Lipids and Membranes Lipid Lipids Greek lipos fat

Lipids and Membranes

Lipid § Lipids: (Greek: lipos, fat) § Are substances of biological origin § Soluble in organic solvents such as chloroform and methanol. § Sparingly soluble, if at all, in water. Fats, oils, § Certain vitamins and hormones, and most nonprotein membrane components are lipids.

FUNCTIONS OF LIPIDS Lipids play three major roles in human biochemistry (1) They store energy within fat cells (2) They are parts of membranes that separate compartments of aqueous solutions from each other. (3) They serve as chemical messengers such as steroid hormones

Membrane Components The lack of water solubility of lipids is an important property because our body chemistry is so heavily based on water. Most body constituents, including carbohydrates and proteins, are soluble in water. However, the body also needs insoluble compounds for the membranes that separate compartments containing aqueous solutions, whether they are cells or organelles within the cells. Lipids provide these membranes. Their water insolubility derives from the fact that the polar groups they contain are much smaller than their alkane like (nonpolar) portions. These nonpolar portions provide the water repellent, or hydrophobic, property.

Biological membranes § Are organized assemblies of lipids and proteins with small amounts of carbohydrate. § They are permeable barriers to the passage of materials § They regulate the composition of the intracellular medium by controlling the flow of nutrients, waste products, ions, etc. , into and out of the cell. § They do this through membrane-embedded “pumps” and “gates” that transport specific substances against an electrochemical gradient or permit their passage with such a gradient.

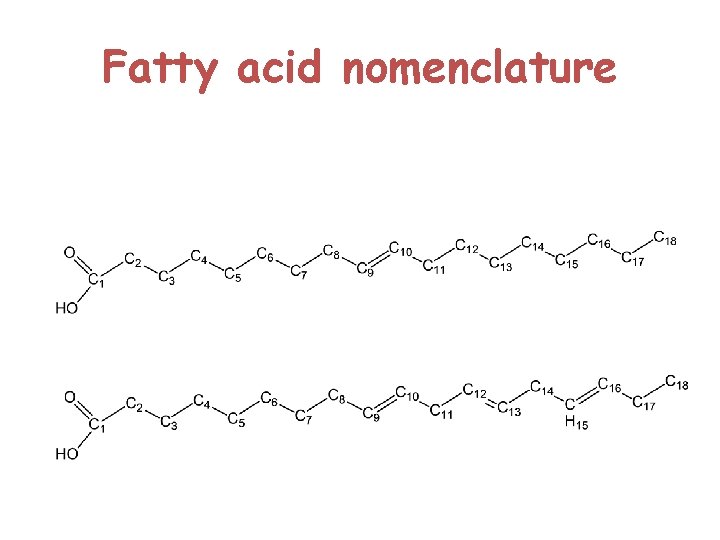
Structure of lipids Animal fats and plant oils are triglycerides (Triacylglycerol ) Triglycerides are triesters of glycerol and long chain carboxylic acids called fatty acids


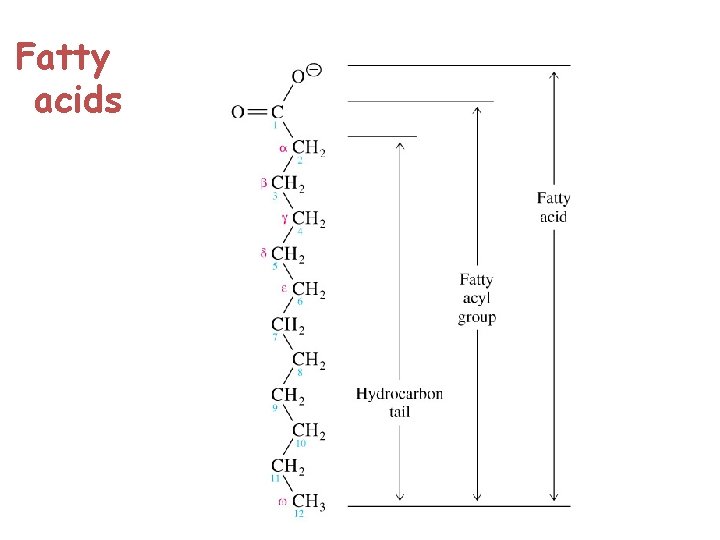
Fatty Acids § Are carboxylic acids with long-chain hydrocarbon side groups § They are rarely free in nature § Occur in esterified form as the major components of the various lipids § In higher plant and animals, the predominant fatty acid residues are those of the C 16 and C 18 species palmitic, oleic, linoleic, and steric acids.

