Link to Phet build an atom Atoms are

• Link to Phet build an atom

Atoms are made of • Protons Neutrons Electrons ↓ ↓ ↓ • Nucleus Outside nucleus • +1 0 -1 • 1 amu ≈ 1 amu 0 (amu = atomic mass unit =1. 66 x 10 -24 g) ↓ ↓ ↓ ATOMIC # ISOTOPE IONS

• ATOMIC NUMBER – number of protons in the nucleus; determines chemical identity of atom • ISOTOPE- atoms of the same element with a different atomic mass; same # of protons but differ in # of neutrons • ION – an atom with a charge (either + or -) + ions have lost electrons; - ions have gained electrons
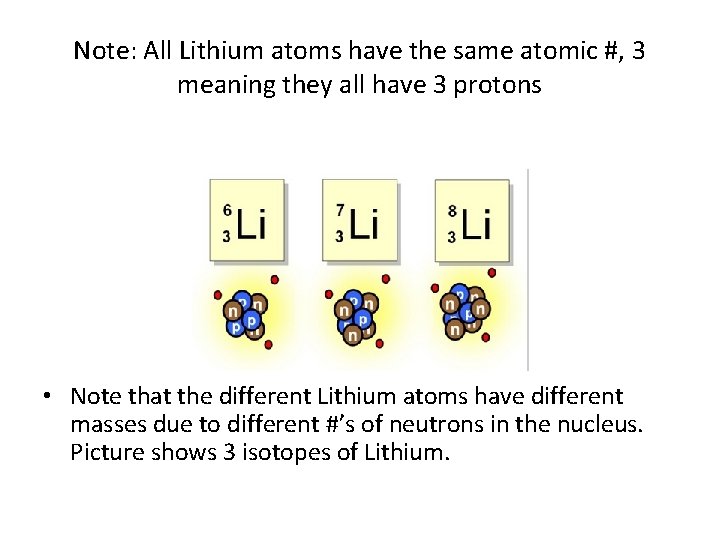
Note: All Lithium atoms have the same atomic #, 3 meaning they all have 3 protons • Note that the different Lithium atoms have different masses due to different #’s of neutrons in the nucleus. Picture shows 3 isotopes of Lithium.

Isotopes have different masses due to different number of neutrons
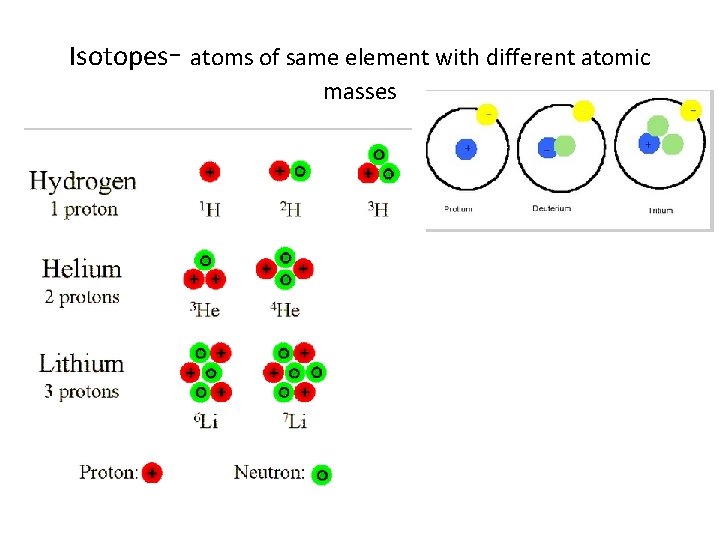
Isotopes- atoms of same element with different atomic masses
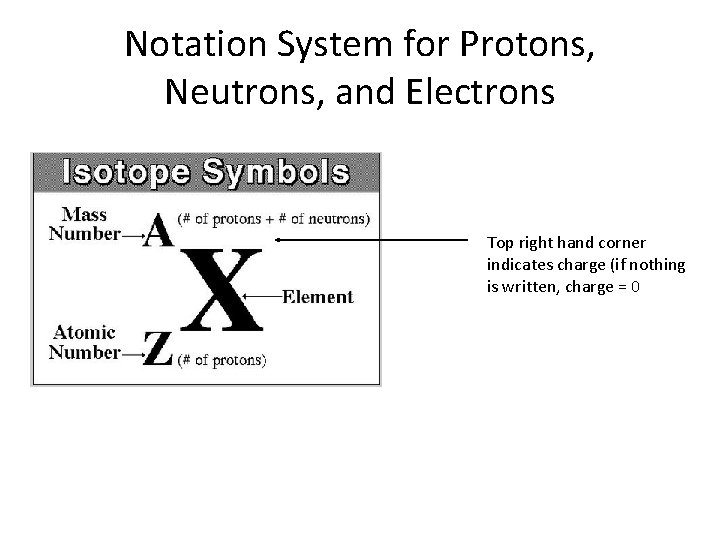
Notation System for Protons, Neutrons, and Electrons Top right hand corner indicates charge (if nothing is written, charge = 0

