Limitations of Antiretroviral Therapy Marshall J Glesby MD

Limitations of Antiretroviral Therapy Marshall J Glesby MD Ph. D Associate Professor of Medicine and Public Health Weill Medical College of Cornell University March 2006

Limitations of Current Antiretrovirals u Adherence u Resistance u Cost u Drug-drug interactions u Side effects 2

Adherence u A major determinant of degree and duration of viral suppression u Poor adherence associated with virologic failure u Optimal suppression requires excellent adherence u Suboptimal adherence is common 3
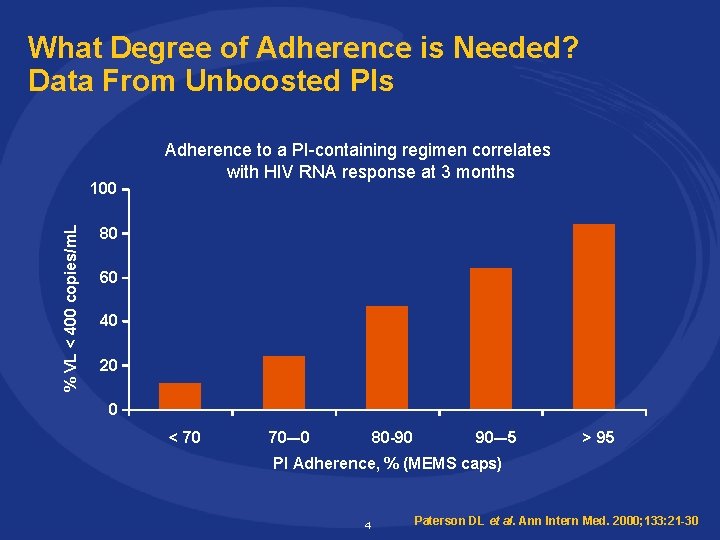
What Degree of Adherence is Needed? Data From Unboosted PIs % VL < 400 copies/m. L 100 Adherence to a PI-containing regimen correlates with HIV RNA response at 3 months 80 60 40 20 0 < 70 70–-0 80 -90 90–-5 > 95 PI Adherence, % (MEMS caps) 4 Paterson DL et al. Ann Intern Med. 2000; 133: 21 -30
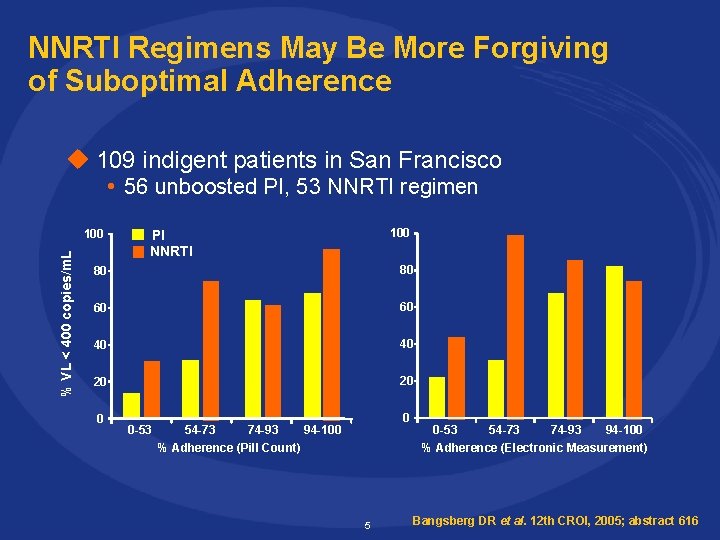
NNRTI Regimens May Be More Forgiving of Suboptimal Adherence u 109 indigent patients in San Francisco • 56 unboosted PI, 53 NNRTI regimen % VL < 400 copies/m. L 100 PI NNRTI 80 80 60 60 40 40 20 20 0 0 -53 0 54 -73 74 -93 94 -100 % Adherence (Pill Count) 5 0 -53 54 -73 74 -93 94 -100 % Adherence (Electronic Measurement) Bangsberg DR et al. 12 th CROI, 2005; abstract 616

