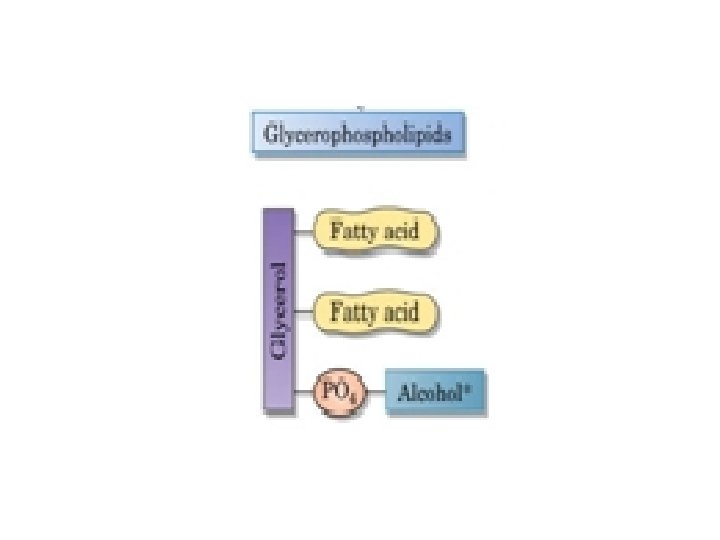

• The third group is esterified not by a fatty acid, but rather by a phosphate group, • which is also esterified to another alcohol. Ø If the other alcohol is choline , the glycerophospholipids are called phosphatidyl cholines (common name lecithin), Ø If the other alcohol is ethanolamine or serine are called cephalins, Ø If the alcohol inositol , is the phosphatidyli nositols.
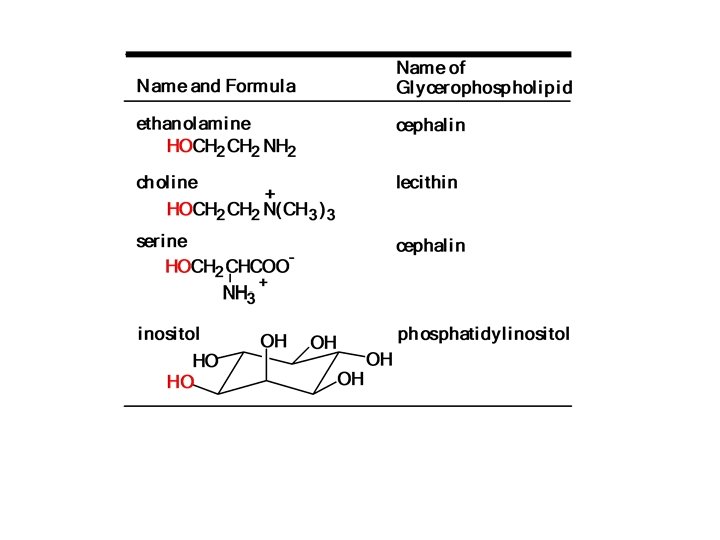

I - lecithin If the other alcohol is choline, a quaternary ammonium compound, the glycerophospholipids are called phosphatidyl cholines (common namelecithin): v Lecithin is a major component of egg yolk. Because it includes both polar and nonpolar portions within one molecule, it is an excellent emulsifier and is used in mayonnaise. v when a phospholipid such as lecithin is part of a lipid bilayer, the hydrophobic tail points toward the middle of the bilayer, and v the hydrophilic heads line both the inner and outer surfaces of the membranes

II - Cephalins, which are similar to the lecithins in every way except that, instead of choline they contain other alcohols, . such as ethanolamine or serine III - glycerophospholipids (phosphatidyli nositols (PI)). In PI, the alcohol inositol is bonded to the rest of the molecule by a phosphate ester bond. • Such compounds are integral structural parts of the biological membranes, and, in their higher phosphorylated form, such as phosphatidylinositol 4, 5 -bisphosphates (PIP 2), serve as signaling molecules in chemical communication

