Limitations of Molecular Dynamics What can Molecular Dynamics










































- Slides: 45

Limitations of Molecular Dynamics What can Molecular Dynamics not do for us? 1

What do we want to do? our goal is to find, understand, and manipulate the adaptation that evolution used to solve biological problems 2

Where to start? with evolution How did evolution solve a problem? At what scale, or scales? 3

What are our goals? to compute the properties of (open) channels as reliably and usefully as physicists can compute the properties of (low frequency*) Field Effect Transistors (FETS) *less then 100 MHz,

Why these goals? So we can control channels and improve them efficiently.

Why these goals? When computations are accurate, thousands of hours of trial and error experimentation are not needed. Experiments are to check results, not derive them.

Standards and Goals Calculations must reproduce biological function 7

Calculations must reproduce biological function Outputs of Calculations need to be the same graphs we produce from experiments 8

Calculations must reproduce biological function Variables that we know are important must actually be in the calculations if we change the variables in experiments, results change so if we change the variables in calculations, the results must change too 9

Experimentalists: Don’t be misled! Primitive incomplete theories often leave out essential variables. ASK theoreticians EXPLICITLY what variables are present what are absent 10

Examples of Dangerous Calculations • Protein folding without water • Protein folding without definite concentrations of ions • Protein folding with a SINGLE energy landscape, independent of concentration of ions, etc. • Calculations at equilibrium (symmetrical solutions zero membrane potential) that are used to explain NONequilibrium properties (asymmetrical solutions, membrane potential, current and flux) 11

Examples of Dangerous Calculations • Rate models of flow without friction (in a condensed phase like ionic solutions) • MD simulations of flow in an equilibrium system • MD simulations in solutions with unspecified concentrations 12

Difficulties for all atom calculations (nickname MD) Most of biology involves flow 13

Difficulties for all atom calculations (nickname MD) Most of biology involves flow Resting potential involves flow Equilibrium potential produces zero flow ONLY for perfectly selective channel and perfectly selective membrane 14

Almost all MD is at equilibrium no flow of any kind no current voltage relations 15

Why not calculate “free energy of binding” and then calculate flow? You tell me what is wrong Hint: can you derive Ohm’s law that way? 16

Answer Free energy profile is derived with a theory that does not allow flow Free energy profile changes A LOT as membrane potential, ionic concentrations in bath, etc change 17

Must not assume your way out of a problem!! 18

Why not compute all the atoms? 19

Why not compute all the atoms? Some physical systems cannot be computed on an atomic scale 1) Propagating Action Potential 2) Artillery Shell 20

Difficulties for all atom calculations (nickname MD) Most of biology depends on trace concentrations of hormones and Ca ion 21

Difficulties for all atom calculations (nickname MD) 10 -7 M Ca occurs in 55 M water for each Ca ion have 5. 5 × 107 water molecules 1. 65 × 108 atoms need ~1000 calcium ions for statistics Must calculate 1. 65 × 1011 atoms and all their interactions! 22

Difficulties for all-atom calculations Most of biology occurs in mixtures MD is calibrated in ZERO concentration MD of mixtures does not exist because calibration fails MD is designed for zero concentrations of pure monovalents 23

Almost no MD calculations exist of divalents NO MD calculations exist of trace concentrations of ions NO MD calculations exist of mixtures of ions 24

MD force fields are calibrated in distilled water, using “free energy of formation” Membrane phenomena depend on the free energy per mole (activity, approximated sometimes by concentration) MD does not calculate activities very well 25

Ions are very concentrated in and near channels, transporters, active sites of enzymes, and nucleic acids Under these conditions, activity and driving force of one ion is coupled to the concentration of all other ions. 26

In concentrated solutions, activity and driving force of one ion is coupled to the concentration of all other ions. MD does not deal with these issues at all MD is calibrated in ZERO concentration and does not describe mixtures or divalents at all well 27

