LIMIT AMI LImitation of Myocardial Injury following Thrombolysis

LIMIT AMI (LImitation of Myocardial Injury following Thrombolysis in Acute Myocardial Infarction) An angiographic safety and efficacy trial of a novel anti-CD 18 therapy in acute myocardial infarction in conjunction with thrombolysis Kenneth W. Baran MD for the LIMIT AMI Investigators St. Paul Heart Clinic, St. Paul, MN, USA Sponsor: Genentech Inc. , South San Francisco, USA

Baran KW August 28, 2000
![rhu. MAb CD 18 Antibody fragment [F(ab’)2] â Single dose administered as IV bolus rhu. MAb CD 18 Antibody fragment [F(ab’)2] â Single dose administered as IV bolus](http://slidetodoc.com/presentation_image/c8c37a67cc880d7dce5db1087869aa5f/image-3.jpg)
rhu. MAb CD 18 Antibody fragment [F(ab’)2] â Single dose administered as IV bolus â Plasma half-life 7 to 10 hours â 90% CD 18 -containing receptor saturation maintained for ~ 24 hours (0. 5 mg/kg) and ~ 48 hours (2. 0 mg/kg) â Baran KW August 28, 2000

The LIMIT AMI Trial of rhu. MAb CD 18 Screening • ST elevation > 2 leads • Symptoms < 12 hrs • No prior coronary surgery • No cardiogenic shock • No thrombolytic exclusions • No major immune-related risks

The LIMIT AMI Trial of rhu. MAb CD 18 Screening • ST elevation > 2 leads • Symptoms < 12 hrs • No prior coronary surgery • No cardiogenic shock • No thrombolytic exclusions • No major immune-related risks Randomization 1: 1: 1 with administration prior to full-dose t. PA 2 mg/kg 0. 5 mg/kg Placebo + usual care including aspirin, IV heparin

The LIMIT AMI Trial of rhu. MAb CD 18 Screening • ST elevation > 2 leads • Symptoms < 12 hrs • No prior coronary surgery • No cardiogenic shock • No thrombolytic exclusions • No major immune-related risks Randomization 1: 1: 1 to study drug or placebo then full-dose t. PA 2 mg/kg 0. 5 mg/kg Placebo + usual care including aspirin, IV heparin Coronary angiography 90 minutes from t. PA initiation • Corrected TIMI Frame Count, TIMI Flow Grade • TIMI Myocardial Perfusion Grade • Angioplasty ± Gp IIb/IIIa inhibitors as required

The LIMIT AMI Trial of rhu. MAb CD 18 Other endpoints • Infarct size • sestamibi at rest, day 6 -9 • CKMB 0 -72 hours • ECG ST segment resolution • 180 minutes • Fever • Antibiotic use • General Safety

Statistical Analysis 1. 2. 3. 4. 5. Efficacy endpoint data analyzed by independent and blinded Core Laboratories Time windows were used for efficacy endpoints using imputation for missing data where possible “As treated” analysis of the evaluable cohort Sensitivity analyses p values: non parametric tests Baran KW August 28, 2000