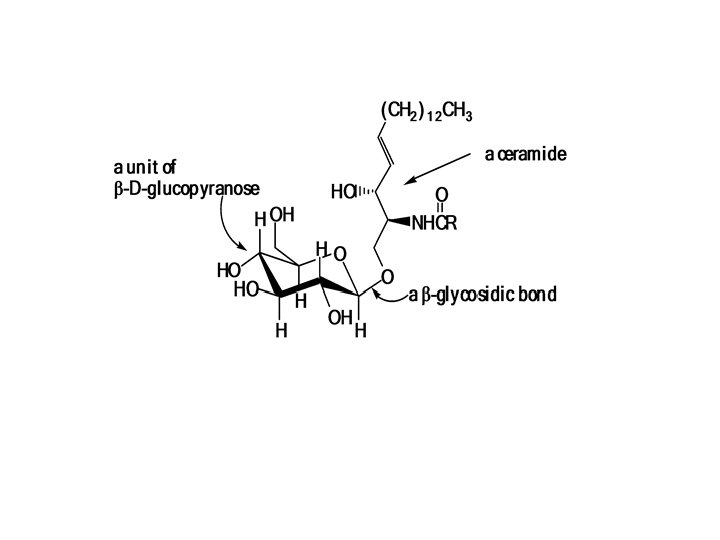
What Are Sphingolipids? Myelin, the coating of nerve axons, contains a different kind of complex lipid In sphingolipids, the alcohol portion is sphingosine The combination of a fatty acid and sphingosine is called the ceramide portion of the molecule The ceramide part of complex lipids may contain different fatty acids. Stearic acid, for example, occurs mainly in sphingomyelin.
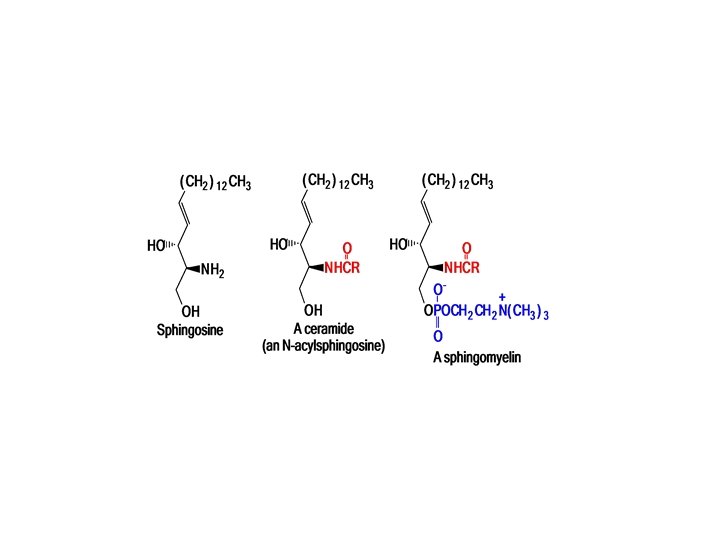

• A long chain fatty acid is connected to the NH 2 group by an amide bond, and the OH group at the end of the chain is esterified by phosphorylcholine:

Sphingomyelins are the most important lipids in the myelin sheaths of nerve cells and are associated with diseases such as multiple sclerosis. Sphingolipids are not randomly distributed in membranes. Johann Thudichum, who discovered sphingolipids in 1874, Sphingolipids appeared to Thudichum as part of a dangerous riddle of the brain.

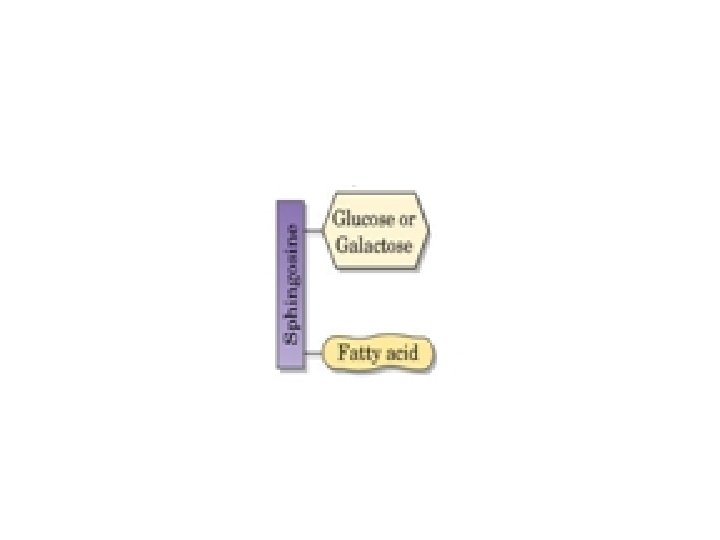
What are Glycolipids? Glycolipids are complex lipids that contain carbohydrates and ceramides. the cerebrosides, consists of ceramide mono or oligosaccharides. tthe gangliosides, contain a more complex carobohydrate structure. A glucose or galactose carbohydrate unit forms a beta glycosidic bond with the ceramide portion of the molecule. The cerebrosides occur primarily in the brain (accounting for 7% of the brain's dry weight) and at nerve synapses.