Predictors of Inadequate Adherence u u u u Regimen complexity and pill burden Poor clinician-patient relationship Active drug use or alcoholism Unstable housing Mental illness (especially untreated depression) Lack of patient education Medication adverse effects (or fear of them) Not age, race, sex, educational level, socioeconomic status, past history of alcoholism or drug use 6
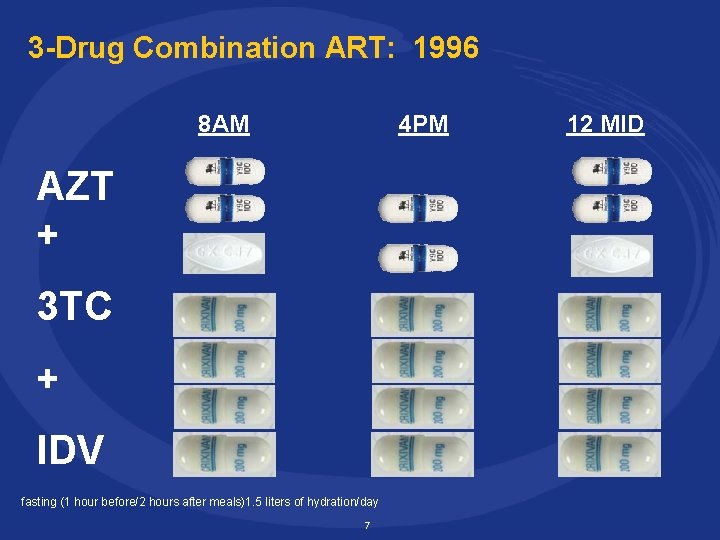
3 -Drug Combination ART: 1996 8 AM 4 PM AZT + 3 TC + IDV fasting (1 hour before/2 hours after meals)1. 5 liters of hydration/day 7 12 MID
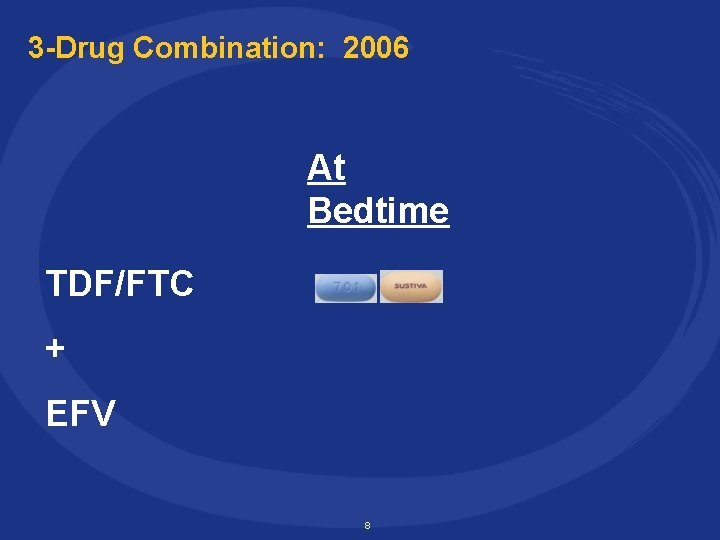
3 -Drug Combination: 2006 At Bedtime TDF/FTC + EFV 8

Improving Adherence u Establish readiness to start therapy u Provide education on medication dosing u Review potential side effects u Anticipate and treat side effects u Utilize educational aids including pictures, pillboxes, and calendars u Individualized adherence programs 9

Limitations of Current Antiretrovirals u Adherence u Resistance u Cost u Drug-drug interactions u Side effects 10

Mutations Occur Spontaneously in the HIV Genome u HIV makes copies of itself very rapidly ~ 1 -10 billion new virus particles/day u During its replication, HIV is prone to make errors when copying itself u This results in mutations or errors in the genetic material of the virus which make the structure of the offspring virus slightly different to that of the parent virus u Some of these mutations will result in an increased ability of the virus to grow in the presence of antiretroviral drugs 11

Partial Viral Suppression Leads to Selection of Resistant Virus u When HIV replication is not blocked completely…. • Sub-optimal therapy regimens (e. g. partially suppressive regimens) • Adherence problems • Pharmacokinetic problems: poor drug absorption, inadequate dosing, drug-drug interactions, interperson differences in PK u …. drug-resistant virus, already present in the population, is selected for and ultimately dominates 12
Drug Levels and Resistance � 1 Increased risk of side effects Drug concentration MEC (Minimum Effective Concentratin) Increased risk of resistance 0 dose 13 dose
Drug Levels and Resistance � 2 Increased risk of side effects Drug concentration MEC (Minimum Effective Concentratin) Increased risk of resistance 0 dose missed dose 14 late dose
CDC Surveillance of Resistance Mutations In Naive Patients u 633 newly diagnosed patients genotyped at 89 sites in 6 states in 2003 -2004 u 14. 5% prevalence of resistance mutations • NRTI, 7. 8% • NNRTI, 3. 0% • PI, 0. 7% • Multiclass, 0. 7% 15 Prevalence (%) 8 7. 8% 6 4 3. 0% 2 0. 7% PI Multi 0 NRTI NNRTI Bennett D et al. 12 th CROI 2005; abstract 674
HAART Observational Medical Evaluation & Research Study u Pts who initiated HAART from 8/96 -9/99 in B. C. u ~25% developed drug resistance mutations during 30 m of f/u u However, with 7 -year f/u of lopinavir/r + d 4 T/3 TC in naïve pts: no d 4 T or LPV resistance; 3% 3 TC resistance 16 Harrigan et al. J Infect Dis 2005; 191: 339 -47 Murphy et al. EACS 2005