§ Fatty acids with <14 or >20 carbon atoms are uncommon. § Most fatty acids have an even number of carbon atoms because they are usually biosynthesized by the condensation of C 2 units (acetate ). § Over half of the fatty acid residues of plant and animal lipids are § Unsaturated (contain double bonds) and § Are often polyunsaturated (contain two or more double bonds)
Fatty acids
Fatty acid nomenclature

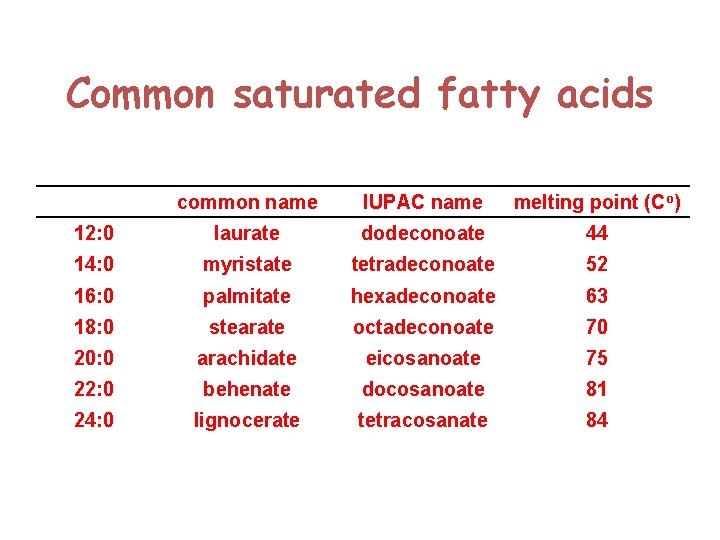
Common saturated fatty acids common name IUPAC name melting point (Co) 12: 0 laurate dodeconoate 44 14: 0 myristate tetradeconoate 52 16: 0 palmitate hexadeconoate 63 18: 0 stearate octadeconoate 70 20: 0 arachidate eicosanoate 75 22: 0 behenate docosanoate 81 24: 0 lignocerate tetracosanate 84

Common unsaturated fatty acids common name IUPAC name melting point (Co) 16: 0 palmitate hexadeconoate 63 16: 1 D 9 palmitoleate cis-D 9 -hexadeconoate -0. 5 18: 0 stearate octadeconoate 70 18: 1 D 9 oleate cis-D 9 - octadeconoate 13 18: 2 D 9, 12 linoleate cis-D 9, 12 - octadeconoate -9 18: 3 D 9, 12, 15 linolenate cis-D 9, 12, 15 - octadeconoate -17 20: 0 arachidate eicosanoate 75 20: 4 D 5, 8, 11, 14 arachindonate cis- D 5, 8, 11, 14 -eicosatetraenoate -49

Figure 6. 1 Structural formula of some C 18 fatty acids. The double bonds all have the cis conformation.

The Physical Properties of fatty acids vary with their degree of unsaturation Ø The first double bond of an unsaturated fatty acid commonly occurs between its C 9 and C 10 atoms counting from the carboxyl C atom (a Δ 9 - or 9 -double bond). Ø In polyunsaturated fatty acids, the double bonds tend to occur at every third carbon atom toward the methyl terminus of the molecule (such as –CH=CH–CH 2– CH=CH–). Ø Double bonds in polyunsaturated fatty acids are almost never conjugated (as in –CH=CH–).

Ø Triple bonds rarely occur in fatty acids or any other compound of biological origin. Ø Two important classes of polyunsaturated fatty acids are denoted n– 3 (or ω– 3) and n-6 (or ω– 6) fatty acids. Ø This nomenclature identifies the last doublebonded carbon atom as counted from the methyl terminal (ω) end of the chain.

