LIPID METABOLISM CHOLESTEROL METABOLISM Functions of Cholesterol a
LIPID METABOLISM: CHOLESTEROL METABOLISM
Functions of Cholesterol • a precursor of steroid hormones (progesterone, testosterone, estradiol, cortisol, etc. ) • a precursor of bile acids • a precursor of vitamin D • important component of many mammalian membranes (modulates the fluidity)
Sources of Cholesterol • from the diet • can be synthesized de novo (about 800 mg of cholesterol per day) - in the liver (major site) - in the intestine • Liver-derived and dietary cholesterol are both delivered to body cells by lipoproteins
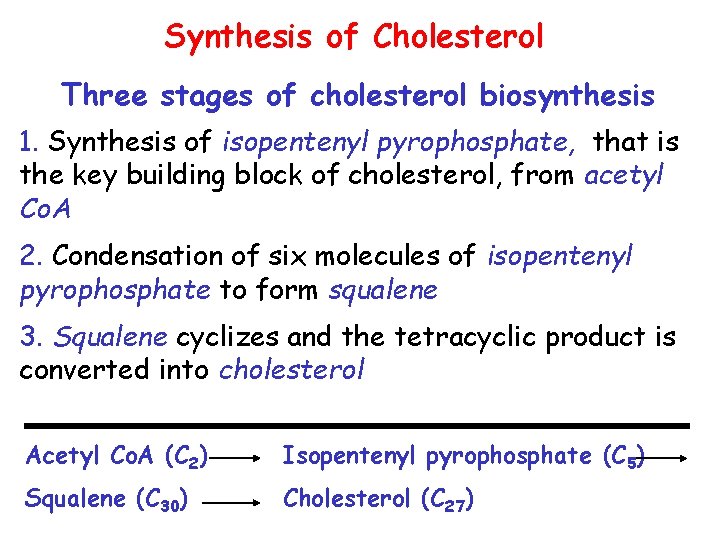
Synthesis of Cholesterol Three stages of cholesterol biosynthesis 1. Synthesis of isopentenyl pyrophosphate, that is the key building block of cholesterol, from acetyl Co. A 2. Condensation of six molecules of isopentenyl pyrophosphate to form squalene 3. Squalene cyclizes and the tetracyclic product is converted into cholesterol Acetyl Co. A (C 2) Isopentenyl pyrophosphate (C 5) Squalene (C 30) Cholesterol (C 27)
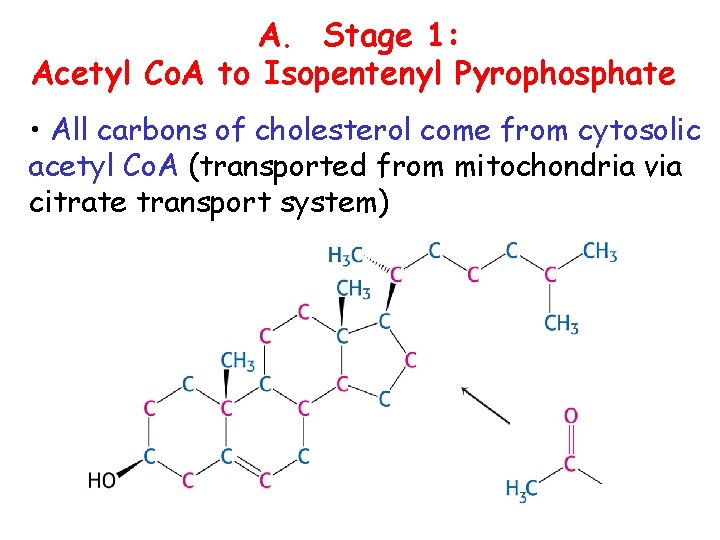
A. Stage 1: Acetyl Co. A to Isopentenyl Pyrophosphate • All carbons of cholesterol come from cytosolic acetyl Co. A (transported from mitochondria via citrate transport system)
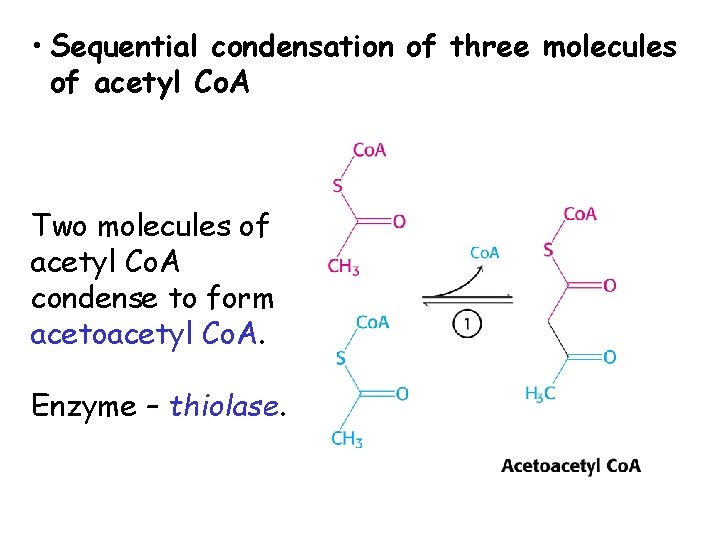
• Sequential condensation of three molecules of acetyl Co. A Two molecules of acetyl Co. A condense to form acetoacetyl Co. A. Enzyme – thiolase.