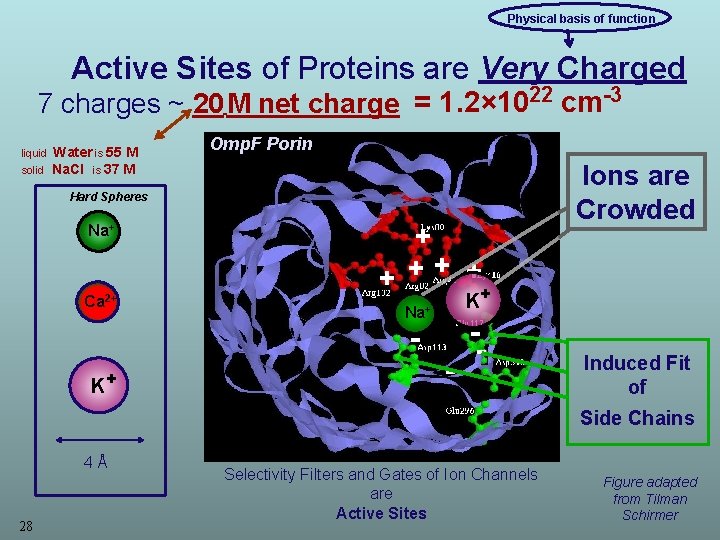
Physical basis of function Active Sites of Proteins are Very Charged 7 charges ~ 20 M net charge = 1. 2× 1022 cm-3 liquid Water is 55 M Omp. F Porin Ions are Crowded solid Na. Cl is 37 M Hard Spheres Na+ Ca 2+ + + + K Na+ K+ 4Å 28 - - Induced Fit of Side Chains Selectivity Filters and Gates of Ion Channels are Active Sites Figure adapted from Tilman Schirmer

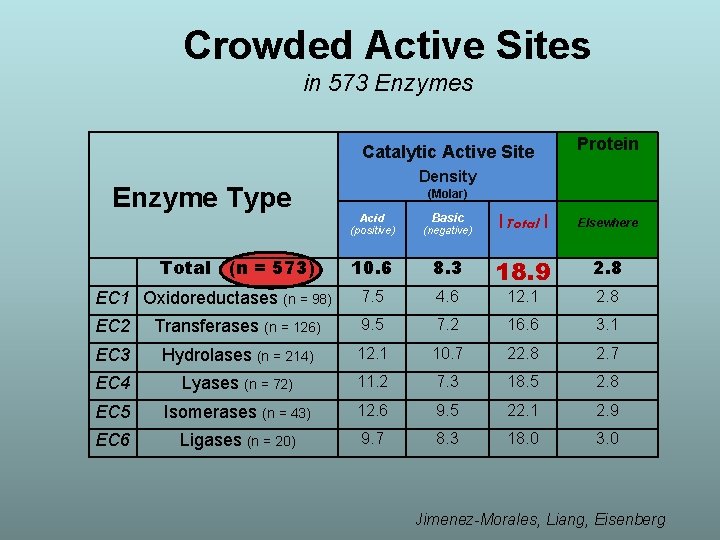
Crowded Active Sites in 573 Enzymes Enzyme Type Catalytic Active Site Density Protein (Molar) Acid (positive) (negative) Basic | Total | Elsewhere 10. 6 8. 3 2. 8 EC 1 Oxidoreductases (n = 98) 7. 5 4. 6 18. 9 12. 1 2. 8 EC 2 Transferases (n = 126) 9. 5 7. 2 16. 6 3. 1 EC 3 Hydrolases (n = 214) 12. 1 10. 7 22. 8 2. 7 EC 4 Lyases (n = 72) 11. 2 7. 3 18. 5 2. 8 EC 5 Isomerases (n = 43) 12. 6 9. 5 22. 1 2. 9 EC 6 Ligases (n = 20) 9. 7 8. 3 18. 0 3. 0 Total (n = 573) Jimenez-Morales, Liang, Eisenberg