Atoms may be: • Neutral = no charge • Positive (+) • Negative (-) • Charged atoms = ION Ions form when atoms gain or lose electrons. Gain 1 e- = -1 charge; Gain 2 e- = -2 charge Lose 1 e- = +1 charge; Loses 2 e- = +2 charge
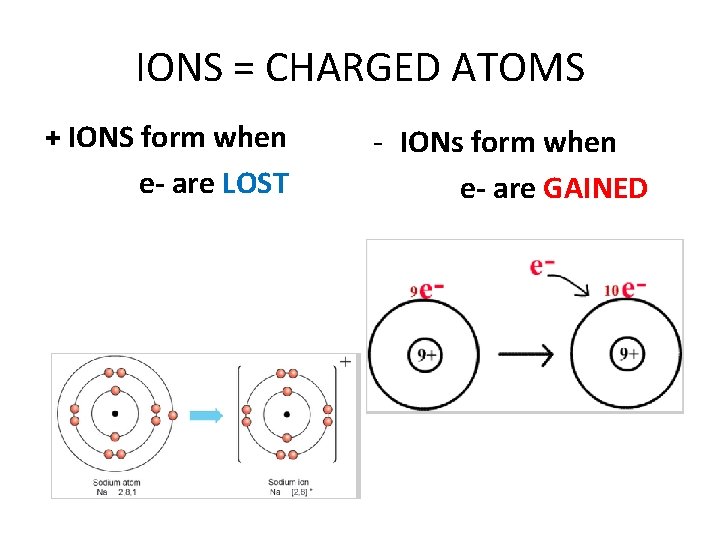
IONS = CHARGED ATOMS + IONS form when e- are LOST - IONs form when e- are GAINED
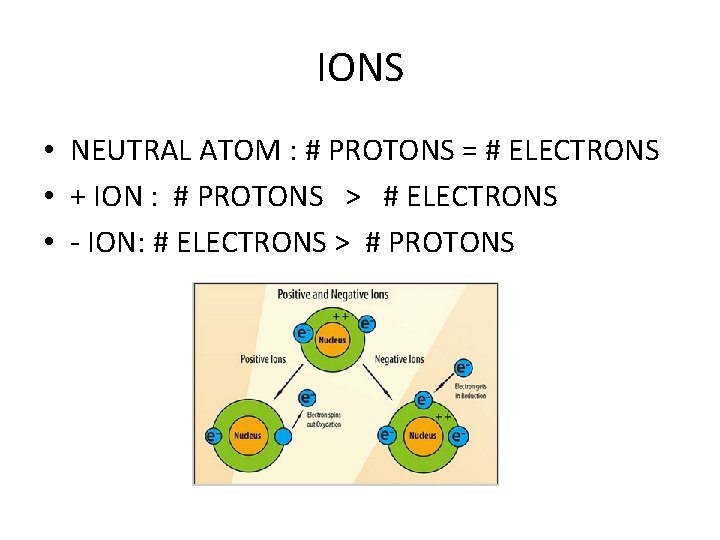
IONS • NEUTRAL ATOM : # PROTONS = # ELECTRONS • + ION : # PROTONS > # ELECTRONS • - ION: # ELECTRONS > # PROTONS

Example: Determine # of proton, neutrons and electrons 25 +2 Mg 12 Protons: Atomic # (Lower left) = Neutrons: Mass (top left) – atomic # = Electrons: # protons (bottom left) – charge (top right) =