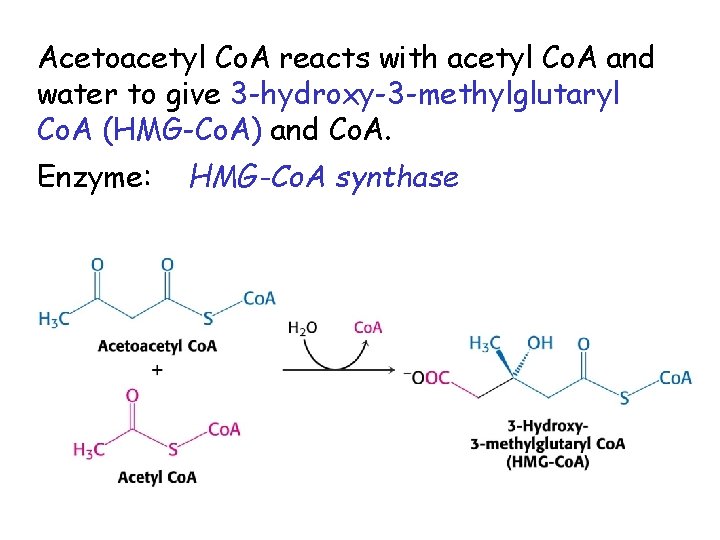
Acetoacetyl Co. A reacts with acetyl Co. A and water to give 3 -hydroxy-3 -methylglutaryl Co. A (HMG-Co. A) and Co. A. Enzyme: HMG-Co. A synthase
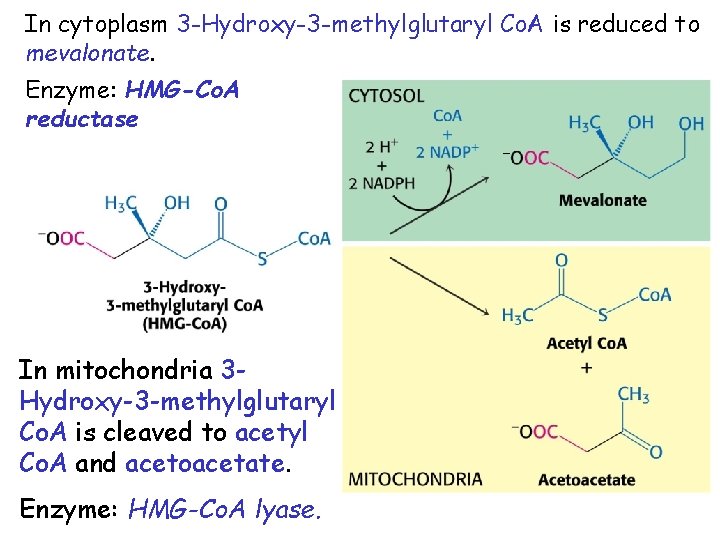
In cytoplasm 3 -Hydroxy-3 -methylglutaryl Co. A is reduced to mevalonate. Enzyme: HMG-Co. A reductase In mitochondria 3 Hydroxy-3 -methylglutaryl Co. A is cleaved to acetyl Co. A and acetoacetate. Enzyme: HMG-Co. A lyase.