Ø Fatty acid double bonds almost always have the cis configuration ØLipid fluidity likewise increases with the degree of unsaturation of their component fatty acid residues Ø This phenomenon has important consequences for membrane properties.

q Only even-numbered acids are found in triglycerides because the body builds these acids entirely from acetate units and therefore puts the carbons in two at a time. q In triglycerides (also called triacylglycerols), all three groups of glycerol are esterified. q Some unsaturated fatty acids (linoleic and linolenic acids) are called essential fatty acids because the body cannot synthesize them from precursors; they must, therefore, be consumed as part of the diet.

Fatty acids, have certain things in common: 1 Fatty acids are practically all unbranched carboxylic acids. 2 They range in size from about 10 to 20 carbons. 3 They contain an even number of carbon atoms. 4 Apart from the COOH group, they have no functional groups, except that some do have double bonds. 5 In most fatty acids that have double bonds, the cis isomers predominate. 6 - Melting points (mp) of saturated fatty acids, increase with molecular mass
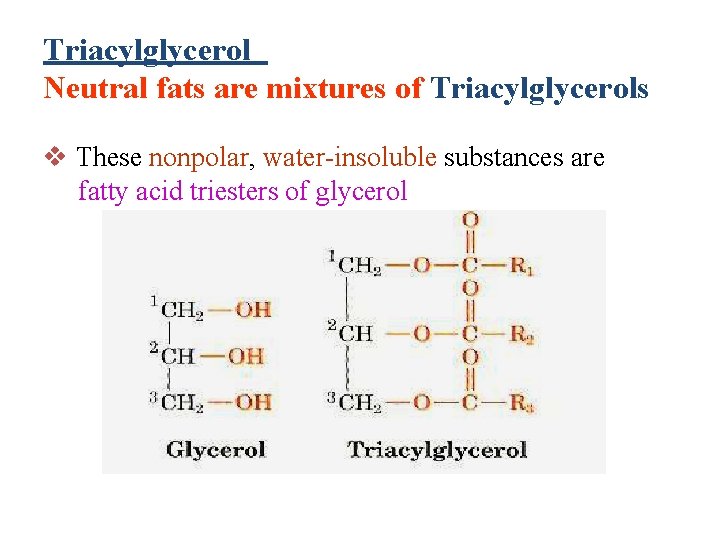
Triacylglycerol Neutral fats are mixtures of Triacylglycerols v These nonpolar, water-insoluble substances are fatty acid triesters of glycerol

v. Fats and oil that occur in plants and animals consist largely of mixtures of Triacylglycerols (also referred to as triglycerides or neutral fats). v Triacylglycerols function as energy reservoirs in animals. v Triacylglycerols differ according to the identity and placement of their three fatty acid residues. 1. Simple triacylglycerols contain one type of fatty acid residue and are named accordingly. For example: A) Tristearoylglycerol or tristearin contains three stearic acid residues B) Trioleoylglycerol or triolein has three oleic acid residues.

2. Mixed triacylglycerols contain two or three different types of fatty acid residues and are named according to their placement on the glycerol moiety. v Fat and oils: v Are differ only in that: v Fats are solid v Oils are liquid at room temperature v. Are complex mixtures of simple and mixed triacylglycerols v Also Plant oils: v Are usually richer in unsaturated fatty acid residues than are animal fats

• Triglycerides (Triacylglycerols )are the most common lipid materials, although mono and diglycerides are not infrequent. In the latter two( mono and di ) types, only one or two OH groups of the glycerol are esterified by fatty acids. • Triglycerides are complex mixtures. Although some of the molecules contain three identical fatty acids, in most cases two or three different fatty acids are present. • The hydrophobic character of triglycerides is caused by the long hydrocarbon chains.

Triacylglycerols are efficient energy reserves • Fats are highly efficient form in which to store metabolic energy: • This is because fats are less oxidized than are carbohydrates or proteins and hence yield significantly more energy on oxidation • Fats, being nonpolar substances, are stored in anhydrous form: • Whereas glycogen bind about twice its weight of water

• Fats therefore provide about six times the metabolic energy of an equal weight of hydrates glycogen. • In animals, • Adipocytes (fat cells) are specialized for the synthesis and storage of triacylglycerols.

• Adipose tissue is most abundant in a subcutaneous layer and in the abdominal cavity • Fat content of normal human (21% for men, 26% for women) enables them to survive starvation for 2 to 3 months. • In contrast, the body’s glycogen supply, which functions are a short-term energy store, can provide for the body’s metabolic need for less than a day. • The subcutaneous fat layer also provides thermal insulation.