Resistance Testing u Genotypic resistance test • Perform test that gives mutations in viral genes u Phenotypic resistance test • Perform test that describes growth of virus in the presence of anti-HIV drugs u Limitations: • Cannot detect minority species (< 10% of viral population) 17
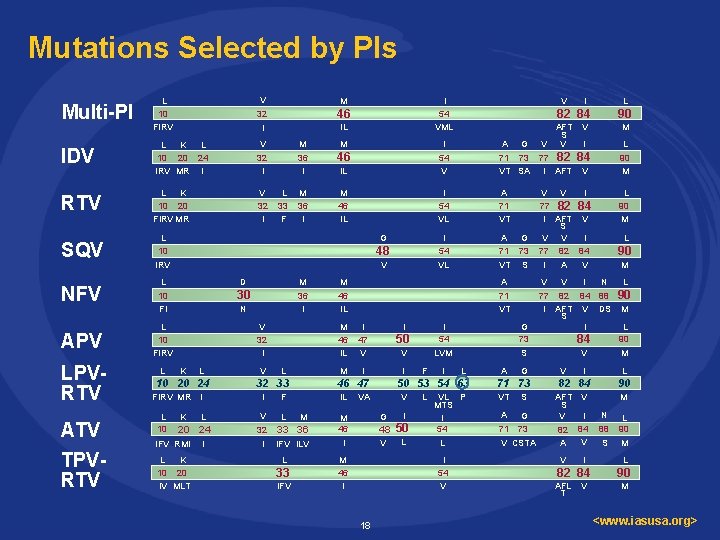
Mutations Selected by PIs Multi-PI L 10 FIRV V M 32 46 I IL IDV L K L 10 20 24 IRV MR I V 32 I RTV L K 10 20 FIRV MR V 32 I SQV IL I 54 V M 46 IL I 54 VL A 71 VT M 36 I M 46 M 36 I I 54 A 71 G 73 IRV V VL VT S APV L 10 FIRV L G D N V 32 I K L 10 20 24 FIRV MR L 10 10 M 36 I 30 K I I L V 20 24 24 20 IFV RMI L K 10 20 IV MLT V I L 32 33 F M 46 IL I 47 V M I IL 32 M 46 I IFV ILV I 33 IFV I F I V G 48 48 V I 50 L L L VL P MTS I 54 L I 54 V 18 G 73 S 50 53 54 63 VA M 46 I I 54 LVM 50 46 47 L M 33 36 L A 71 VT M 46 IL I L 82 84 AFT S A G V V 71 73 77 82 VT SA I AFT 48 NFV ATV TPVRTV V L 10 FI LPVRTV L 33 F I 54 VML A G 90 V M I L 90 M 84 V V V I 77 82 84 I AFT V S V V I 77 82 84 I A L 90 M L 90 V V V I 77 82 84 I AFT V S I M N 88 DS M L 90 M 84 V V L 90 I L 71 73 82 84 90 VT S M A 71 G 73 AFT V S I V 84 82 V A V CSTA V I 82 84 AFL T V N 88 L 90 S M L 90 M <www. iasusa. org>
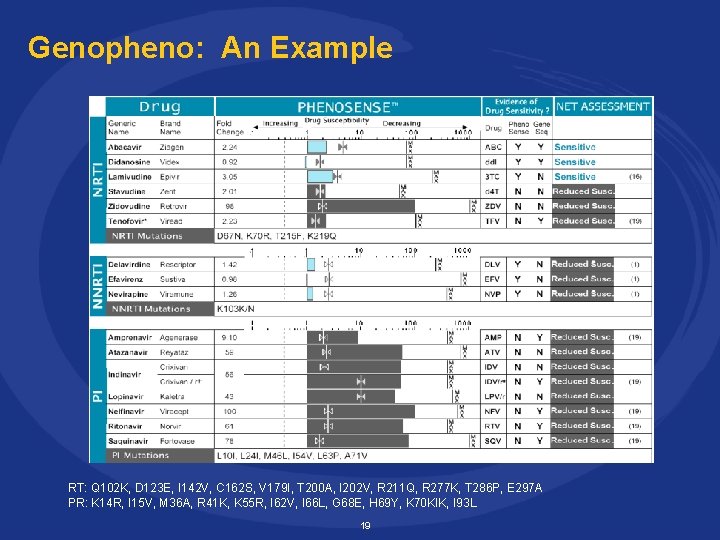
Genopheno: An Example RT: Q 102 K, D 123 E, I 142 V, C 162 S, V 179 I, T 200 A, I 202 V, R 211 Q, R 277 K, T 286 P, E 297 A PR: K 14 R, I 15 V, M 36 A, R 41 K, K 55 R, I 62 V, I 66 L, G 68 E, H 69 Y, K 70 KIK, I 93 L 19

Recommendations for Resistance Tests Clinical Setting Recommended u Virologic failure u Suboptimal virologic suppression u Acute HIV infection Consider u Chronic HIV infection prior to starting ART Not generally recommended u >4 weeks after ART drugs are stopped u Viral load levels <1000 cpm 20 DHHS Guidelines, 4/7/05

Antiretroviral Resistance: Conclusions u HIV growth leads to diversity. u Not suppressing viral load levels in the presence of antiretroviral drugs leads to resistant virus. u HIV drugs have unique resistance patterns, but cross-resistance may occur. u Resistance testing offers benefits in choosing the next drug combination. 21

Limitations of Current Antiretrovirals u Adherence u Resistance u Cost u Drug-drug interactions u Side effects 22

Metabolism of PIs/NNRTIs u Metabolized by cytochrome P 450, especially CYP 3 A 4 u Levels of PIs and NNRTIs may be affected by concurrently administered drugs u PIs, especially ritonavir, inhibit CYP 3 A 4 potentially leading to increased levels of concurrently administered drugs u Efavirenz and nevirapine can induce and inhibit CYP 3 A 4 u Fewer drug-drug interactions with NRTIs 23

Drug Interactions with ARVs: Dose Modification or Cautious Use u Oral contraceptives (may require second method) u Methadone u Erectile dysfunction agents u Herbs - St. John’s wort u Lipid-lowering agents u Anti-mycobacterials, especially rifampin u Psychotropics – midazolam, triazolam u Ergot Alkaloids u Antihistamines – astemizole u Anticonvulsants 24

