LIME SOFTENING STABILIZATION Murder Parties Ralph Rottingrape Marilyn

LIME SOFTENING & STABILIZATION

Murder Parties Ralph Rottingrape Marilyn Merlot George W. Taliban

Queen Illicibeth; Gridlock, the Merchant of Tennis Pen Ta Gon Advisor to the Emperor

Classifying Water Hardness Description of water Hardness expressed as mg/L of Ca. CO 3 Soft less than 50 Moderately soft 50– 75 Slightly hard 75– 150 Hard 150– 300 Very hard greater than 300

What is “lime”? • Limestone, mainly calcium carbonate, Ca. CO 3 • Quicklime, calcium oxide, Ca. O • Slaked lime, calcium hydroxide, Ca(OH)2

Limestone mining, preparation and use

Limestone Mining, cont’d

Limestone Mining, cont’d

Limestone Handling and Shipping

Lime Conversions • Limestone can be converted to quick lime: Ca. CO 3 Ca. O + CO 2 (1800 F) • Quick lime is slaked with water: Ca. O + H 2 O Ca(OH)2 • Slaked lime slowly reacts with CO 2 in air: …Ca(OH)2 + CO 2 Ca. CO 3 + H 2 O • Dissolved lime (hardness) can precipitate: Ca(HCO 3)2 Ca. CO 3 + H 2 O + CO 2

Lime Kilns

Calcination of Limestone Ca. CO 3 Ca. O + CO 2

Hydration: slaking with water Water reacts with the quicklime to form hydrated lime: Ca. O + H 2 O Ca(OH)2

The removal of divalent metal ions Precipitation/settling or IX resins
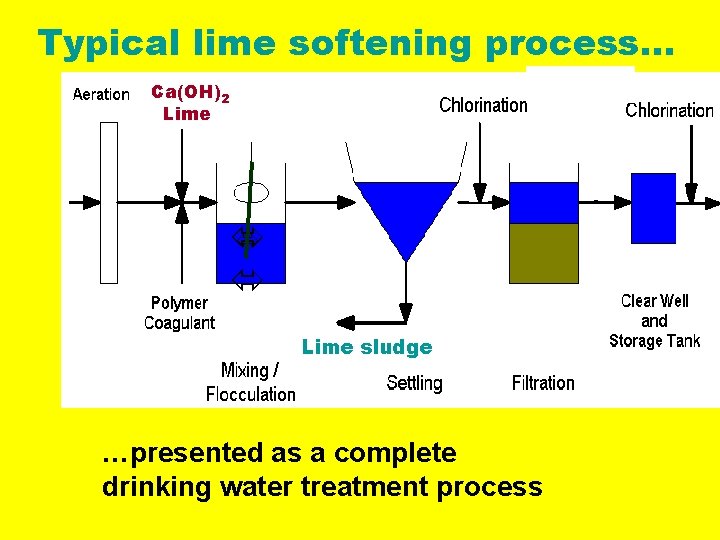
Typical lime softening process… Ca(OH)2 Lime sludge …presented as a complete drinking water treatment process

LIME SLAKING …and DOSING

Precipitation occurs in a settling tank

LAGOON DEWATERING

Why are we concerned about corrosion?

Stability Relationship • The basic Relationship in Stabilization Has been simplified by Powell, Bacon and Lill to p. Hs = p(Ca 2+) + p(Alk) + C
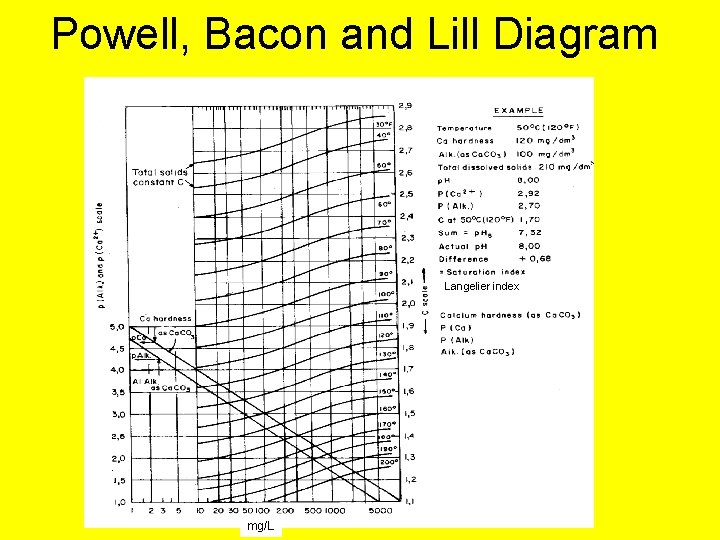
Powell, Bacon and Lill Diagram Langelier index mg/L
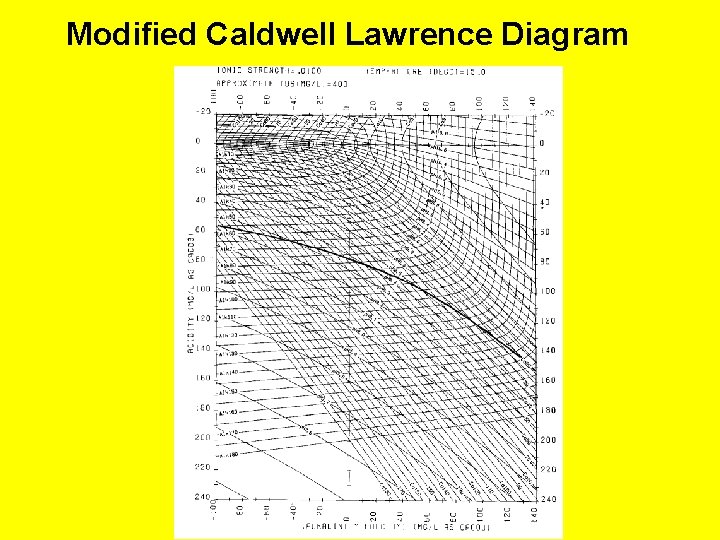
Modified Caldwell Lawrence Diagram
- Slides: 23