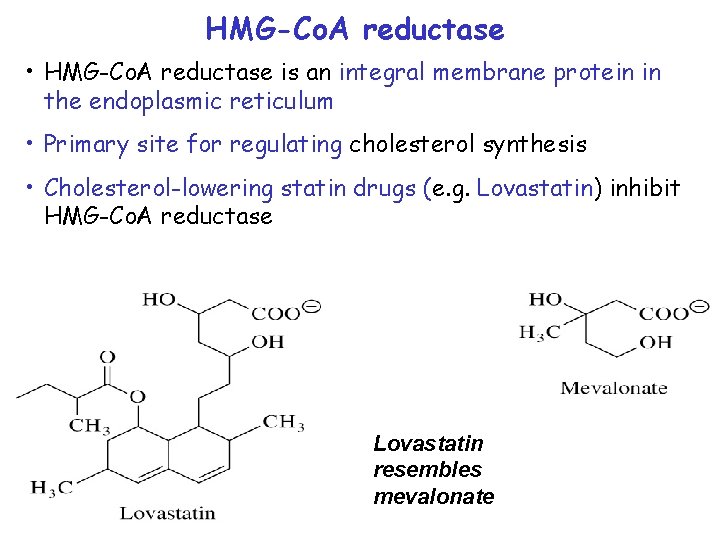
HMG-Co. A reductase • HMG-Co. A reductase is an integral membrane protein in the endoplasmic reticulum • Primary site for regulating cholesterol synthesis • Cholesterol-lowering statin drugs (e. g. Lovastatin) inhibit HMG-Co. A reductase Lovastatin resembles mevalonate
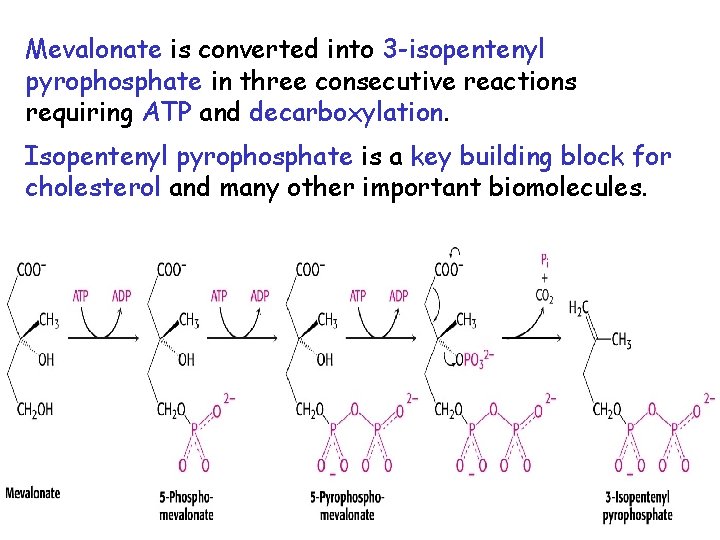
Mevalonate is converted into 3 -isopentenyl pyrophosphate in three consecutive reactions requiring ATP and decarboxylation. Isopentenyl pyrophosphate is a key building block for cholesterol and many other important biomolecules.
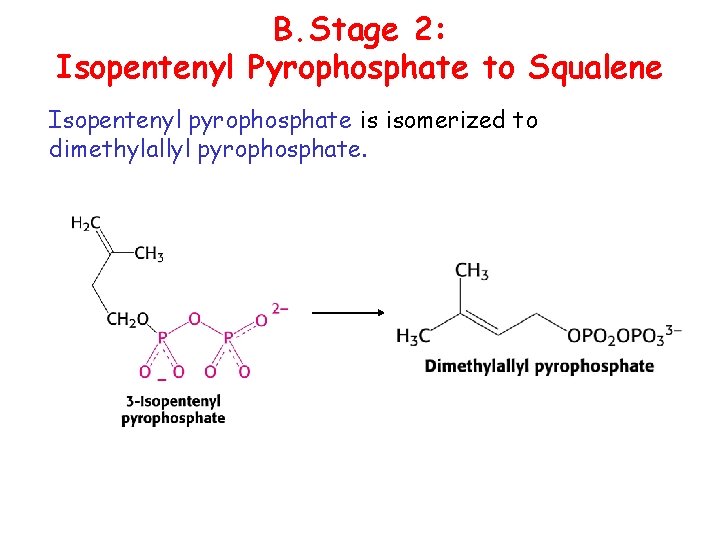
B. Stage 2: Isopentenyl Pyrophosphate to Squalene Isopentenyl pyrophosphate is isomerized to dimethylallyl pyrophosphate.