Limitations of Current Antiretrovirals u Adherence u Resistance u Cost u Drug-drug interactions u Side effects 25
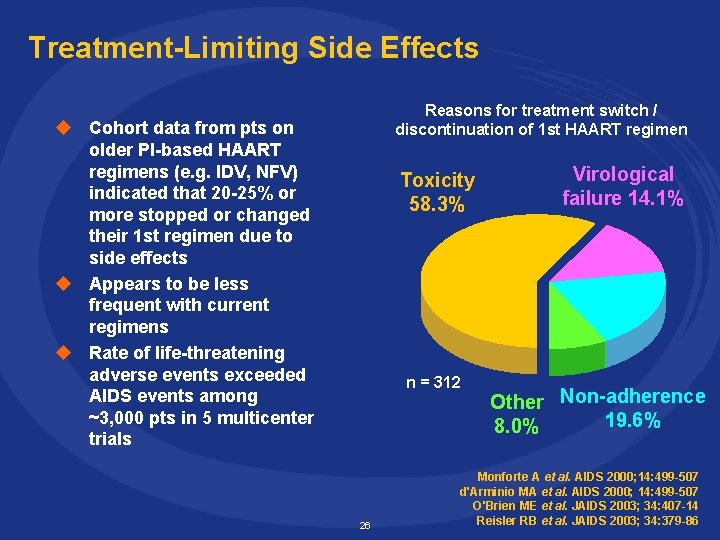
Treatment-Limiting Side Effects Reasons for treatment switch / discontinuation of 1 st HAART regimen u Cohort data from pts on older PI-based HAART regimens (e. g. IDV, NFV) indicated that 20 -25% or more stopped or changed their 1 st regimen due to side effects u Appears to be less frequent with current regimens u Rate of life-threatening adverse events exceeded AIDS events among ~3, 000 pts in 5 multicenter trials Toxicity 58. 3% n = 312 26 Virological failure 14. 1% Other Non-adherence 19. 6% 8. 0% Monforte A et al. AIDS 2000; 14: 499 -507 d'Arminio MA et al. AIDS 2000; 14: 499 -507 O'Brien ME et al. JAIDS 2003; 34: 407 -14 Reisler RB et al. JAIDS 2003; 34: 379 -86

Adverse Effects of NRTIs* u Zidovudine (AZT)- headache, GI intolerance, bone marrow suppression u Abacavir - hypersensitivity reaction u Didanosine (dd. I) - GI intolerance, pancreatitis, peripheral neuropathy u Stavudine (d 4 T) - peripheral neuropathy, pancreatitis, lipoatrophy u Zalcitabine (dd. C) - peripheral neuropathy, oral ulcers u Lamivudine (3 TC) – rare side effects u Emtricitabine (FTC) – side effects uncommon; hyperpigmentation of palms/soles < 2% (non-Whites) u Tenofovir - headache, GI intolerance, renal insufficiency *Lactic acidosis is a class effect, most strongly associated with d 4 T/dd. I; 3 TC, FTC, and tenofovir are active against HBV. Development of HBV resistance may lead to flare of hepatitis. 27

Adverse Effects of NNRTIs u Rash, including Stevens-Johnson syndrome with nevirapine u Elevated liver enzymes (nevirapine > efavirenz, delavirdine) • Incidence of hepatotoxicity highest in women with pre-nevirapine CD 4 counts >250 cells/mm 3 and men with >400 cells/mm 3 u Efavirenz - neuropsychiatric, teratogenic in primates (FDA Pregnancy Class D) 28

Acute Adverse Effects of PIs u GI intolerance, diarrhea u Hyperbilirubinemia –atazanavir, indinavir u Hepatotoxicity u Increased bleeding in hemophiliacs u Adverse metabolic effects • Dyslipidemia • Insulin resistance • ? Lipodystrophy/fat redistribution • Atazanavir has favorable metabolic profile 29

