lignin Dissolved Organic Matter lignin OH O Me










- Slides: 44

lignin Dissolved Organic Matter lignin OH O Me O OH lignin OH O O Me O OMe OH O OMe HO O O Me lignin Slides and figures from Mark Williams, Diane Mc. Knight, Bailey Simone, Rose Cory, Matt Miller, Rachel Gabor, Eran Hood

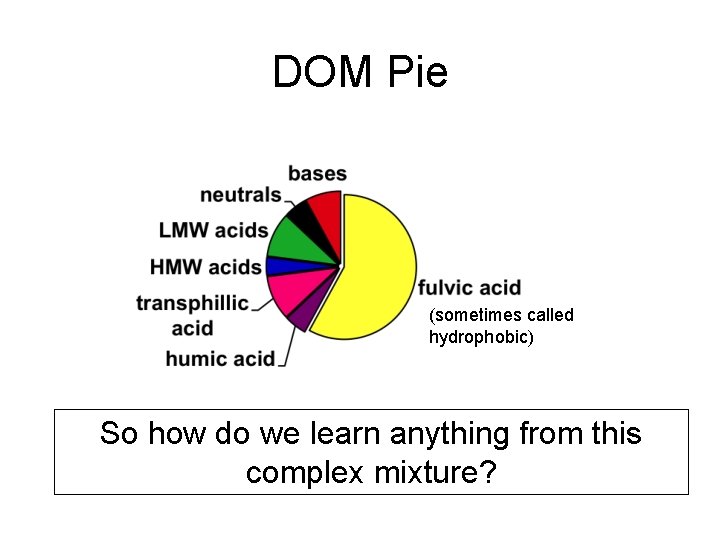
• • • Complex, heterogeneous mixture Truly ubiquitous (ALL waters) Colored (gives water its color) Moderate molecular weight Mass Distribution: – – – 50% C 35 -40% O 4 -6% H 1 -6% N <1 -3%S

Why DOM Matters • Light absorbing DOM can act as sunscreen and control depth of photic zone • Some DOM is bio-available and contains nutrients • Organic acid portion of DOM can buffer p. H • Some DOM can act as metal complexing agents (fate & transport and bioavailability of metals) • Hydrophobic organic pollutants can partition into some portion of DOM • Some DOM can act as electron acceptors and donors • Reducer and oxidizer • Some DOM can “sensitize” the photochemical breakdown of pollutants • Reactions with some DOM can produce toxic disinfection byproducts during drinking water treatment

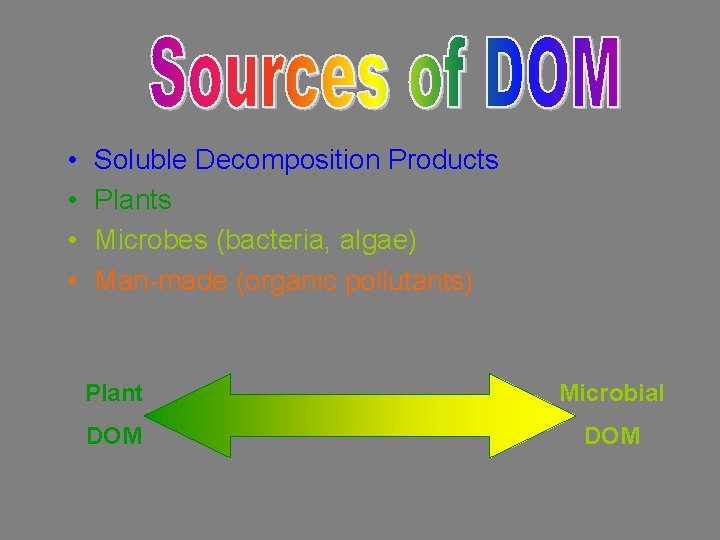
• • Soluble Decomposition Products Plants Microbes (bacteria, algae) Man-made (organic pollutants) Plant Microbial DOM

DOM Pie (sometimes called hydrophobic) So how do we learn anything from this complex mixture?

CHEMISTRY!