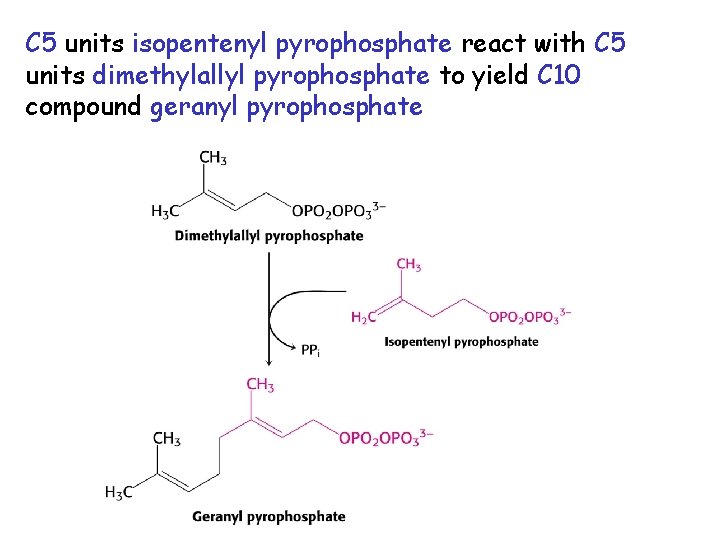
C 5 units isopentenyl pyrophosphate react with C 5 units dimethylallyl pyrophosphate to yield C 10 compound geranyl pyrophosphate
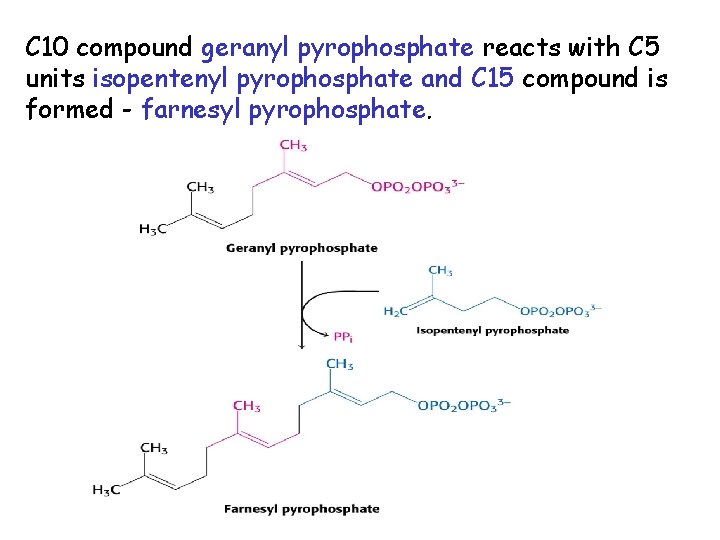
C 10 compound geranyl pyrophosphate reacts with C 5 units isopentenyl pyrophosphate and C 15 compound is formed - farnesyl pyrophosphate.

Reductive tail-to-tail condensation of two molecules of farnesyl pyrophosphate results in the formation of C 30 compound squalene
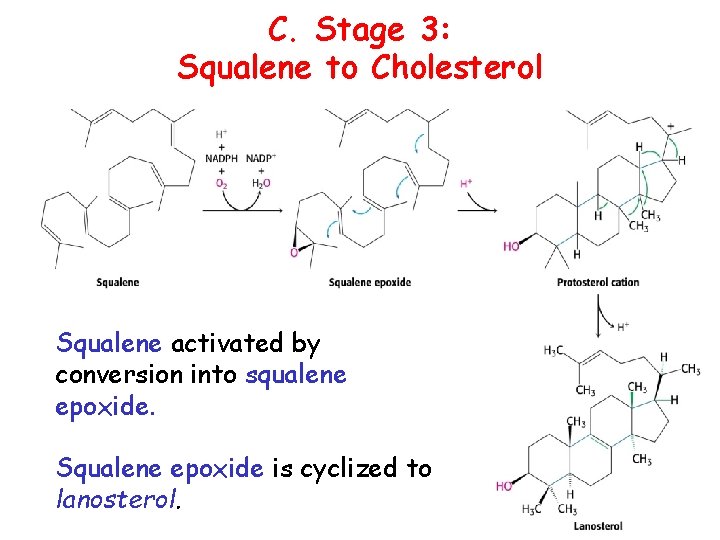
C. Stage 3: Squalene to Cholesterol Squalene activated by conversion into squalene epoxide. Squalene epoxide is cyclized to lanosterol.
