Limiting Reactants and ICE Charts Chemistry Cake You


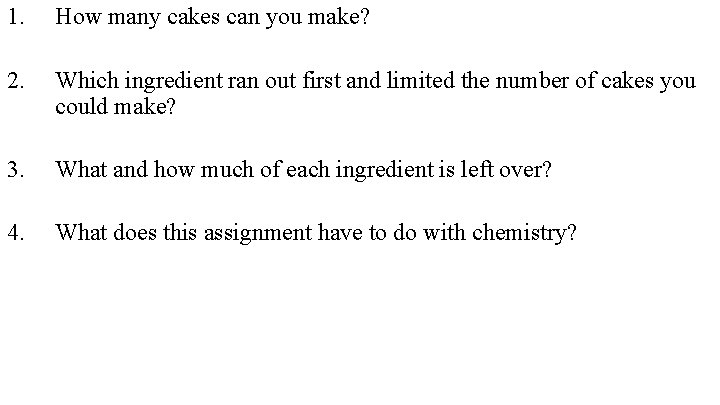






- Slides: 10

Limiting Reactants and ICE Charts

Chemistry Cake You have 20 cups of flour, 8 cups of sugar, 30 litres of milk and 48 eggs in your kitchen. The recipe for chemistry cake is: + 3 cups of flour 2 cups of sugar 2 litres of milk 6 eggs = 1 chemistry cake
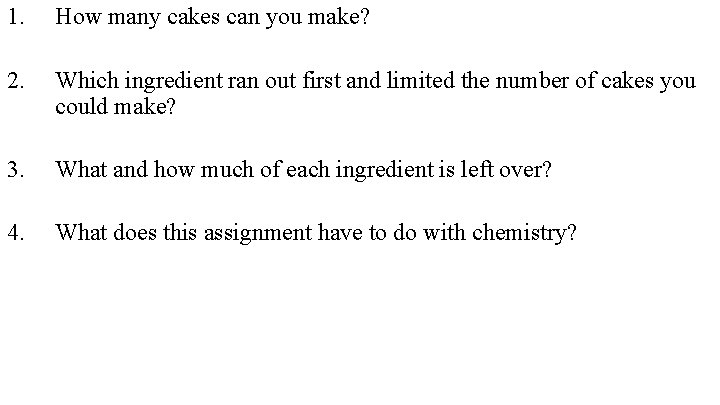
1. How many cakes can you make? 2. Which ingredient ran out first and limited the number of cakes you could make? 3. What and how much of each ingredient is left over? 4. What does this assignment have to do with chemistry?

3 F + 2 S + 2 M + 6 E → 1 Cake Initial 20 8 30 48 0 Change 12 8 8 24 4 End 8 0 22 24 4 The sugar runs out- limiting ingredient 8 cups of sugar will make 4 cakes- so multiple the recipe by 4 All other ingredients are in excess

Limiting Reactant Problems

14. 0 mole Ga and 12. 0 mole O 2 react. Find the limiting reactant, the mass of excess reactant and product made. 4 Ga + 3 O 2 → 2 Ga 2 O 3 0 I 14. 0 12. 0 4 C 16. 0 12. 0 3 Guess that one of the reactants runs out- then check Cannot consume 16. 0 moles – we only have 14 moles

14. 0 mole Ga and 12. 0 mole O 2 react. Find the limiting reactant, the mass of excess reactant and product made. 4 Ga + 3 O 2 → 2 Ga 2 O 3 0 I 14. 0 12. 0 3 2 C 7. 00 14. 0 10. 5 4 3 7. 00 x 187. 4 g E 0 1. 5 x 32. 0 g 1 mole 48. g 1. 31 x 103 g limiting excess product The other reactant must run out- calculate the change Subtract to get what is left- Convert back to grams

14. 0 g of Al reacts with 94. 0 g of Br 2. Find the limiting reactant, the mass of the excess reactant and product. 2 Al + 3 Br 2 14. 0 g x 1 mole 27. 0 g I 0. 5185 mole C 0. 5185 mole → 2 Al. Br 3 94. 0 g x 1 mole 159. 8 g 3 2 0. 5882 mole 0. 7778 mole 0 mole

14. 0 g of Al reacts with 94. 0 g of Br 2. Find the limiting reactant, the mass of the excess reactant and product. 2 Al + 3 Br 2 14. 0 g x 1 mole 27. 0 g I C E 0. 5185 mole 0. 3921 mole 94. 0 g x 1 mole 159. 8 g 2 3 0. 1264 mole x 27. 0 g 1 mole 3. 41 g → 2 Al. Br 3 0. 5882 mole 0 mole 2 3 0. 3921 mole 0. 0000 0. 3921 mole x 266. 7 g 1 mole limiting 105 g

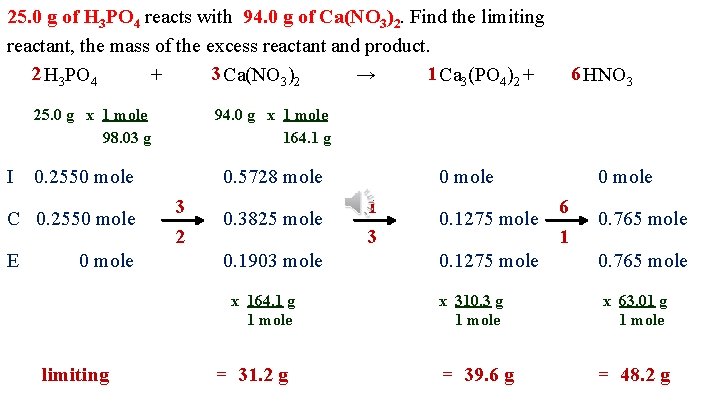
25. 0 g of H 3 PO 4 reacts with 94. 0 g of Ca(NO 3)2. Find the limiting reactant, the mass of the excess reactant and product. 2 H 3 PO 4 3 Ca(NO 3)2 1 Ca 3(PO 4)2 + + → 25. 0 g x 1 mole 98. 03 g I E 94. 0 g x 1 mole 164. 1 g 0. 2550 mole C 0. 2550 mole 0. 5728 mole 3 2 0. 3825 mole 0. 1903 mole x 164. 1 g 1 mole limiting 6 HNO 3 = 31. 2 g 0 mole 1 3 0. 1275 mole x 310. 3 g 1 mole = 39. 6 g 0 mole 6 1 0. 765 mole x 63. 01 g 1 mole = 48. 2 g