Chemistry is all about electrons

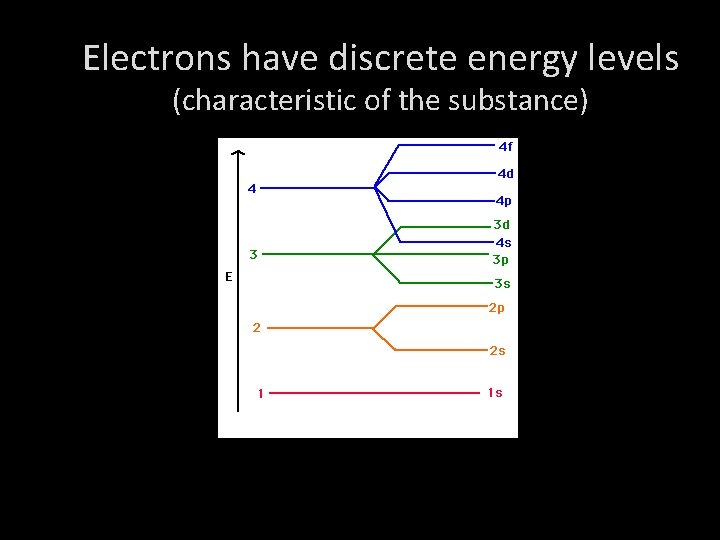
Electrons have discrete energy levels (characteristic of the substance)

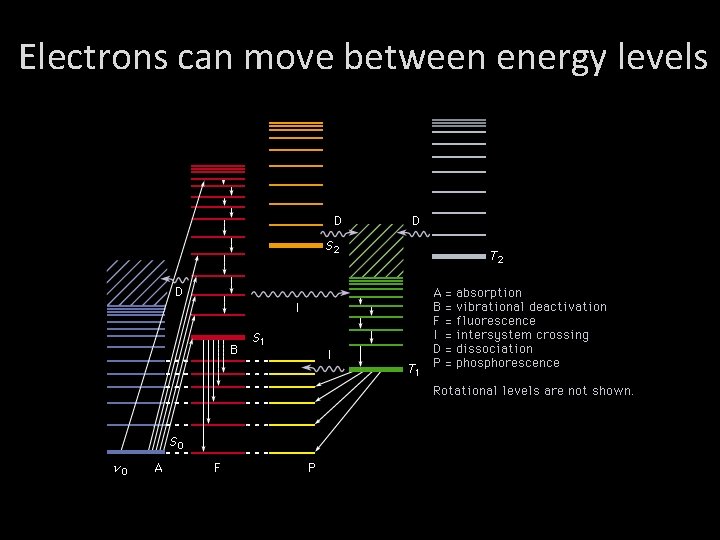
Electrons can move between energy levels

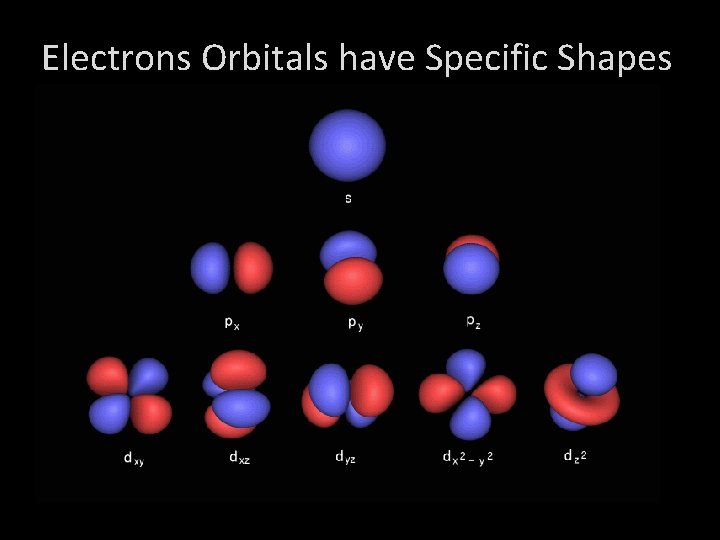
Electrons Orbitals have Specific Shapes

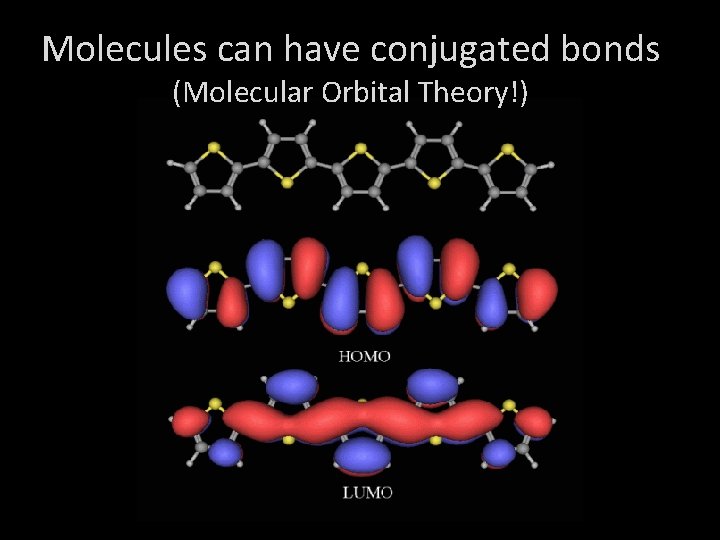
Molecules can have conjugated bonds (Molecular Orbital Theory!)