Adverse Effects of Entry Inhibitors u Enfuvirtide (T-20) • Injection-site reactions • Hypersensitivity reaction • Increased incidence of bacterial pneumonia 30
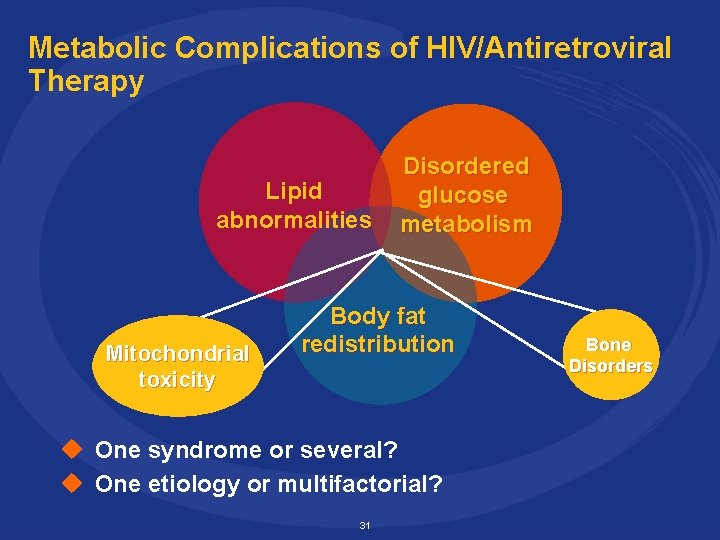
Metabolic Complications of HIV/Antiretroviral Therapy Lipid abnormalities Mitochondrial toxicity Disordered glucose metabolism Body fat redistribution u One syndrome or several? u One etiology or multifactorial? 31 Bone Disorders
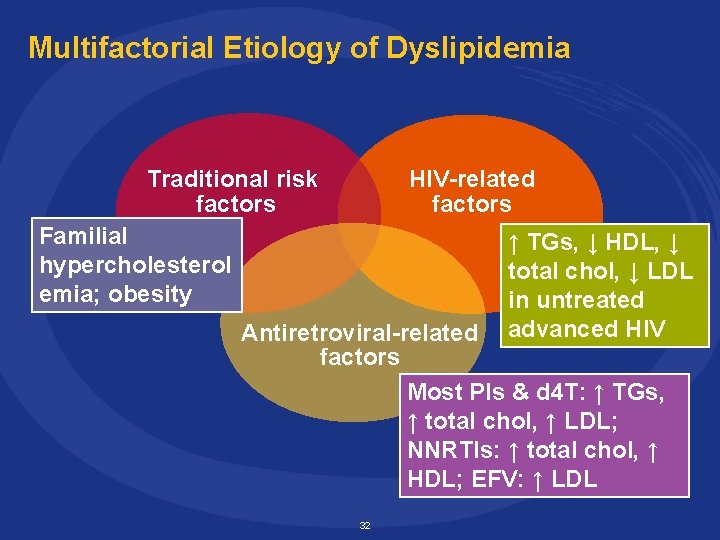
Multifactorial Etiology of Dyslipidemia Traditional risk factors HIV-related factors Familial hypercholesterol emia; obesity ↑ TGs, ↓ HDL, ↓ total chol, ↓ LDL in untreated advanced HIV Antiretroviral-related factors Most PIs & d 4 T: ↑ TGs, ↑ total chol, ↑ LDL; NNRTIs: ↑ total chol, ↑ HDL; EFV: ↑ LDL 32
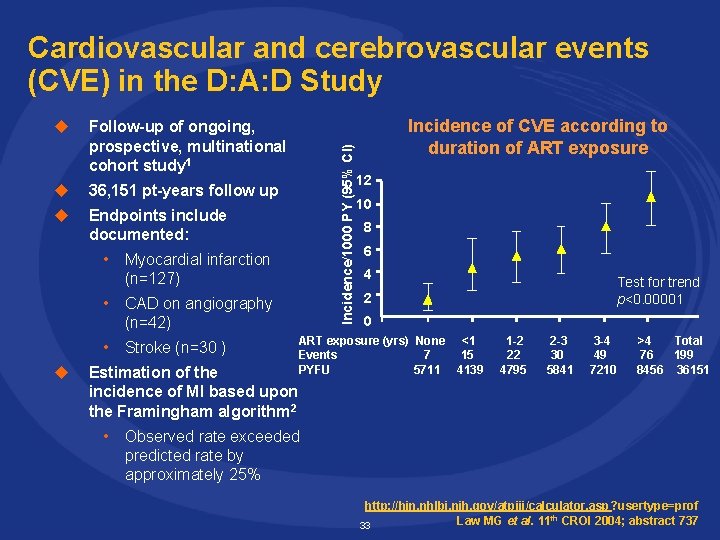
u Follow-up of ongoing, prospective, multinational cohort study 1 u 36, 151 pt-years follow up u Endpoints include documented: Incidence/1000 PY (95% Cl) Cardiovascular and cerebrovascular events (CVE) in the D: A: D Study • Myocardial infarction (n=127) • CAD on angiography (n=42) • Stroke (n=30 ) u Estimation of the incidence of MI based upon the Framingham algorithm 2 Incidence of CVE according to duration of ART exposure 12 10 8 6 4 Test for trend p<0. 00001 2 0 ART exposure (yrs) None <1 Events 7 15 PYFU 5711 4139 1 -2 22 4795 2 -3 30 5841 3 -4 49 7210 >4 Total 76 199 8456 36151 • Observed rate exceeded predicted rate by approximately 25% http: //hin. nhlbi. nih. gov/atpiii/calculator. asp ? usertype=prof Law MG et al. 11 th CROI 2004; abstract 737 33