Example: Determine # of proton, neutrons and electrons 25 +2 Mg 12 Protons: Atomic # (Lower left) = 12 Neutrons: Mass (top left) – atomic # = Electrons: # protons (bottom left) – charge (top right) =

Example: Determine # of proton, neutrons and electrons 25 +2 Mg 12 Protons: Atomic # (Lower left) = 12 Neutrons: Mass (top left) – atomic # = 13 Electrons: # protons (bottom left) – charge (top right) =

Example: Determine # of proton, neutrons and electrons 25 +2 Mg 12 Protons: Atomic # (Lower left) = 12 Neutrons: Mass (top left) – atomic # = 13 Electrons: # protons (bottom left) – charge (top right) = 12 – (+2) = 10 electrons

Determine the # of protons, neutrons and electrons. • 11 27 B 5 P: N: E: +3 14 Al -3 N 13 7 P: N: E:

Write notation in vertical format and then determine # of proton, neutrons and electrons • C-12 C-13 C-14
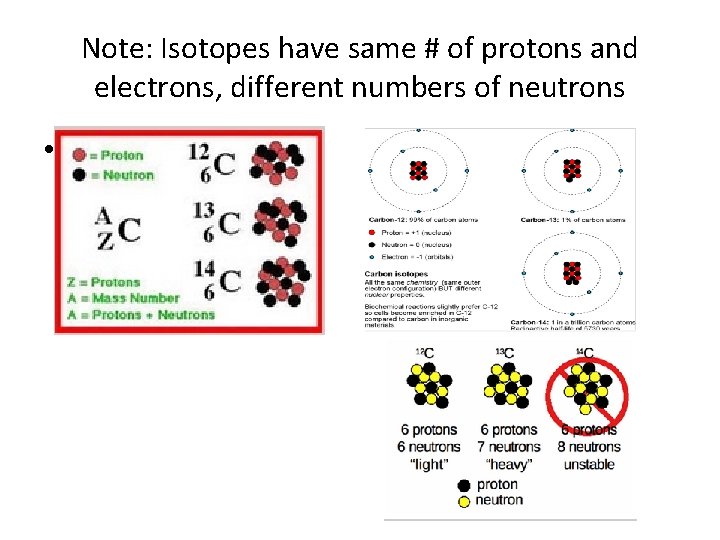
Note: Isotopes have same # of protons and electrons, different numbers of neutrons •

Isotopes of Elements in Nature • A sample of the atoms of a naturally occurring element will contain a mixture of the different isotopes of that element.

Link to Isotopes

Thought Question • The mass of boron on the periodic table is listed as 10. 8 amu, although there are no atoms of carbon that actually weigh 10. 8 amu. Where does the number 10. 8 come from? • Hint #1: How would you calculate the average of 10 and 11? • Hint #2: How would you calculate the average of 10, 11, 11, ?

Link to Isotopes

Average Atomic Mass on the Periodic Table • The atomic mass of an element given on the periodic table represents the weighted average of all of the isotopes of the element, taking into account the relative abundance of each isotope.


• Link to Phet build an atom

Homework 1 -10 • p. 107 , 29 -32 • 29) Explain what we mean when we say an atom has several isotopes. • 30)Define atomic # and mass # of the nucleus • 31) For a neutral atom the number of protons and electrons is ______ (different or same). • 32) The ____ number represents the sum of protons and neutrons in the nucleus.

HW 1 -10 • p. 107 • How many protons and neutrons are contained in the nucleus of each of the following atoms? • 40 a) 39 K 40 c) 84 Se 40 e)91 Kr 19 34 36

HW 1 -10 • p. 104, #2: • The average mass of a carbon atom is 12. 011. Assuming you could pick up one carbon atom, what is the chance that you would randomly get one with a mass of 12. 011? • • A) 0% B) 0. 011% C) about 12% D) 12. 011% E) greater than 50% F) none of the above. Explain your answer.

Homework 1 -10 • p. 105, #17 • 17) Chlorine has 2 prominent isotopes Cl-37 and Cl-35. Which is more abundant? How do you know? (Hint: Use the periodic table)
- Slides: 28