What Are Steroids? They are compounds containing the following ring system:

In this structure, three cyclohexane rings (A, B, and C); a fused cyclopentane ring (D). v Steroids are thus completely different in structure from the lipids already discussed. v Note that they are not necessarily esters, although some of them are.

A. Cholesterol The most abundant steroid in the human body, and the most important Cholesterol serves as: q a plasma membrane component in all animal cells ( in red blood cells). q. Its second important function is to serve as a raw material for the synthesis of other steroids, such as the sex and adrenocorticoid hormones and bile salts.
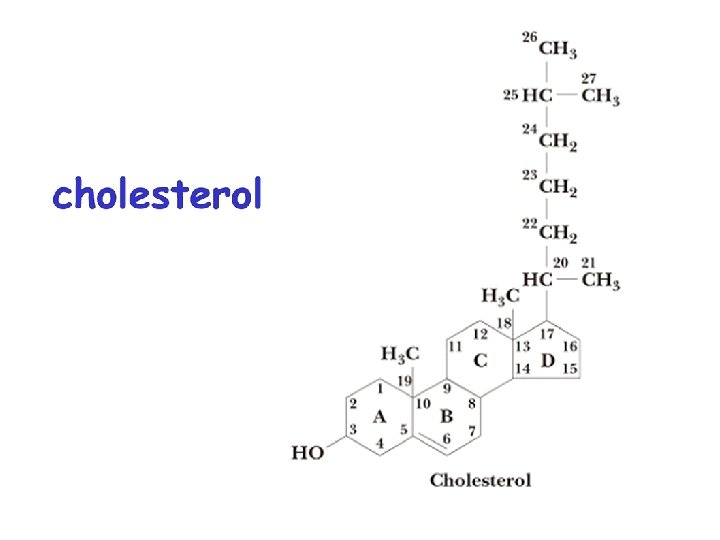

Cholesterol exists both in the free form (Gallstones) and esterified with fatty acids. When the cholesterol level exceeds 150 mg/l 00 m. L, cholesterol synthesis in the liver is reduced to half the normal rate of production.