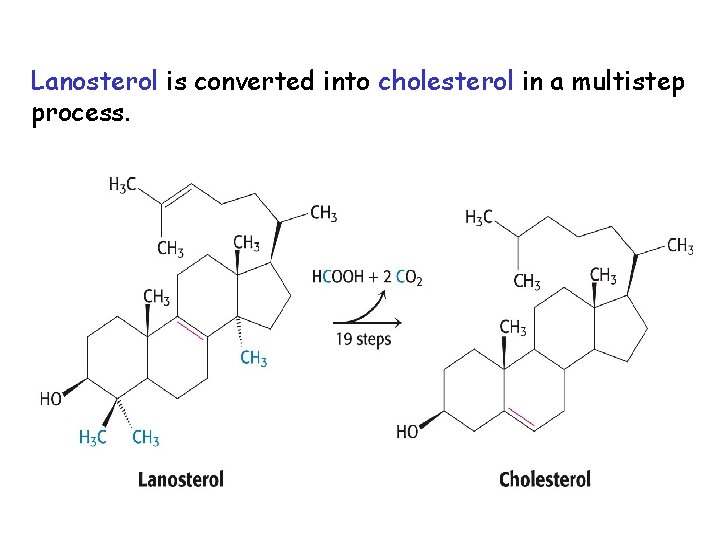
Lanosterol is converted into cholesterol in a multistep process.

THE REGULATION OF CHOLESTEROL BIOSYNTHESIS Regulatory enzyme - 3 -hydroxy-3 -methylglutaryl Co. A reductase. Tetrameric enzyme. NADPH coenzyme

HMG Co. A reductase is controlled in multiple ways: 1. The rate of synthesis of reductase m. RNA is controlled by the sterol regulatory element binding protein (SREBP). When cholesterol levels fall this protein migrates to the nucleus and enhance transcription. 2. The rate of translation of reductase m. RNA is inhibited by cholesterol 3. The degradation of the reductase is controlled. The increase of cholesterol concentration makes the enzyme more susceptible to proteolysis. 4. Phosphorylation decreases the activity of the reductase. Enzyme is switched off by an AMP-activated protein kinase. Thus, cholesterol synthesis ceases when the ATP level is low.
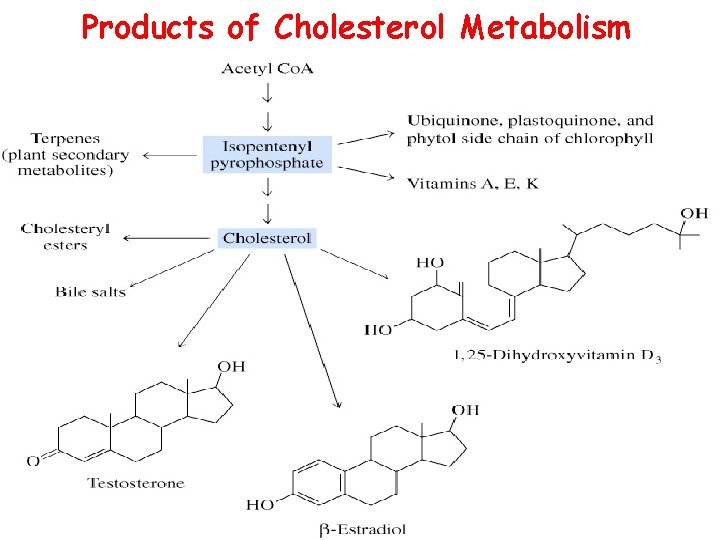
Products of Cholesterol Metabolism