Pure fats and oils are colorless, odorless, and tasteless. This statement may seem surprising because we all know the tastes and colors of such fats and oils as butter. and olive oil. The tastes, odors, and colors are caused by small amounts of other substances dissolved in the fat or oil

What Are the Structures of Complex Lipids? Complex lipids can be classified into two groups: . Ø 1 Phospholipids contain an alcohol, two fatty acids, and a phosphate group. There are two types of phospholipids : glycerophospholipids , the alcohol is glycerol sphingolipids, the alcohol is sphingosine. Ø 2 Glycolipids are complex lipids that contain carbohydrates.
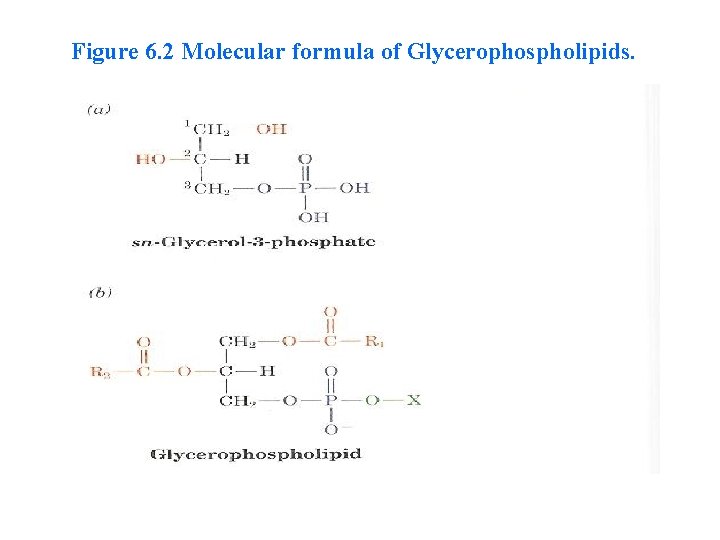
Figure 6. 2 Molecular formula of Glycerophospholipids.

Glycerophospholipids (Phosphoglycerides) • Glycerophospholipids are major lipid components of biological membranes. q The alcohol in Glycerophospholipids is glycerol. q Two of the three carbons of glycerol (C 1 and C 2 ) are esterified by fatty acids, these fatty acids may be any long-chain carboxylic acids, with or without double bonds. q In all glycerophospholipids, lecithins, cephalins, and phosphatidylinositols, the fatty acid on carbon 2 of glycerol , is always unsaturated.

• The third carbon of glycerol is esterified not by a fatty acid, but rather by a phosphate group, and the phosphoryl group connect to a group (X) • The simplest Glycerophospholipids, in which X=H, are phosphatidic acids; they are present only in small amounts in biological membranes (Table 6. 2).