Results â â Enrollment from September 1998 to March 2000 413 subjects randomized 394 treated with rhu. MAb CD 18 or placebo 19 randomized but not treated Ý mainly due to review of exclusion criteria â 60 centers in US and Canada Baran KW August 28, 2000
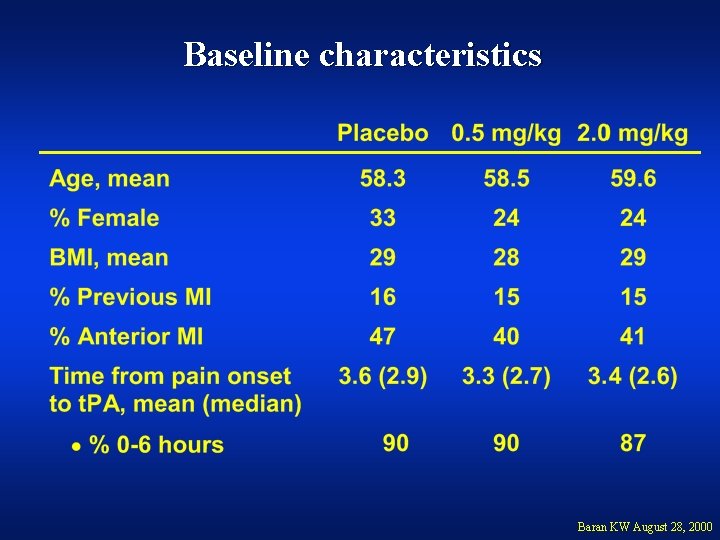
Baseline characteristics Baran KW August 28, 2000
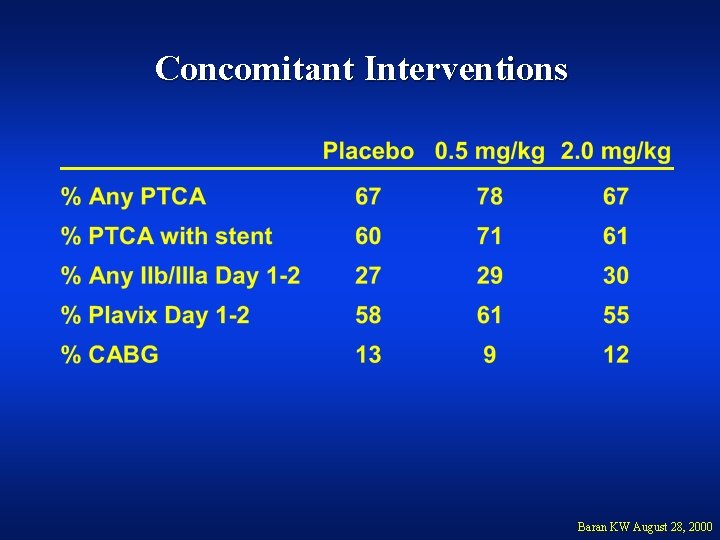
Concomitant Interventions Baran KW August 28, 2000
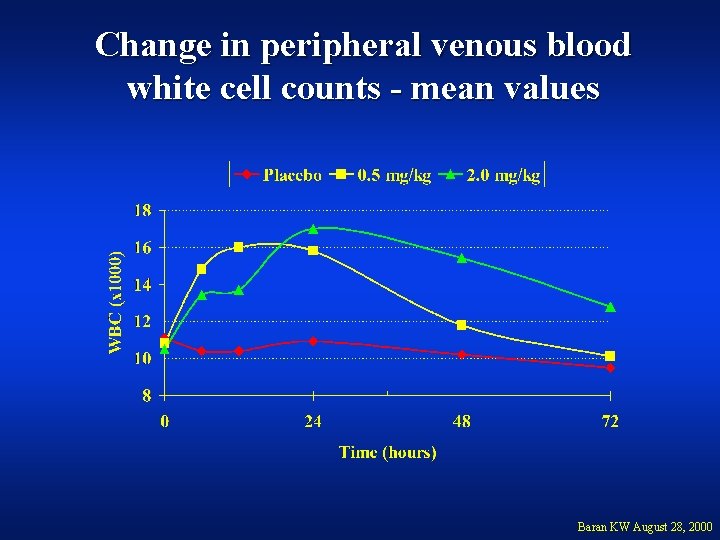
Change in peripheral venous blood white cell counts - mean values Baran KW August 28, 2000
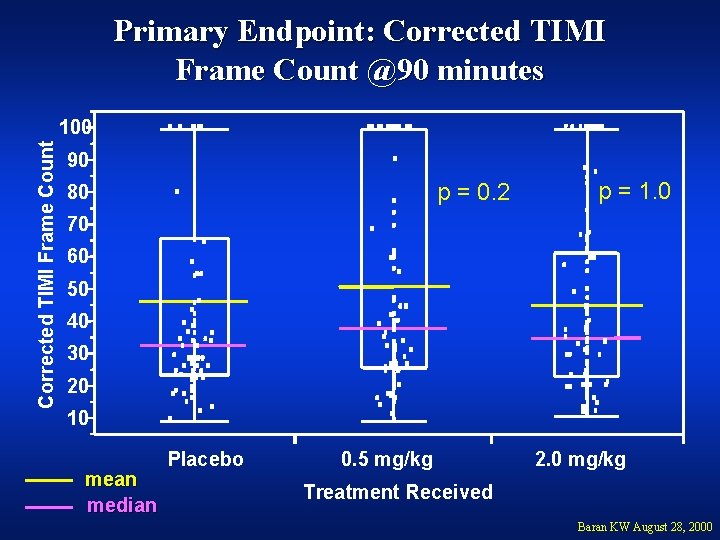
Primary Endpoint: Corrected TIMI Frame Count @90 minutes Corrected TIMI Frame Count 100 90 p = 0. 2 80 p = 1. 0 70 60 50 40 30 20 10 mean median Placebo 0. 5 mg/kg 2. 0 mg/kg Treatment Received Baran KW August 28, 2000