ATHEROSCLEROSIS The desirable level of cholesterol in blood plasma: < 200 mg/dl (< 5 mmol/l)
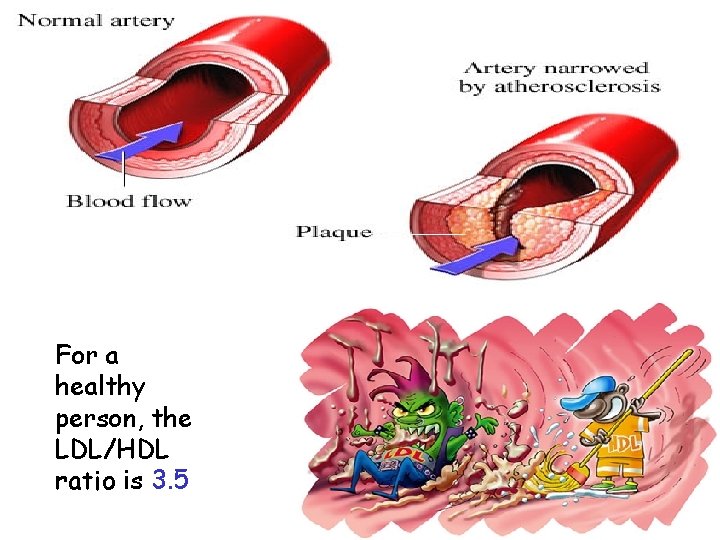
For a healthy person, the LDL/HDL ratio is 3. 5
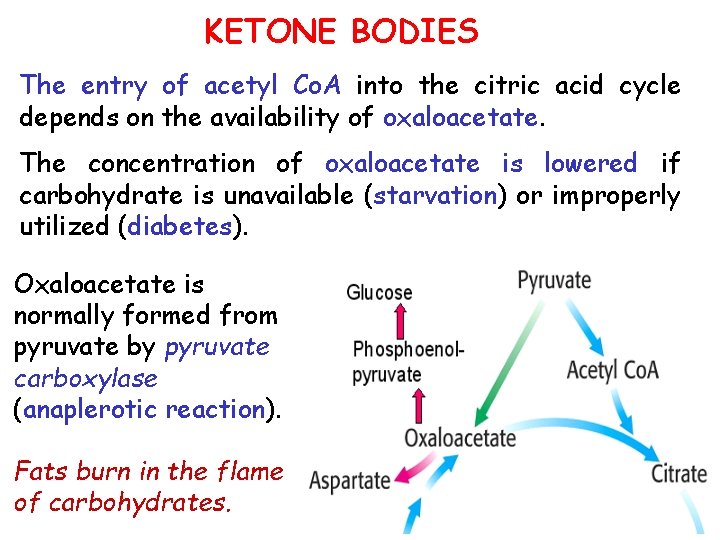
KETONE BODIES The entry of acetyl Co. A into the citric acid cycle depends on the availability of oxaloacetate. The concentration of oxaloacetate is lowered if carbohydrate is unavailable (starvation) or improperly utilized (diabetes). Oxaloacetate is normally formed from pyruvate by pyruvate carboxylase (anaplerotic reaction). Fats burn in the flame of carbohydrates.
In fasting or diabetes the gluconeogenesis is activated and oxaloacetate is consumed in this pathway. Fatty acids are oxidized producing excess of acetyl Co. A which is converted to ketone bodies: b-Hydroxybutyrate Acetoacetate Acetone Ketone bodies are synthesized in liver mitochondria and exported to different organs. Ketone bodies are fuel molecules (can fuel brain and other cells during starvation)