In the common classes of Glycerophospholipids , the X group is another alcohol that is esterified to the phosphate group. ØIf the another alcohol is choline , the glycerophospholipids are called phosphatidyl cholines (common name lecithin), ØIf the another alcohol is ethanolamine or serine are called cephalins. ØIf the another alcohol inositol , is called phosphatidyli nositols.
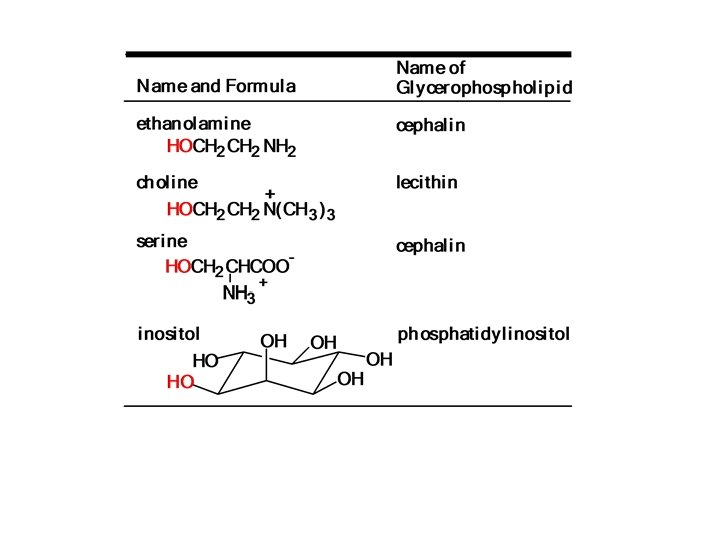
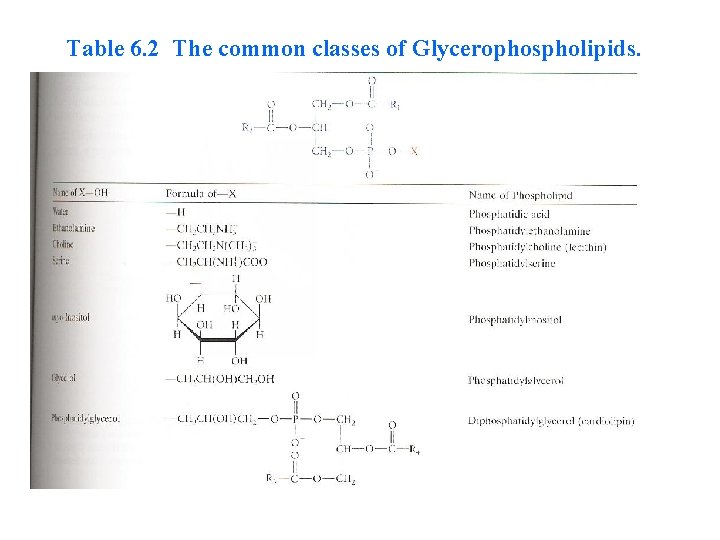
Table 6. 2 The common classes of Glycerophospholipids.

Sphingolipids comprise a second class of molecules in biological membranes In sphingolipids, the alcohol portion is sphingosine ( not glycerol ) The combination of a fatty acid and sphingosine is called the ceramide portion of the molecule. The phosphate group of sphingosine is esterified with the alcohol choline. The ceramide part of complex lipids may contain different fatty acids. Stearic acid, for example, occurs mainly in sphingomyelin. •

Sphingomyelins are the most common Sphingolipids. Myelin, the coating of nerve axons, contains a different kind of complex lipid Sphingomyelins are the most important lipids in the myelin sheaths of nerve cells. Sphingolipids are not randomly distributed in membranes.
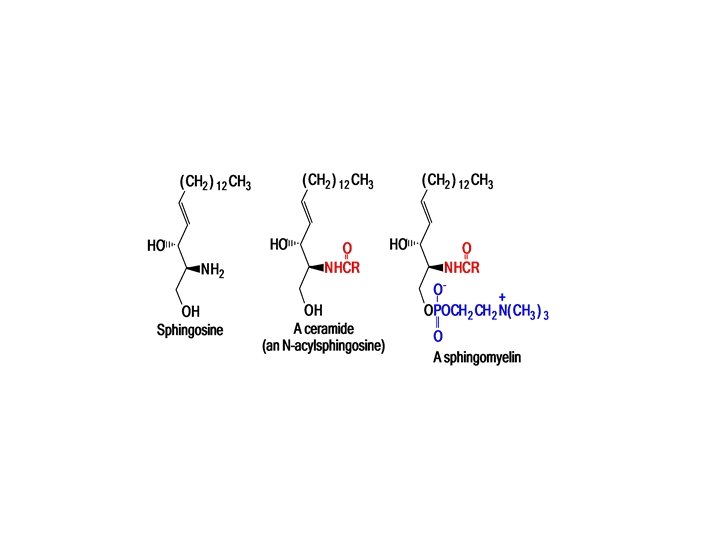

• A long chain fatty acid is connected to the NH 2 group of sphingosine by an amide bond, • And • the OH group at the end of the chain is esterified by phosphorylcholine.

