Limulus Amebocyte Lysate LAL Test Methods LAL Test

Limulus Amebocyte Lysate (LAL) Test Methods

LAL Test Methods • The gel-clot method • The kinetic turbidimetric method • The chromogenic methods (kinetic and endpoint)
Gel-Clot Turbidimetric Chromogenic
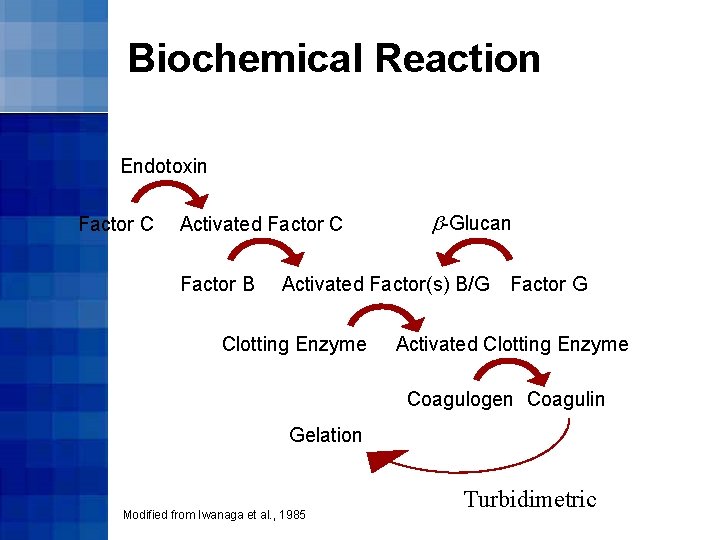
Biochemical Reaction Endotoxin Factor C Activated Factor C Factor B b-Glucan Activated Factor(s) B/G Clotting Enzyme Factor G Activated Clotting Enzyme Coagulogen Coagulin Gelation Modified from Iwanaga et al. , 1985 Turbidimetric

The Gel-Clot Method • Simplest and most widely used • The USP referee method • The labeled gel-clot reagent sensitivity (l) is the least concentration of endotoxin to cause a solid clot under standard conditions

Reading the Gel-Clot Test positive cloudy negative

Turbidimetric Methods • As coagulin molecules coalesce forming particles, the reaction mixture becomes turbid • The rate of increase in turbidity is a function of endotoxin concentration
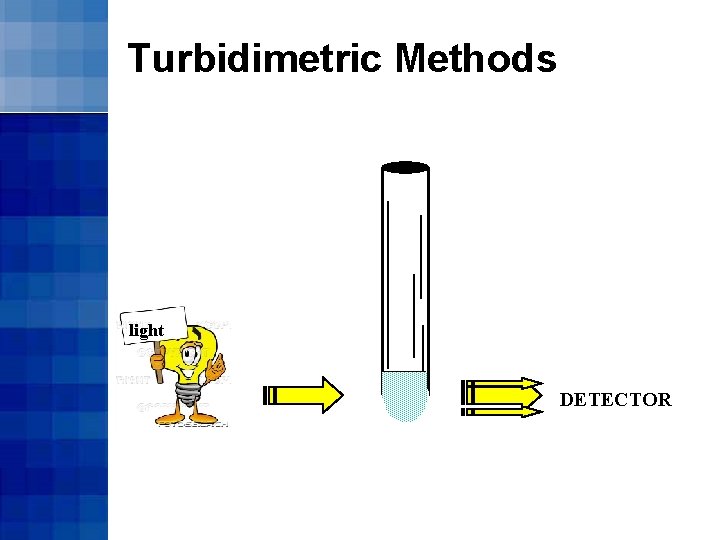
Turbidimetric Methods light DETECTOR
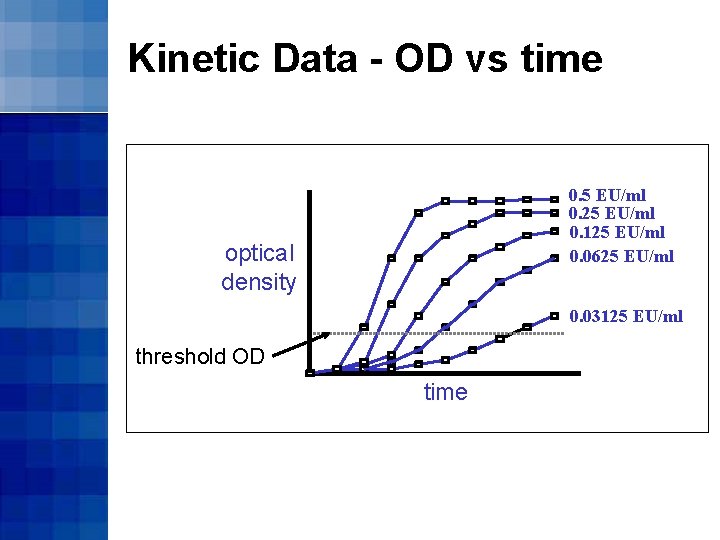
Kinetic Data - OD vs time 0. 5 EU/ml 0. 25 EU/ml 0. 125 EU/ml 0. 0625 EU/ml optical density 0. 03125 EU/ml threshold OD time