MD is calibrated in ZERO concentration. It does not describe mixtures or divalents and so MD cannot deal with Ringer’s solutions or intracellular solutions 30

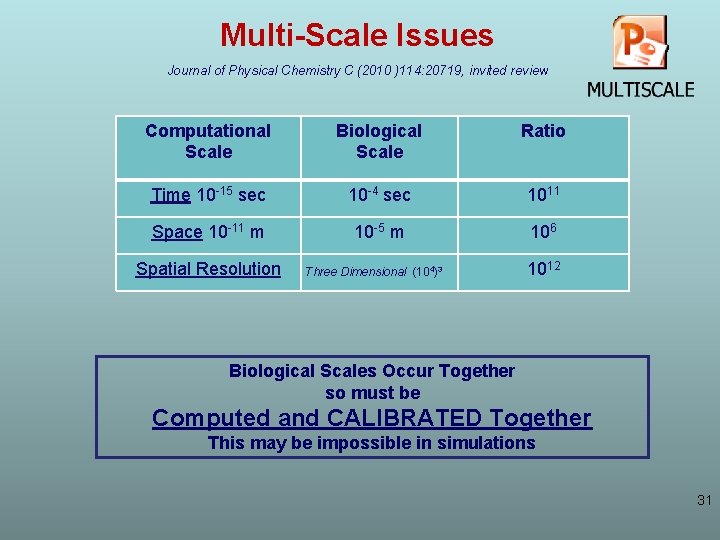
Multi-Scale Issues Journal of Physical Chemistry C (2010 )114: 20719, invited review Computational Scale Biological Scale Ratio Time 10 -15 sec 10 -4 sec 1011 Space 10 -11 m 10 -5 m 106 Spatial Resolution Three Dimensional (104)3 1012 Biological Scales Occur Together so must be Computed and CALIBRATED Together This may be impossible in simulations 31

Time scale of protein structural changes Need for stable structures, preparing the calculation Drift from initial guess Special conditions of crystallization 32

What should be done? Physics as Usual Guess and Check 33

Guess cleverly, but with abandon Don’t think too much Check everything, compulsively or the lithium battery will catch fire Orthogonal Personality Traits Not usually found in one person 34

Biology will become a mature science when it realizes both structure and function are needed across all relevant scales 35

When dealing with theory and simulations, use the same standards of care that you use in the lab! 36

Multi-scale Issues in General

Can Molecular Simulations serve as “Fundamental Physical Laws”? Only if they are calibrated ! 38

Can Molecular Simulations Serve as “Fundamental Physical Laws”? Simulations are Reliable Science when they are Calibrated Simulations are not Mathematics! (e. g. , results depend on numerical procedures and round-off error) 39

Calibration is Hard Work Force Fields must be RE-calibrated in each Biological Solution to verify equilibrium potentials (chemical potentials) Fitting Real Experiments requires Accurate Chemical Potentials in mixtures 2+ like Ringer Solution, with Ca Channels are Identified by Equilibrium Potentials 40

Force Fields are Calibrated Ignoring Interactions with ions but Chemically Specific Properties come from Interactions in Ionic Solutions Life occurs in Interacting Solutions 41

Simulations as fundamental physical laws (? ) First Principle of Numerical Integration The larger the calculation, the more work done, the greater the error First Principle of Experimentation The more work done, the less the error 42

It is very difficult for Molecular Dynamics to count well enough to reproduce Conservation Laws (e. g. , of energy) Concentration (i. e. , number density) or activity Energy of Electric Field Ohm’s ‘law’ (in simple situations) Fick’s ‘law’ (in simple situations) Fluctuations in number density (e. g. , entropy) 43

Simulations produce too many numbers 106 trajectories each 10 -6 sec long, with 109 samples in each trajectory, in background of 1022 atoms 44

Simulations need a theory that Estimates Parameters (e. g. , averages) Theories and Reduced Models are needed to create estimators. Theories and Models are Unavoidable! (in my opinion) 45