Glycolipids are complex lipids that contain carbohydrates and ceramides. The cerebrosides, consists of ceramide mono or oligosaccharides. The gangliosides, contain a more complex carobohydrate structure. The cerebrosides occur primarily in the brain (accounting for 7% of the brain's dry weight) and at nerve synapses.
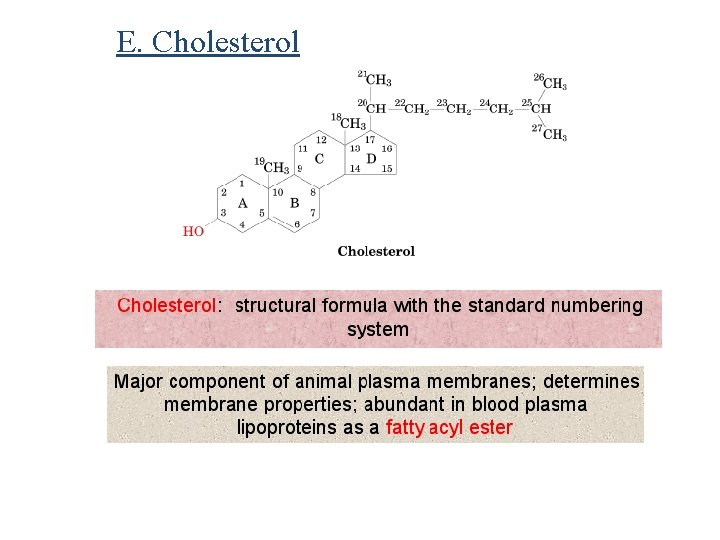
E. Cholesterol

Ø Cholesterol is another membrane component, also positions the hydrophilic portion of the molecule on the surface of the membranes and the hydrophobic portion inside the bilayer. Ø It is also abundant in blood plasma lipoproteins Ø Cholesterol is the metabolic precursor of steroid hormones: Ø Substances that regulate a great variety of physiological functions including: Ø Sexual development Ø Carbohydrate metabolism.

Lipoproteins: Carriers of Cholesterol • Cholesterol, along with fat, is 'transported by lipoproteins. There are four kinds of lipoproteins: v High-density lipoprotein (HDL) ("good cholesterol"), which consists of about 33% protein and about 30% cholesterol v Low-density lipoprotein (LDL) ("bad cholesterol"), which contains only 25% protein but 50% cholesterol v Very-Iow-density lipoprotein (VLDL), which mostly carries triglycerides (fats) synthesized by the liver v Chylomicrons, which carry dietary lipids synthesized in the intestines

Role of Lipids in the Structure of Membranes The complex lipids form the membranes around body cells and around small structures inside the cell's. (organelles. ) Unsaturated fatty acids are important components of these lipids. Most lipid molecules in the bilayer contain at least one unsaturated fatty acid. The cell membranes separate cells from the external environment and provide selective transport for nutrients and waste products into and out of cells. (against an electrochemical gradient or permit their passage with such a gradient ).

• These membranes are made of lipid bilayers. • In a lipid bilayer, two rows (layers) of complex lipid molecules are arranged tail to tail. • The hydrophobic tails point toward each other, which enables them to get as far away from the water as possible. • This arrangement leaves the hydrophilic heads projecting to the inner and outer surfaces of the membrane.
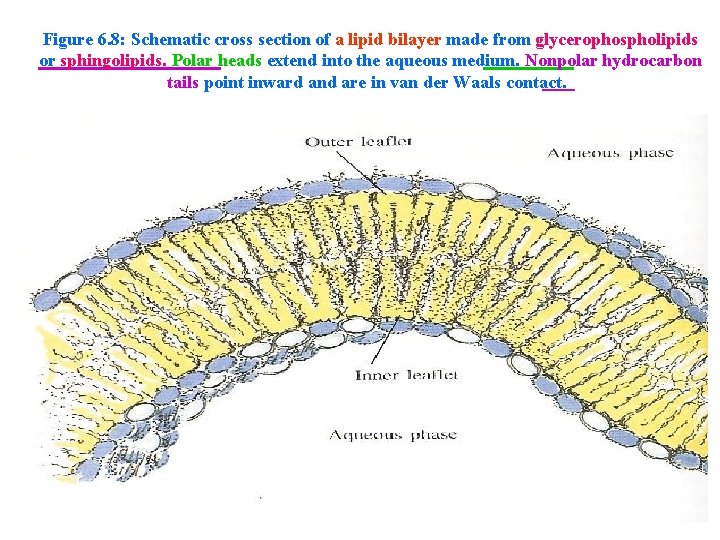
Figure 6. 8: Schematic cross section of a lipid bilayer made from glycerophospholipids or sphingolipids. Polar heads extend into the aqueous medium. Nonpolar hydrocarbon tails point inward and are in van der Waals contact.