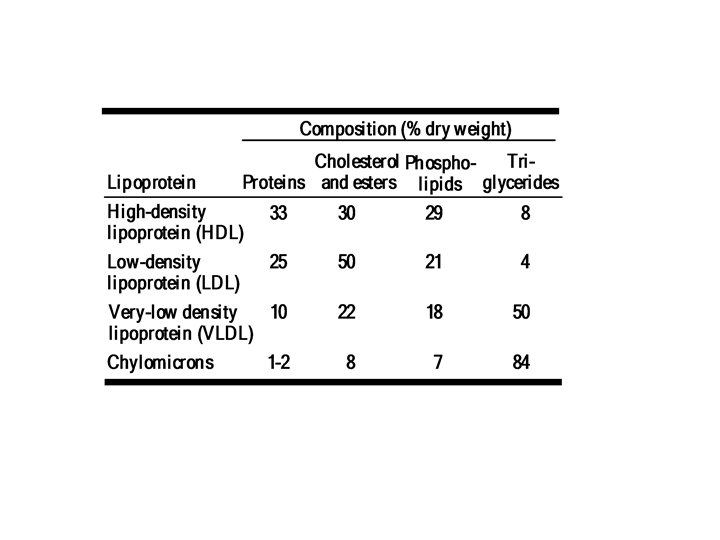
B. Lipoproteins: Carriers of Cholesterol • Cholesterol, along with fat, is 'transported by lipoproteins. Most lipoproteins contain a core of hydrophobic lipid molecules surrounded by a shell of hydrophilic molecules such as proteins and phospholipids There are four kinds of lipoproteins: v High density lipoprotein (HDL) ("good cholesterol"), which consists of about 33% protein and about 30% cholesterol v Low density lipoprotein (LDL) ("bad cholesterol"), which contains only 25% protein but 50% cholesterol v Very Iow density lipoprotein (VLDL), which mostly carries triglycerides (fats) synthesized by the liver v Chylomicrons, which carry dietary lipids synthesized in the intestines
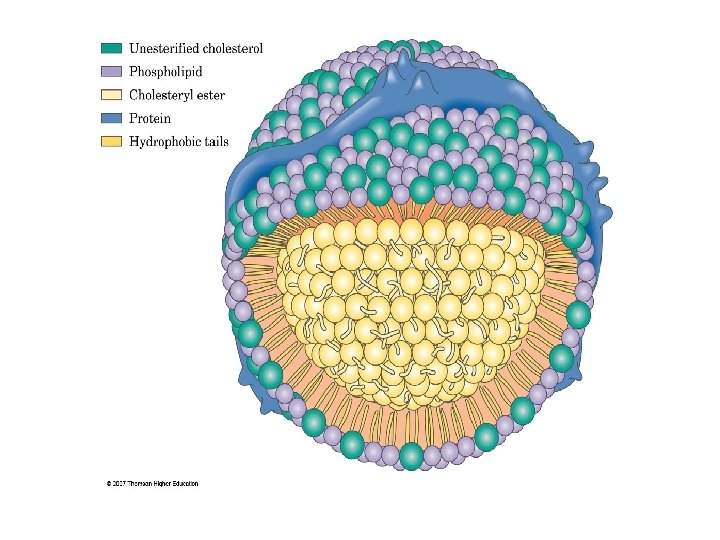
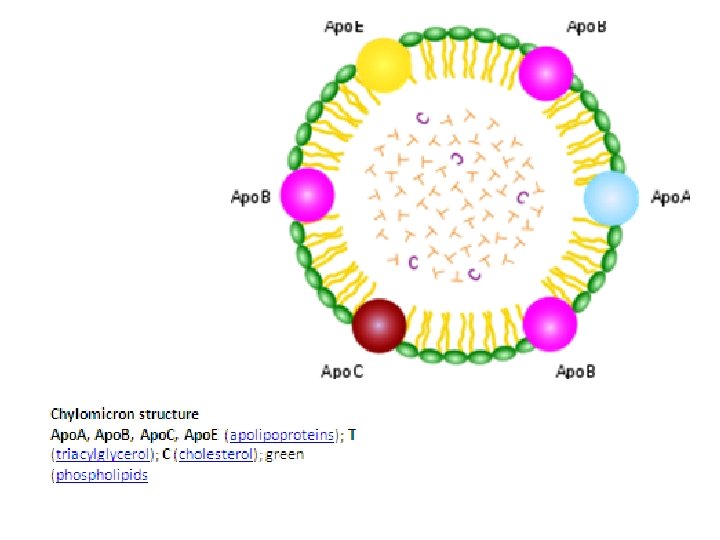

C. Transport of Cholesterol in LDL Ttransport of cholesterol from the liver starts out as a large VLDL (55 nanometers in diameter). 1. The core of VLDL contains triglycerides cholesteryl esters, (cholesteryl linoleate). It is surrounded by a polar coat of phospholipids and proteins. 2. The VLDL is carried in the serum. When the capillaries reach muscle or fat tissues, the triglycerides and all proteins except apo. B l 00 are removed from the VLDL. 3. At this point, the diameter of the lipoprotein shrinks to 22 nanometers and its core contains only cholesteryl esters. 4. Because of the removal of fat, its density increases and it becomes LDL.

5. The LDL carries cholesterol to the cells, where specific LDL receptor molecules line the cell surface in certain concentrated areas called coated pits. 6. The apo. B l 00 protein on the surface of the LDL binds specifically to the LDL receptor molecules in the coated pits. 7. After such binding, the LDL is taken inside the cell (endocytosis), where enzymes break down the lipoprotein. 8. In the process, they liberate free cholesterol from cholesteryl esters. In this manner, the cell can, for example, use cholesterol as a component of a membrane.