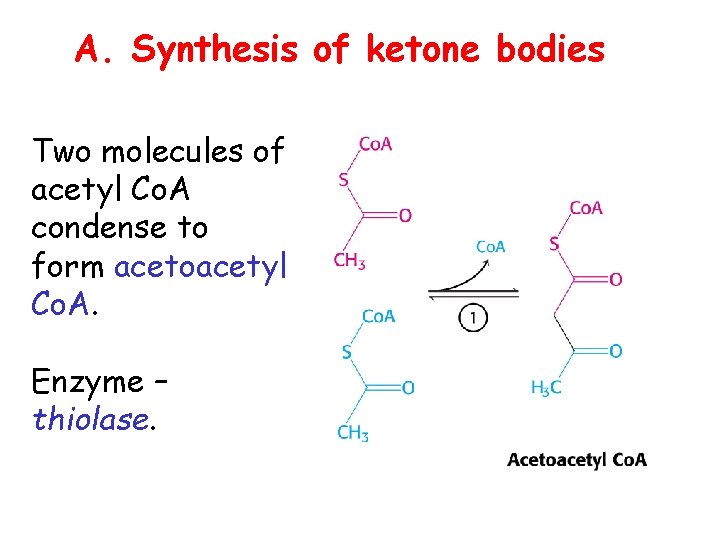
A. Synthesis of ketone bodies Two molecules of acetyl Co. A condense to form acetoacetyl Co. A. Enzyme – thiolase.
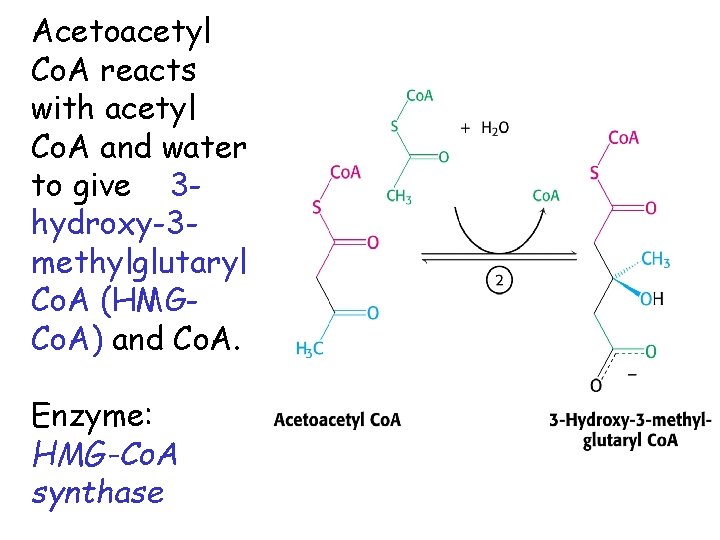
Acetoacetyl Co. A reacts with acetyl Co. A and water to give 3 hydroxy-3 methylglutaryl Co. A (HMGCo. A) and Co. A. Enzyme: HMG-Co. A synthase
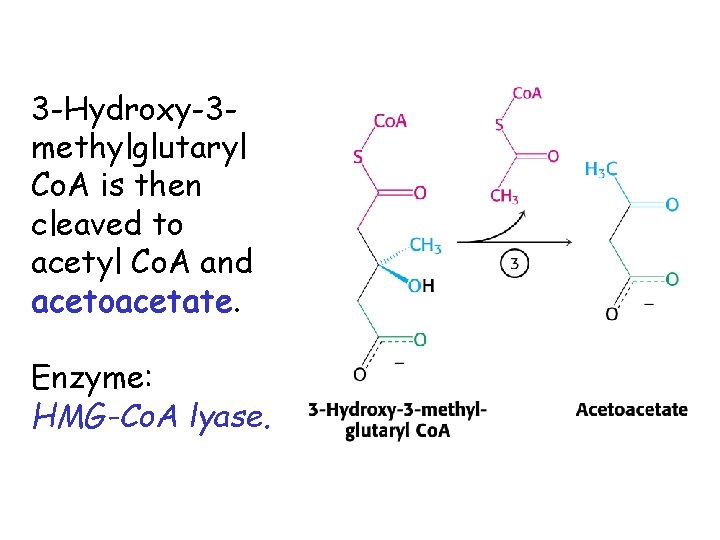
3 -Hydroxy-3 methylglutaryl Co. A is then cleaved to acetyl Co. A and acetoacetate. Enzyme: HMG-Co. A lyase.
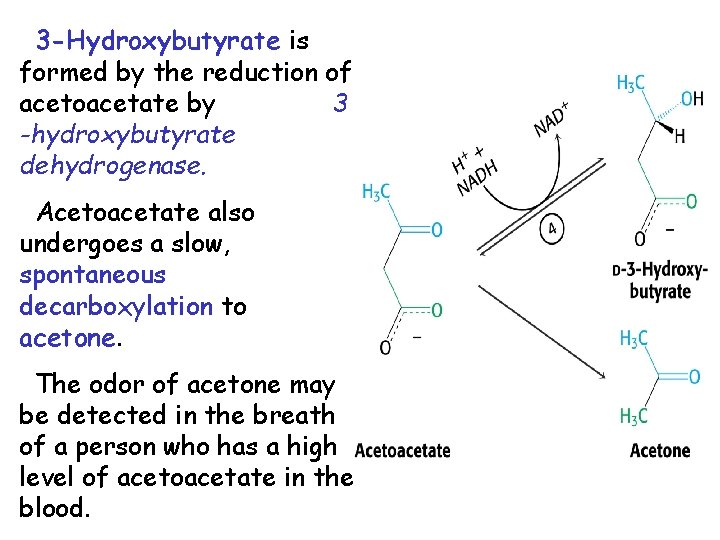
3 -Hydroxybutyrate is formed by the reduction of acetoacetate by 3 -hydroxybutyrate dehydrogenase. Acetoacetate also undergoes a slow, spontaneous decarboxylation to acetone. The odor of acetone may be detected in the breath of a person who has a high level of acetoacetate in the blood.