Lipid Bilayers Are Fluid Ø A lipid bilayer is about 6 mm across. This is so thin that we can regard the membrane as a twodimensional fluid. Ø Lipid molecules in a bilayer are highly oriented. Ø The unsaturated fatty acids prevent the tight packing of the hydrophobic chains in the lipid bilayer, thereby providing a liquid-like character to the membranes. ØThis effect is similar to the one that causes unsaturated fatty acids to have lower melting points than saturated fatty acids

• This property of membrane fluidity is of extreme importance because many products of the body's biochemical processes must cross the membrane, and the liquid nature of the lipid bilayer allows such transport. • The lipid part of the membrane serves as a barrier against any movement of ions or polar compounds into and out of the cells.

In the lipid bilayer, protein molecules are either suspended on the surface (peripheral proteins) or partly or fully embedded in the bilayer (integral proteins). The model shown in the Figure, called the fluid mosaic model of membranes, allows the passage of nonpolar compounds by diffusion, as these compounds are soluble in the lipid membranes. The term mosaic refers to the topography of the bilayers: protein molecules dispersed in the lipid

Ø In contrast, polar compounds are transported either via: Ø specific channels through the protein regions or Ø by a mechanism called active transport.
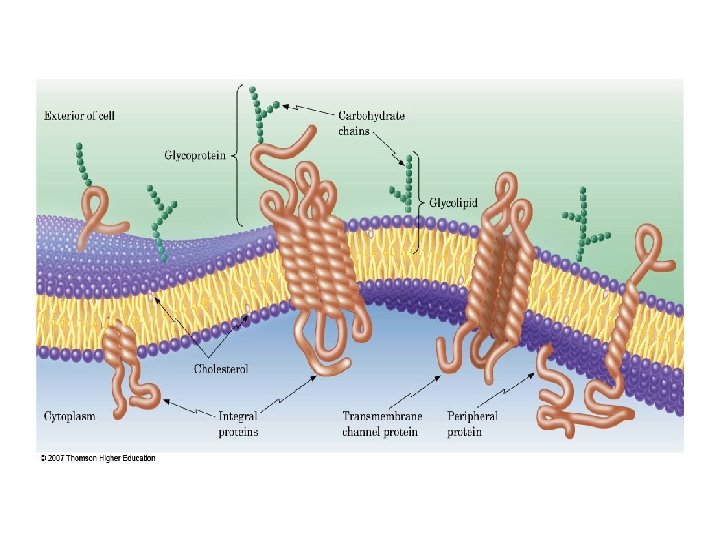

Lipid Bilayers Are Selective. Permeability Barriers § The hydrophobic hydrocarbon chains in the lipid bilayer separates the aqueous compartment inside the vesicle from its aqueous surroundings. § This thin layer of hydrophobic hydrocarbon acts as a selective-permeability barrier, which allows hydrophobic molecules to diffuse across the bilayer at relatively rapid rates while serving as an almost impenetrable barrier to hydrophilic molecules.

The direction in which net diffusion of hydrophilic molecules across a bilayer occurs depends upon the concentration of a solute inside and outside the vesicle. The difference in the concentration of a substance on the two sides of the bilayer is called the concentration gradient §Spontaneous diffusion occurs down the concentration gradient from the side with the higher concentration to the side with the lower concentration.

§ The rate of diffusion of a hydrophilic substance across a lipid bilayer parallels the solubility of that substance in a nonpolar solvent relative to its solubility water. § Polar and ionic substances, on the other hand, are much less soluble in nonpolar solvents than they are in water. § As a result, Polar and ionic substances diffuse slowly across the hydrophobic hydrocarbon interior of the bilayer.

§ Water is a surprising exception to this rule that polar molecules diffuse slowly across lipid bilayer § The high concentration of water surrounding the bilayer facilitates the diffusion of water molecules. § Therefore, even though water is a relatively polar substance, it diffuses more rapidly than other polar substances as a result of its abundance

§ Hydrophobic molecules are more soluble in nonpolar solvents (which have properties similar to those of the hydrocarbon interior of the bilayer) than they are in water § Therefore, they diffuse across lipid bilayers fairly rapidly.


- Slides: 59