• Michael Brown and Joseph Goldstein of the University of Texas shared the Nobel Prize in medicine in 1986 for the discovery of the LDL receptor mediated pathway. If the LDL receptors are not sufficient in number, cholesterol accumulates in the blood; this accumulation can happen even with low intake of dietary cholesterol. Both genetics and diet play a role in determining cholesterol levels in the blood.

D. Transport of Cholesterol in HDL While in the serum, the free cholesterols in HDL are converted to cholesteryl esters. These esterified cholesterols are delivered to the liver for synthesis of bile acids and steroid hormones.

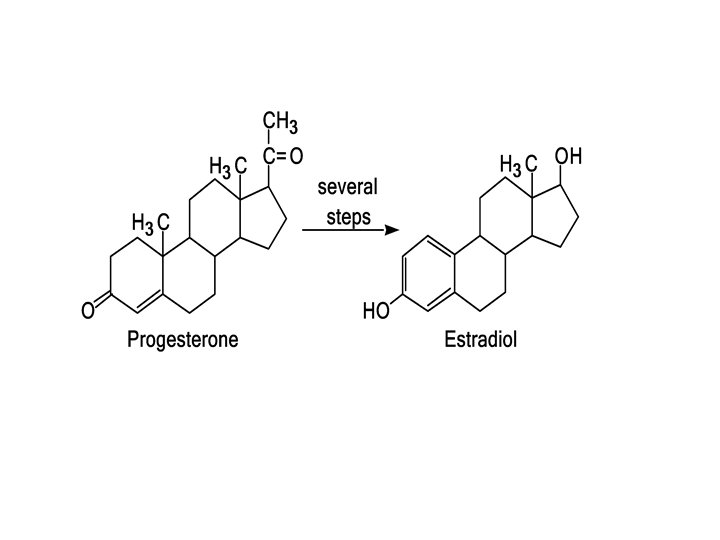
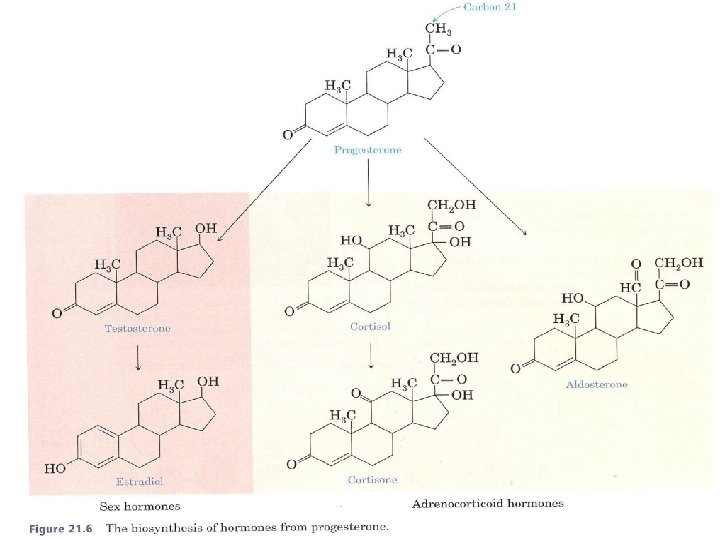
What Are Some of the Physiological Roles of Steroid Hormones? Cholesterol is the starting material for the synthesis of steroid hormones. In this process, the aliphatic side chain on the D ring is shortened by the removal of a six carbon unit, and the secondary alcohol group on carbon 3 is oxidized to a ketone. The resulting molecule, progesterone, serves as the starting compound for both the sex hormones and the adrenocorticoid hormones.
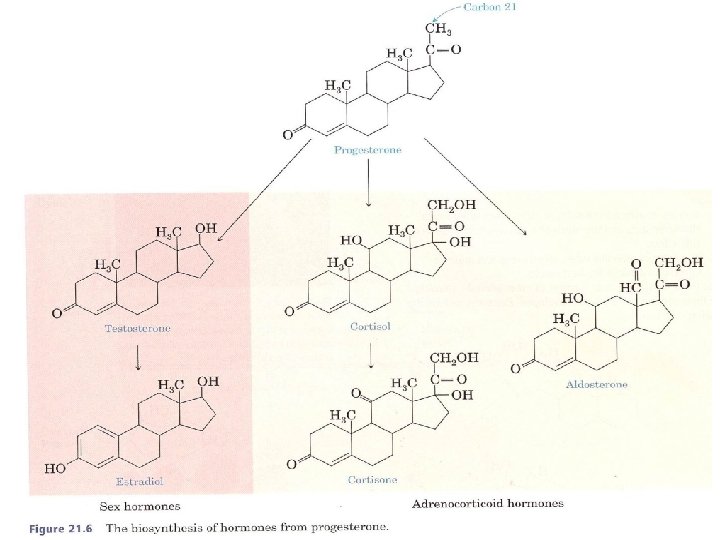