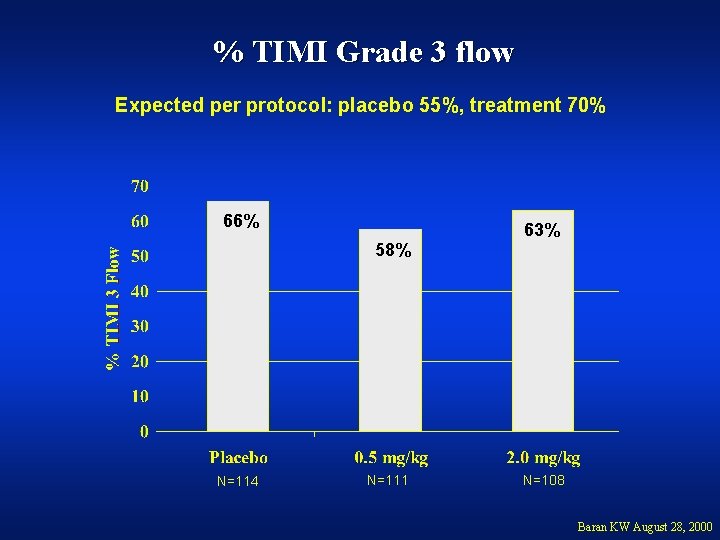
% TIMI Grade 3 flow Expected per protocol: placebo 55%, treatment 70% 66% 58% N=114 N=111 63% N=108 Baran KW August 28, 2000
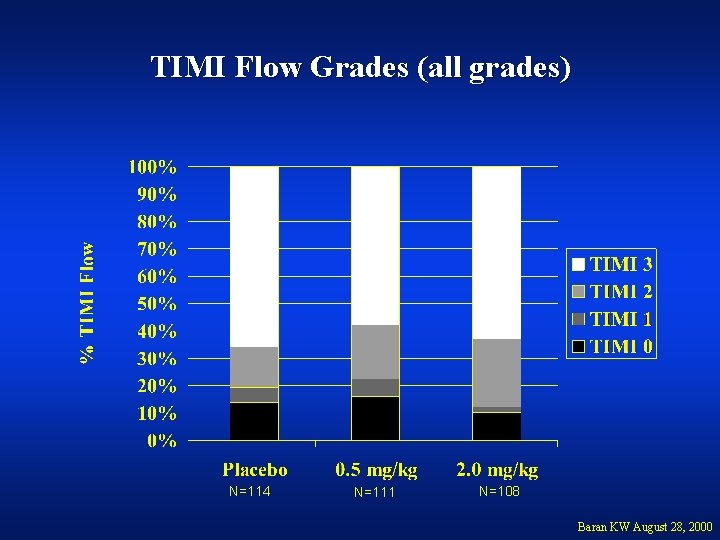
TIMI Flow Grades (all grades) N=114 N=111 N=108 Baran KW August 28, 2000
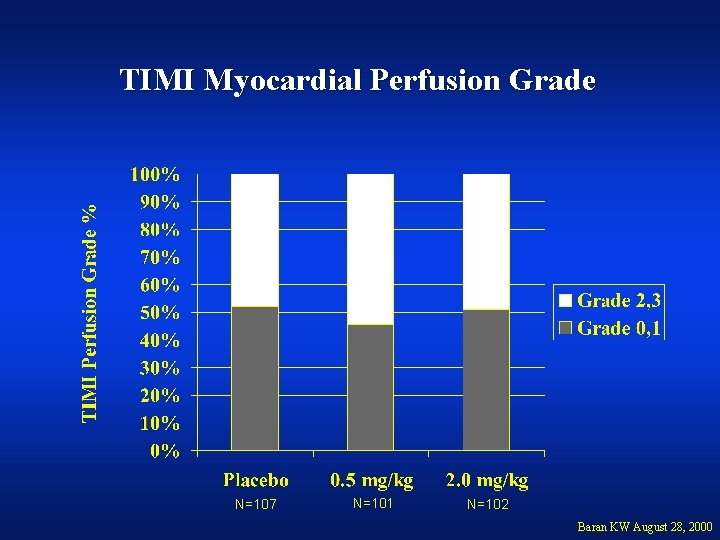
TIMI Myocardial Perfusion Grade N=107 N=101 N=102 Baran KW August 28, 2000
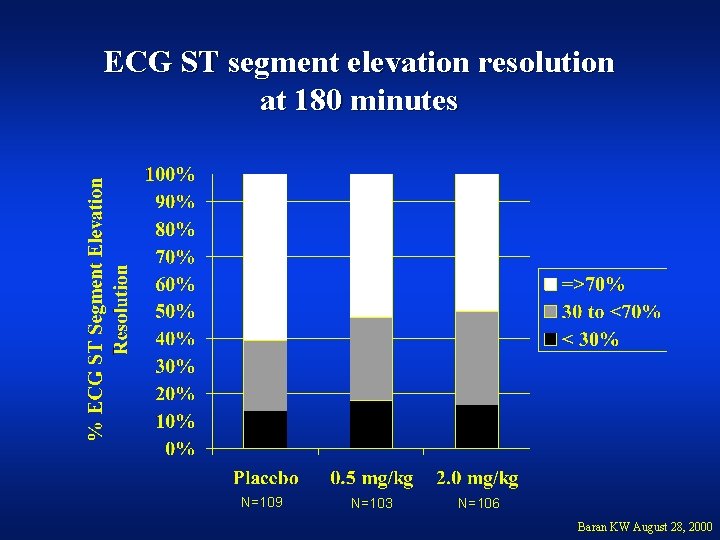
ECG ST segment elevation resolution at 180 minutes N=109 N=103 N=106 Baran KW August 28, 2000
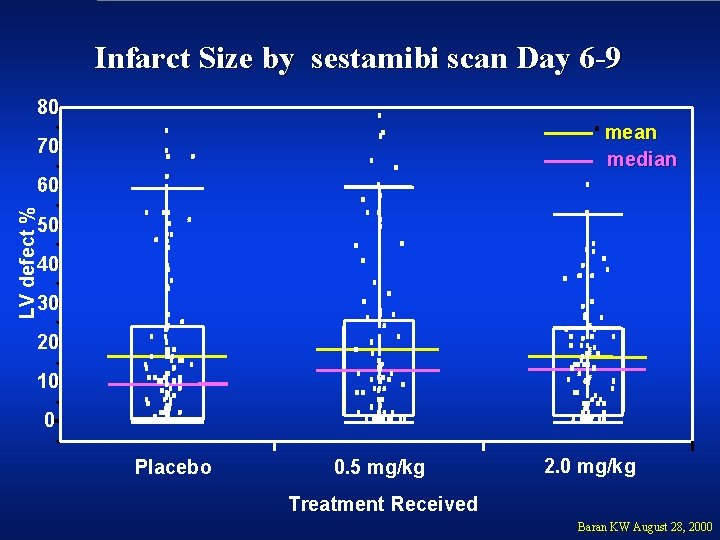
Infarct Size by sestamibi scan Day 6 -9 80 mean median 70 LV defect % 60 50 40 30 20 10 0 Placebo 0. 5 mg/kg 2. 0 mg/kg Treatment Received Baran KW August 28, 2000
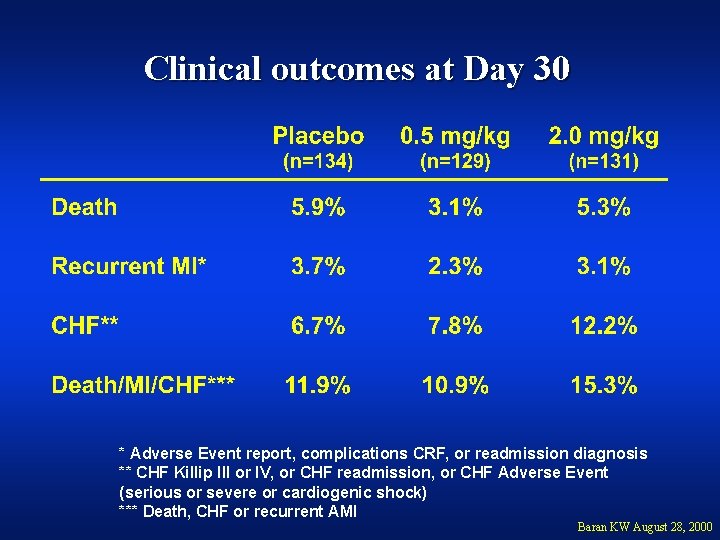