Disordered Glucose Metabolism u Prevalence of diabetes mellitus increased among HIV+ pts on protease inhibitors • Prevalence ~2 -14% u Insulin resistance (higher concentrations of insulin required for usual effects) more common u MACS: Risk of new onset DM ~ 4 x higher in HIV+ men vs. HIV- men (adjusted for age, BMI) 34 Dube M Clin Infect Dis 2000; 31: 1467 -75 Brown TT et al. Arch Intern Med 2005; 165: 1179 -84
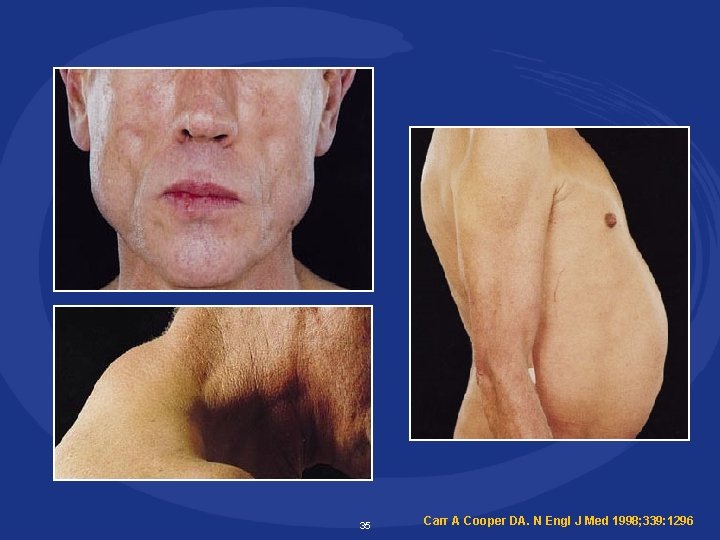
35 Carr A Cooper DA. N Engl J Med 1998; 339: 1296
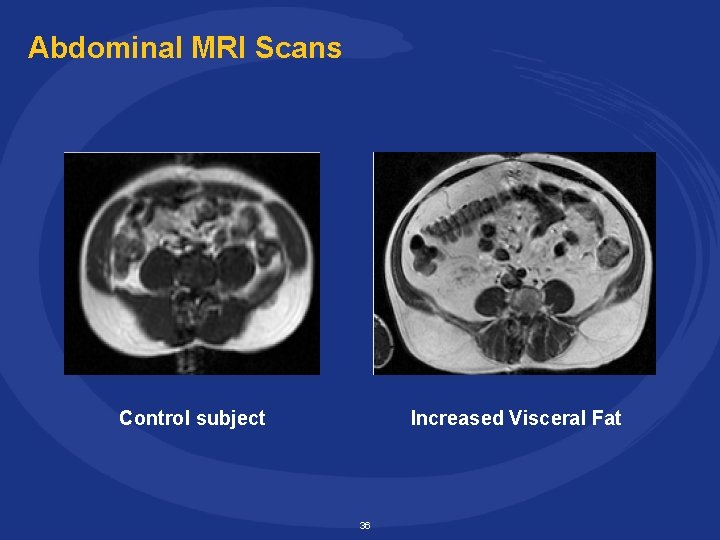
Abdominal MRI Scans Control subject Increased Visceral Fat 36

“Lipodystrophy Syndrome” u No generally accepted case definition of syndrome(s) u Initial reports suggested clustering of: • Central fat accumulation/adiposity • Lipoatrophy/fat wasting • Dyslipidemia • Insulin resistance/type 2 diabetes mellitus u Recent cross-sectional epidemiological data question linkage of lipoatrophy and fat accumulation 37 Fram J Acquir Immune Defic Syndr 2005; 40: 121 -131
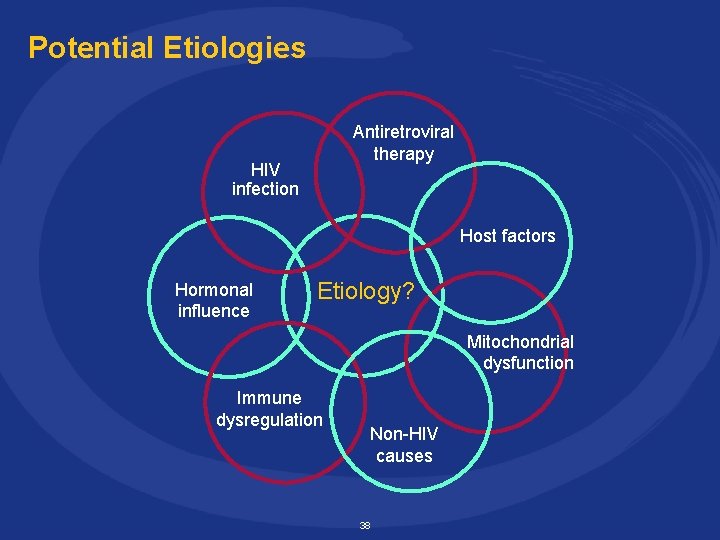
Potential Etiologies Antiretroviral therapy HIV infection Host factors Hormonal influence Etiology? Mitochondrial dysfunction Immune dysregulation Non-HIV causes 38
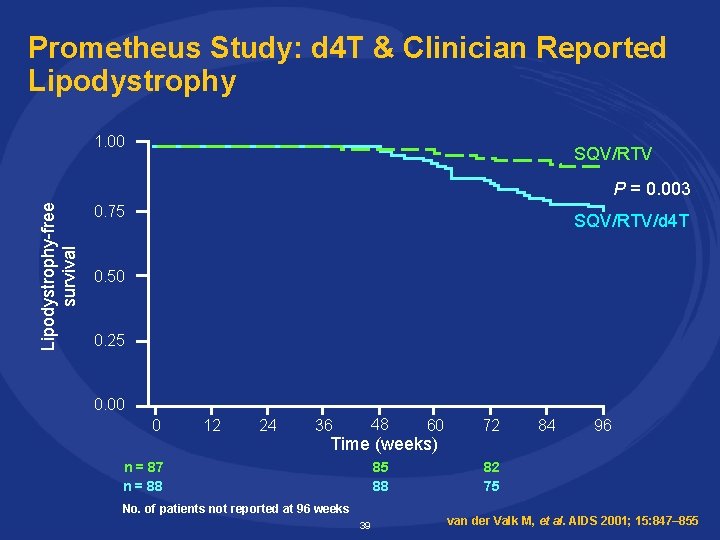
Prometheus Study: d 4 T & Clinician Reported Lipodystrophy 1. 00 SQV/RTV Lipodystrophy-free survival P = 0. 003 0. 75 SQV/RTV/d 4 T 0. 50 0. 25 0. 00 0 12 24 36 48 60 Time (weeks) n = 87 n = 88 85 88 No. of patients not reported at 96 weeks 39 72 84 96 82 75 van der Valk M, et al. AIDS 2001; 15: 847– 855
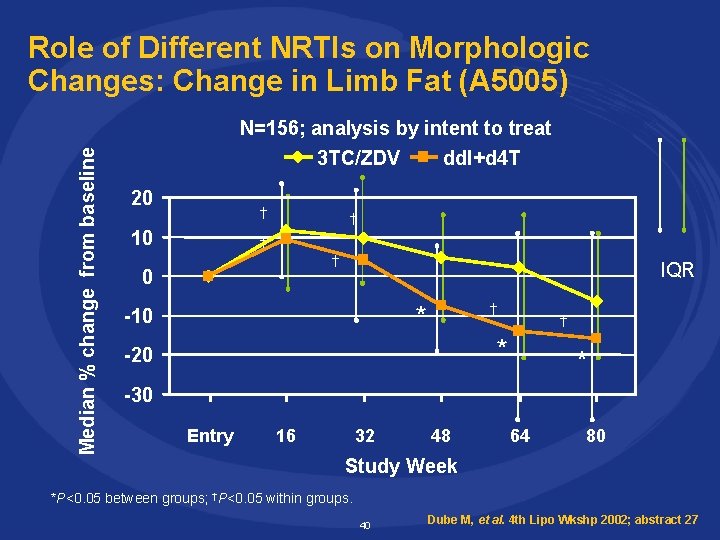
Role of Different NRTIs on Morphologic Changes: Change in Limb Fat (A 5005) Median % change from baseline N=156; analysis by intent to treat 3 TC/ZDV 20 † 10 dd. I+d 4 T † † † 0 IQR † * -10 † * -20 * -30 Entry 16 32 48 64 80 Study Week *P<0. 05 between groups; †P<0. 05 within groups. 40 Dube M, et al. 4 th Lipo Wkshp 2002; abstract 27

