Limiting Reactants and Percentage Yield vocab Limiting reactant

Limiting Reactants and Percentage Yield

vocab Limiting reactant- the one that runs out first, limits the amount of product that can be made Excess- the one with reactant leftovers, not used up completely

What it looks like C + O 2 = CO 2 Mole ratio is 1 to 1 Given 5 mol of C and 10 mol of O 2, which one is limiting?

What it looks like C + O 2 = CO 2 Mole ratio is 1 to 1 Given 5 mol of C and 10 mol of O 2, which one is limiting? The 5 mol of C is limiting, it will run out first

example If 6 mol of HF is added to 4. 5 mol of Si. O 2, which is the limiting reactant? First write the balanced equation and classify. Compare the mole ratios of each of the reactants to the product using the given amount The limiting reactant is the one that produces the smallest number of moles

example Si. O 2 + 4 HF = Si. F 4 + 2 H 2 O Double displacement Mole ratio of HF to Si. F 4 is 6 mol HF x 1 mol Si. F 4 / 4 mol HF = 1. 5 mol Si. F 4 Mole ratio of Si. O 2 to Si. F 4 is 4. 5 mole Si. O 2 x 1 mol Si. F 4 /1 mol Si. O 2 = 4. 5 mol Si. F 4 6 mol HF produces smaller amount of product so it is the limiting reactant and Si. O 2 is the excess

You try When 36. 0 g of H 2 O is mixed with 67. 0 g Fe it produces hydrogen gas and magnetite Fe 3 O 4, which is the limiting reactant?

equation 3 Fe + 4 H 2 O = Fe 3 O 4 + 4 H 2 Single displacement Fe = 56 g/mol H 2 O = 18 g/mol Fe 3 O 4 = 232 g/mol

Molar mass and mole ratio 67 g Fe/ 56 g Fe x 1 mol magnetite/ 3 mol Fe = 0. 4 mol magnetite 36 g water/ 18 g water x 1 mol magnetite/ 4 mol water = 0. 499 mol magnetite Fe produces the smaller amount of product, so it is the limiting reactant, and water is the excess

One step further Using the limiting reactant, how many moles of magnetite are produced? 0. 4 mol magnetite x 232 g/ 1 mol magnetite = 92 g magnetite produced
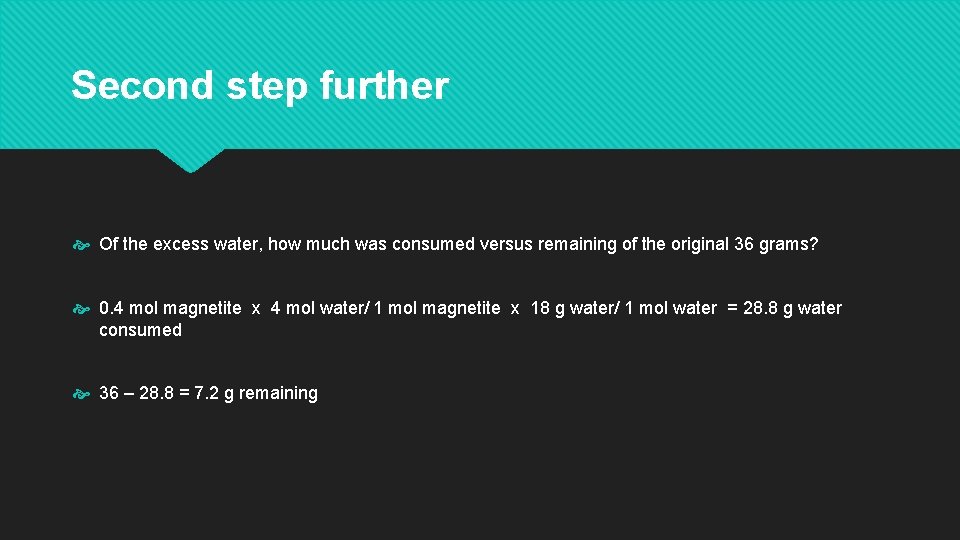
Second step further Of the excess water, how much was consumed versus remaining of the original 36 grams? 0. 4 mol magnetite x 4 mol water/ 1 mol magnetite x 18 g water/ 1 mol water = 28. 8 g water consumed 36 – 28. 8 = 7. 2 g remaining

yield Theoretical yield- maximum amount of product that can be produced from a given amount of reactant, found mathematically Actual yield- the measure amount of a product obtained from a reaction, the amount you actually get Percentage yield- the ratio of actual to theoretical yield, shows how accurate your work is Actual/ theoretical x 100 = percentage yield

Percentage yield Theoretical yield 53 g of magnetite Actual yield 38. 8 g of magnetite What is the percentage yield? 38. 8/ 53 x 100 = 73. 2 %
- Slides: 13