Clinical outcomes at Day 30 * Adverse Event report, complications CRF, or readmission diagnosis ** CHF Killip III or IV, or CHF readmission, or CHF Adverse Event (serious or severe or cardiogenic shock) *** Death, CHF or recurrent AMI Baran KW August 28, 2000
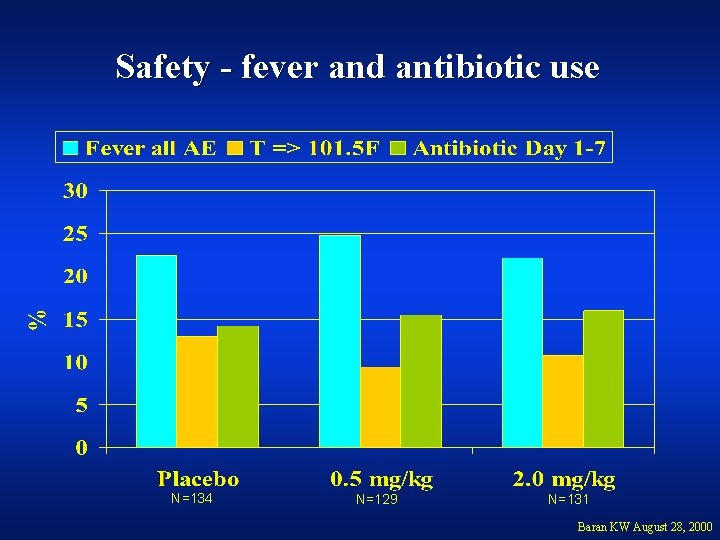
Safety - fever and antibiotic use N=134 N=129 N=131 Baran KW August 28, 2000
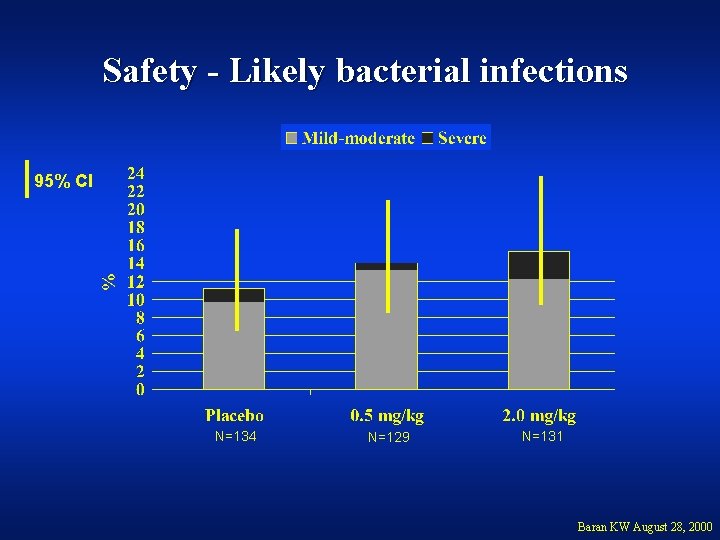
Safety - Likely bacterial infections 95% CI N=134 N=129 N=131 Baran KW August 28, 2000

General Safety 1. 2. 3. 4. 5. No effect on fibrinogen or d-dimer levels No increase in “serious and life-threatening bleeding” Slight trend towards increases in overall bleeding adverse events ( 10%) and transfusions ( 20%) Small number of mild thrombocytopenia cases < 100, 000/mm 3: placebo 2 cases, 0. 5 mg/kg 1 case, 2. 0 mg/kg 5 cases No specific anti-rhu. MAb CD 18 antibody formation* * Except for one 30 Day sample with possible low level anti-CDR activity still under investigation. Baran KW August 28, 2000

Conclusions â rhu. MAb CD 18, in conjunction with thrombolysis, had no effect on coronary flow, infarct size or ECG ST segment resolution â These results are consistent with the findings of another Phase II study of an anti-CD 18 antibody in Primary Angioplasty on infarct size (HALT MI*) â There may be non CD 18 - dependent leukocyte binding and transmigration in AMI â Leukocytes may not be key to ischemiareperfusion injury in human AMI * Faxon D, Annual Meeting of the American Heart Association, Atlanta, November 1999 Baran KW August 28, 2000
- Slides: 23