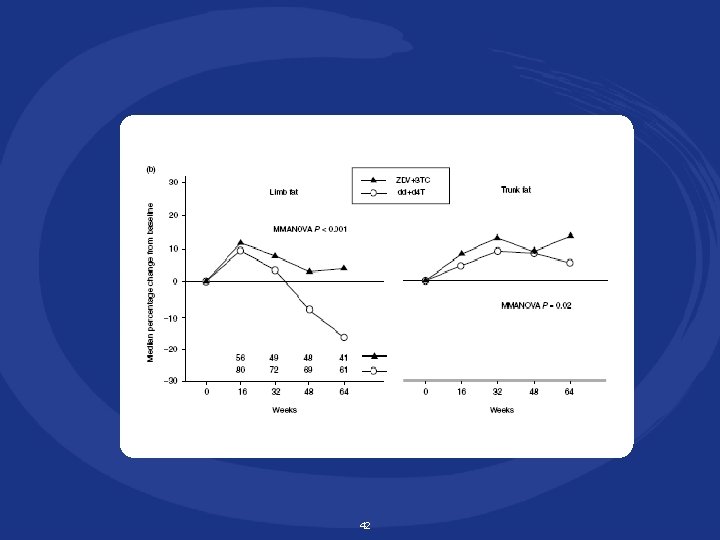
41 Dube MP et al. AIDS 2005; 19: 1807 -18
42
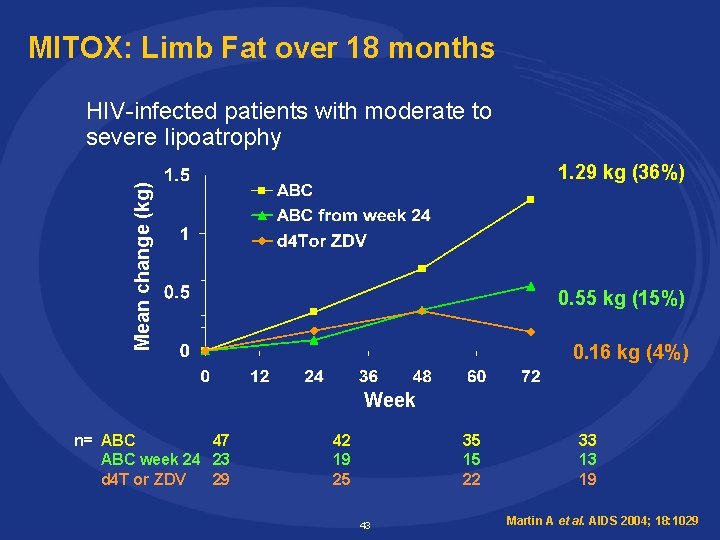
MITOX: Limb Fat over 18 months HIV-infected patients with moderate to severe lipoatrophy Mean change (kg) 1. 29 kg (36%) 0. 55 kg (15%) 0. 16 kg (4%) Week n= ABC 47 ABC week 24 23 d 4 T or ZDV 29 42 19 25 35 15 22 43 33 13 19 Martin A et al. AIDS 2004; 18: 1029
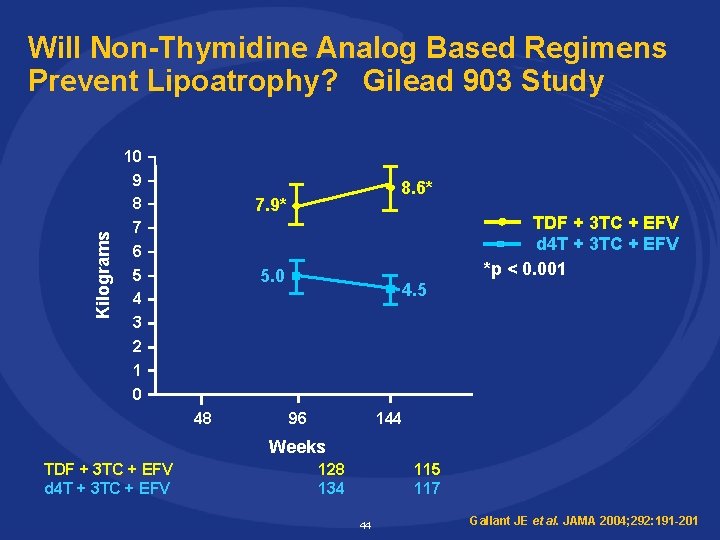
Kilograms Will Non-Thymidine Analog Based Regimens Prevent Lipoatrophy? Gilead 903 Study 10 9 8 7 8. 6* 7. 9* 6 5 4 3 2 1 0 TDF + 3 TC + EFV d 4 T + 3 TC + EFV *p < 0. 001 5. 0 48 4. 5 96 144 Weeks TDF + 3 TC + EFV d 4 T + 3 TC + EFV 128 134 115 117 44 Gallant JE et al. JAMA 2004; 292: 191 -201