B. Ketone bodies are a major fuel in some tissues Ketone bodies diffuse from the liver mitochondria into the blood and are transported to peripheral tissues. Ketone bodies are important molecules in energy metabolism. Heart muscle and the renal cortex use acetoacetate in preference to glucose in physiological conditions. The brain adapts to the utilization of acetoacetate during starvation and diabetes.
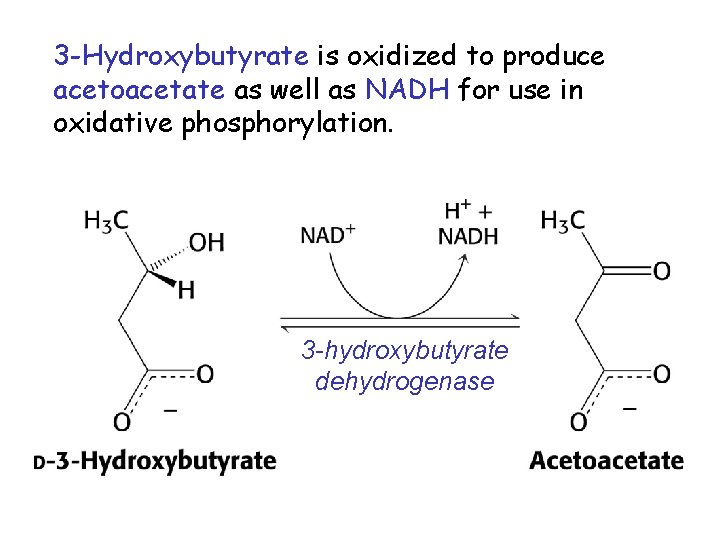
3 -Hydroxybutyrate is oxidized to produce acetoacetate as well as NADH for use in oxidative phosphorylation. 3 -hydroxybutyrate dehydrogenase
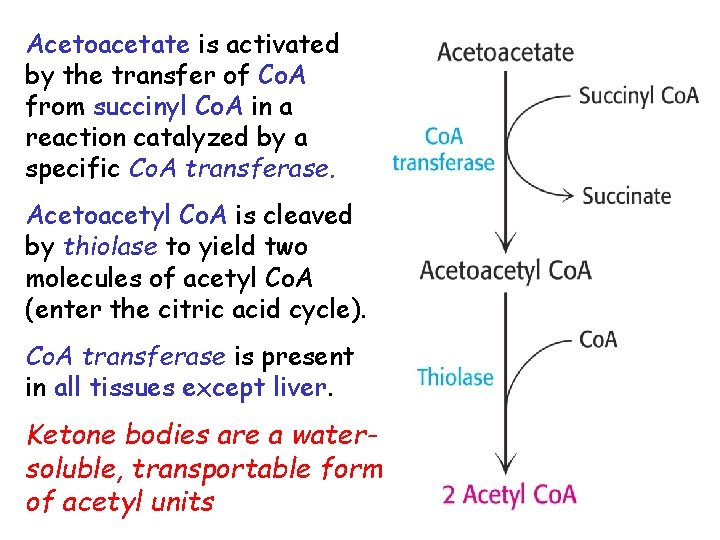
Acetoacetate is activated by the transfer of Co. A from succinyl Co. A in a reaction catalyzed by a specific Co. A transferase. Acetoacetyl Co. A is cleaved by thiolase to yield two molecules of acetyl Co. A (enter the citric acid cycle). Co. A transferase is present in all tissues except liver. Ketone bodies are a watersoluble, transportable form of acetyl units

KETOSIS The absence of insulin in diabetes mellitus § liver cannot absorb glucose § inhibition of glycolysis § activation of gluconeogenesis § deficit of oxaloacetate § activation of fatty acid mobilization by adipose tissue § large amounts of acetyl Co. A which can not be utilized in Krebs cycle § large amounts of ketone bodies (moderately strong acids) § severe acidosis (ketosis) Impairment of the tissue function, most importantly in the central nervous system
- Slides: 31