A. Adrenocorticoid Hormones The adrenocorticoid hormones are products of the adrenal glands. The term adrenal means "adjacent to the renal" (which refers to the kidney).

We classify these hormones into two groups according to function: 1 -Mineralocorticoids regulate the concentrations of ions (mainly Na+ and K+), q Aldosterone is one of the most important mineralocorticoids. Increased secretion of aldosterone enhances the reabsorption of N a + and Cl ions in the kidney tubules and Increases the loss of K+. Because Na + concentration controls water retention in the tissues, aldosterone controls tissue swelling.

2 - glucocorticoids control carbohydrate metabolism. q Cortisol is the major glucocorticoid. Its function is to increase the glucose and glycogen concentrations in the body. This accumulation occurs at the expense of other nutrients. Fatty acids and amino acids from body proteins are transported to the liver, which, under the influence of cortisol, manufactures glucose and glycogen from these sources. Cortisol and its ketone derivative, cortisone, have remarkable anti inflammatory effects. These or similar synthetic derivatives, such as prednisolone, are used to treat inflammatory diseases of many organs, rheumatoid arthritis, and bronchial asthma.

B. Sex Hormones The most important male sex hormone is testosterone. This hormone, which promotes the normal growth of the male genital organs, is synthesized in the testes from cholesterol. During puberty, increased testosterone production leads to such secondary male sexual characteristics as deep voice and facial and body hair.

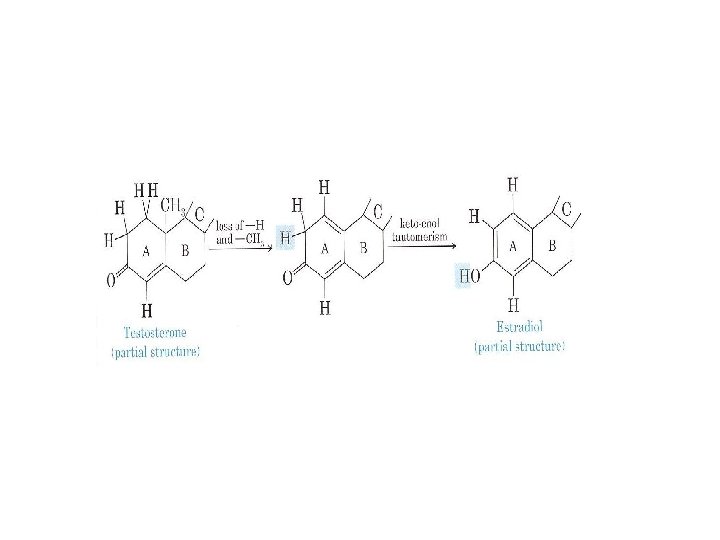
Female sex hormones, the most important of which is estradiol, are synthesized from the corresponding male hormone (testosterone) by aromatization of the A ring:

Estradiol, together with its precursor progesterone, regulates the cyclic changes occurring in the uterus and ovaries known as the menstrual cycle. Ø As the cycle begins, the level of estradiol in the body rises, which in turn causes the lining of the uterus to thicken. Another hormone, called luteinizing hormone (LH), then triggers ovulation. Ø If the ovum is fertilized, increased progesterone levels will inhibit any further ovulation. Both estradiol and progesterone promote further preparation of the uterine lining to receive the fertilized ovum. Ø If no fertilization takes place, progesterone production stops altogether, and estradiol production decreases. This halt decreases the thickening of the uterine lining, which is sloughed off with accompanying bleeding during menstruation
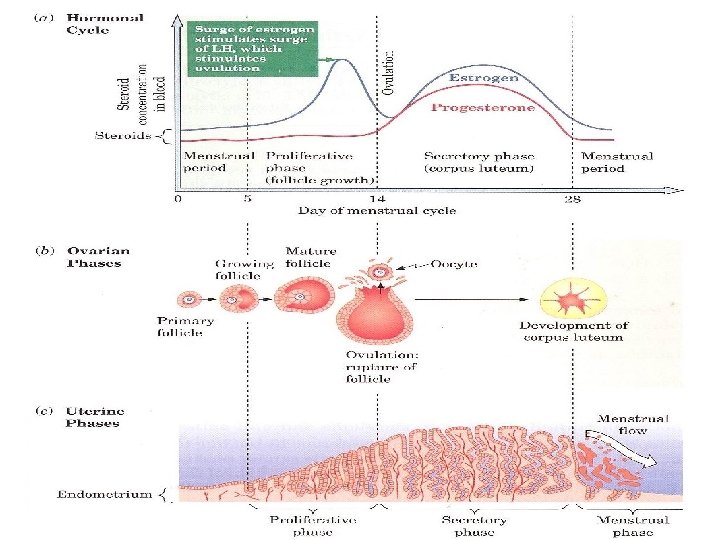

Because progesterone is essential for the implantation of the fertilized ovum, § Progesterone interacts with a receptor in the nucleus of cells. § The receptor changes its shape when progesterone binds to it its action leads to termination of pregnancy

What Are Bile Salts? Bile salts are oxidation products of cholesterol. First the cholesterol is oxidized to the trihydroxy derivative, and the end of the aliphatic chain is oxidized to the carboxylic acid. The latter, in turn, forms an amide bond with an amino acid, either glycine or taurine:
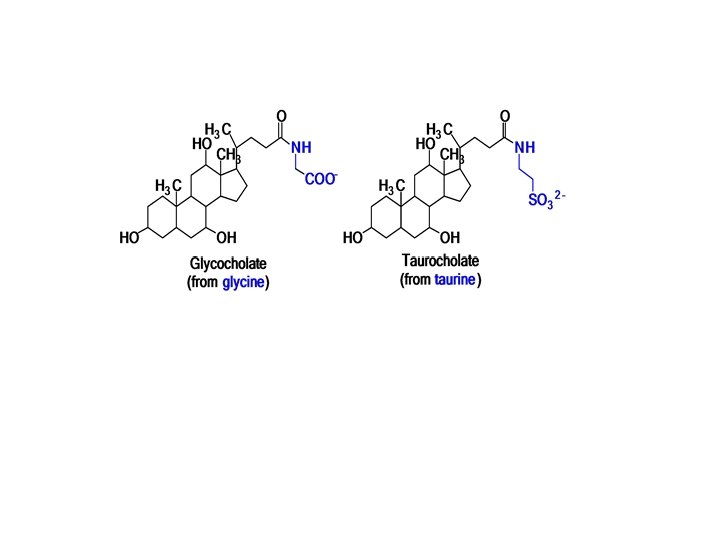

Taurine has developed a certain amount of commercial importance in recent years as an ingredient in sports drinks. The drink marketed under the trade name Red Bull contains various sugars , caffeine, and B vitamins in addition to taurine,

Bile salts are powerful detergents. One end of the molecule is strongly hydrophilic because of the negative charge, and the rest of the molecule is largely hydrophobic. As a consequence, bile salts can disperse dietary lipids in the small intestine into fine emulsions, thereby facilitating digestion. The dispersion of dietary lipids by bile salts is similar to the action of soap on dirt.

Because they are eliminated in the feces, bile salts remove excess cholesterol in two ways: (1) They are themselves breakdown products of cholesterol (so cholesterol is eliminated via bile salts), and (2) they solubilize deposited cholesterol in the form of bile salt cholesterol particles.


- Slides: 92