Kinetic Turbidimetric Method • The threshold OD (onset OD) is used as a point of reference for data collection • The greater the endotoxin concentration, the shorter the time taken to reach the onset OD

Kinetic Turbidimetric Method • The onset time is the time that it takes (in seconds) for the reaction to reach the onset OD • Standard curves are constructed by plotting log 10(onset time) on log 10(endotoxin concentration)
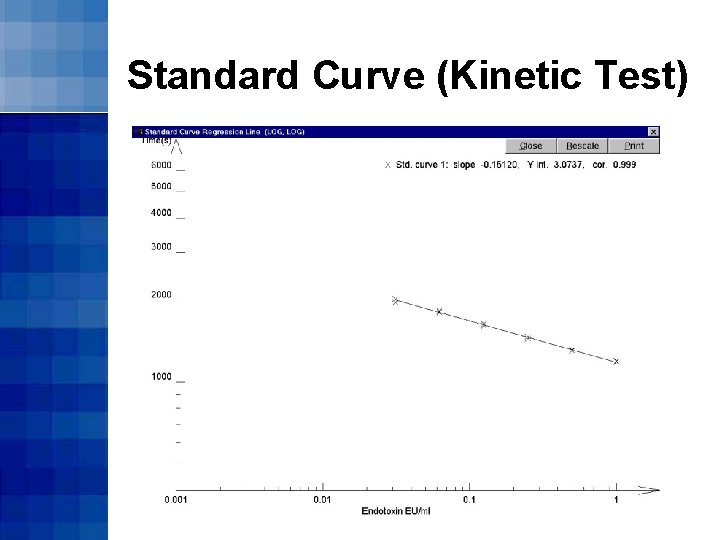
Standard Curve (Kinetic Test)

Kinetic Turbidimetric Method • Calculate sample endotoxin content by comparing with standards • Take sample onset time and reference against standard curve to determine its endotoxin content

Interference • Most samples, at some concentration, interfere with the LAL reaction • Interference is caused by – sample interaction with the LAL reagent – sample interaction with endotoxin

Inhibition • Inhibition is a reduction in sensitivity of the assay which causes an underestimation of the concentration of endotoxin • Inhibition controls (PPC’s) prevent misinterpretation of negative results

Enhancement • Enhancement is an increase in the sensitivity of the assay which causes an overestimation of the concentration of endotoxin • Positive product controls (PPC’s) prevent misinterpretation of positive results in the photometric methods

False Positives • Enhancement is not a false positive! • A false positive test is a positive in the absence of endotoxin • False positives are rare – trypsin (all methods) – activated serine proteases (chromogenic) – beta-glucans (suspected, all methods)

Positive Product Control • All LAL tests must have a control to demonstrate that the sample itself does not cause a false negative result • A known quantity of endotoxin is added to a portion of the sample under test to provide an inhibition or positive product control (PPC)

Remove Interference • Dilute with LRW first • Use a more sensitive LAL reagent or method to increase the MVD • Reconstitute LAL with Pyrosol (strongly buffered products outside the p. H range, highly concentrated electrolytes, or for sample/endotoxin interactions)

Maximum Valid Dilution • The maximum valid dilution (MVD) is the greatest possible dilution at which the limit can be detected • This is the dilution used for the pass/fail test • The MVD increases with increasing test sensitivity

Maximum Valid Dilution • If l is 0. 125 EU/m. L and the unknown has an endotoxin limit of 2 EU/m. L, calculate the MVD: Limit in EU/m. L = 2 EU/m. L = 16 l in EU/m. L 0. 125 EU/m. L Limit in EU/m. L = 2 EU/m. L = 64 l in EU/m. L 0. 03125 EU/m. L

Select a Sensitivity • • Sensitivity is lowest point on curve Consider the endotoxin limit and MVD Perform preliminary tests If interference cannot be overcome without exceeding the MVD of a product, go to a more sensitive reagent or method
- Slides: 22