Cardiovascular Risk Factors in Lipodystrophy u Compared with age and BMI matched controls from the Framingham Offspring Study, HIV+ pts with self-reported lipodystrophy had: • • • higher prevalence of impaired glucose tolerance/diabetes higher diastolic blood pressure elevated triglycerides, total cholesterol (not LDL-C) lower HDL-C increased PAI-1 and t. PA (markers of impaired fibrinolysis-- ability to break down blood clots ) u Some pts with lipodystrophy appear to have a metabolic syndrome that theoretically could predispose to accelerated atherosclerosis and diabetes 45 Hadigan et al, Clin Infect Dis 2001; 32: 130 Hadigan et al, JCEM 2001; 86: 939 -43

Osteopenia/Osteoporosis u Decreased bone mineral density (BMD) initially reported in HIV+ on PIs (plus NRTIs) u Subsequent reports of higher prevalence of decreased BMD in ARV naïve pts and increases in BMD while on PI-containing HAART u Multifactorial etiology: HIV, cytokines, endocrine factors, liver disease, smoking, ? antiretrovirals 46 Tebas P et al, AIDS 2000; 14: F 63 -7 Mondy K et al, Clin Infect Dis 2003 ; 36: 482 -90
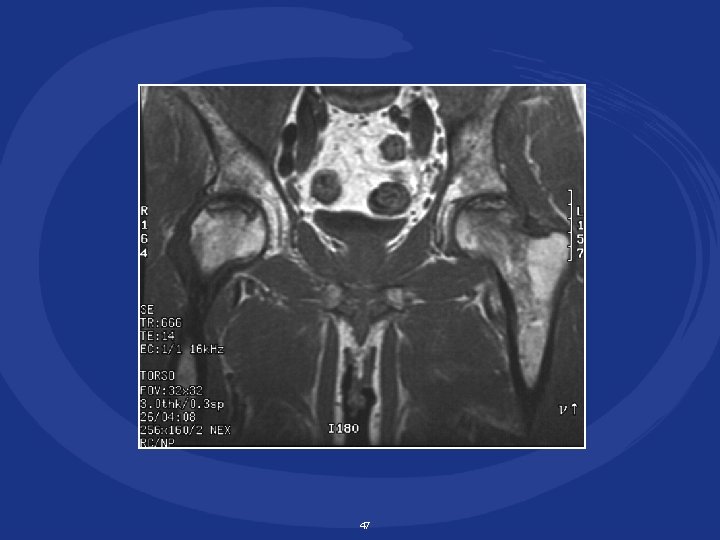
47

Osteonecrosis u Avascular necrosis = aseptic necrosis = osteonecrosis • death of cellular constituents of bone & marrow due to ischemia (decreased blood supply) u Associated with corticosteroid use, possibly duration of antiretroviral therapy & immune recovery u Most commonly presents as hip pain u MRI is test of choice if symptoms suggest dx u Conservative management for early stages of disease u Surgery • total hip replacement vs. core decompression 48 Allison et al, AIDS 2003; 17: 1 -9

Conclusions u Adherence, resistance, drug-drug interactions, and side effects (short- and long-term) are important limitations of antiretroviral therapy u Regimen choices usually based on potential advantages/options • Decreased dosing frequency and pill burden • Tolerability • Pharmacokinetic profiles • Resistance considerations • Improved metabolic profiles 49
- Slides: 49