Molecules can be aromatic

So how does this awesome chemistry help us? Spectroscopy! Interaction of light and matter Specifically…. • UV-VIS • Fluorescence

1. Grab your sample 2. Filter your sample 3. Run it on a DOC analyzer for Dissolved Organic Carbon (DOC) and Dissolved Organic Nitrogen (DON) concentrations 4. Spectroscopy!

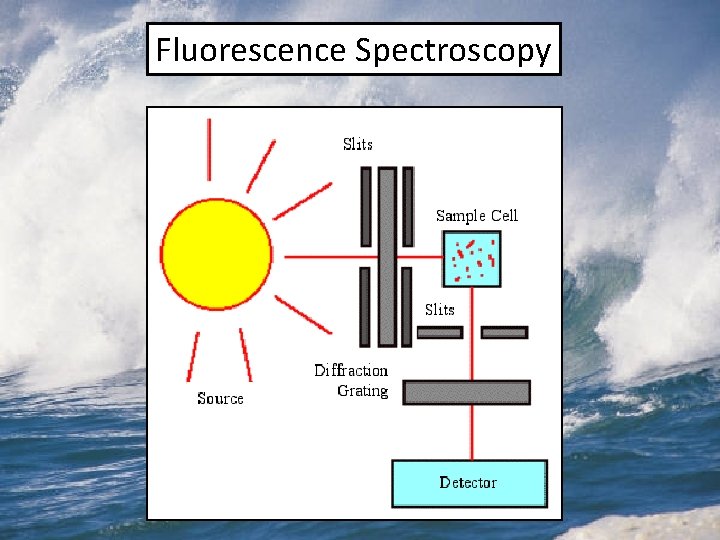
UV-VIS Spectroscopy and SUVA 254

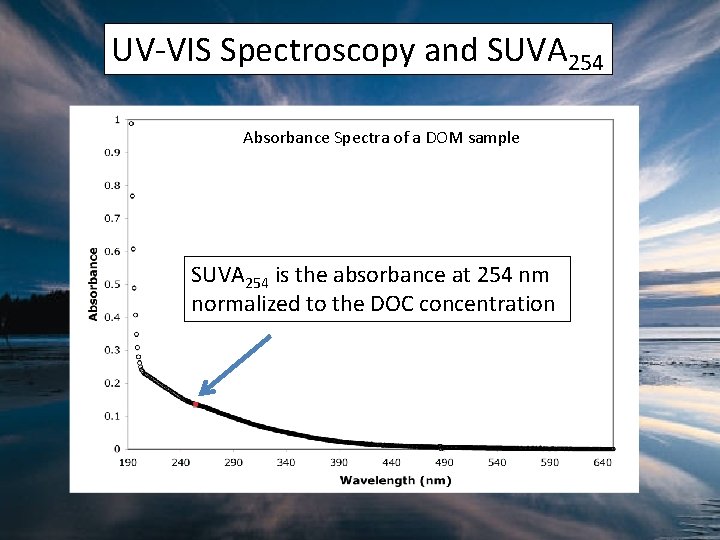
UV-VIS Spectroscopy and SUVA 254 Absorbance Spectra of a DOM sample SUVA 254 is the absorbance at 254 nm normalized to the DOC concentration

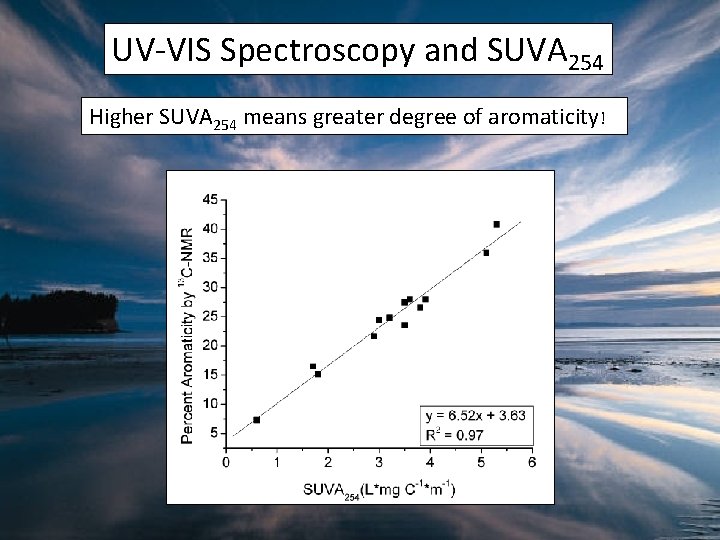
UV-VIS Spectroscopy and SUVA 254 Higher SUVA 254 means greater degree of aromaticity!

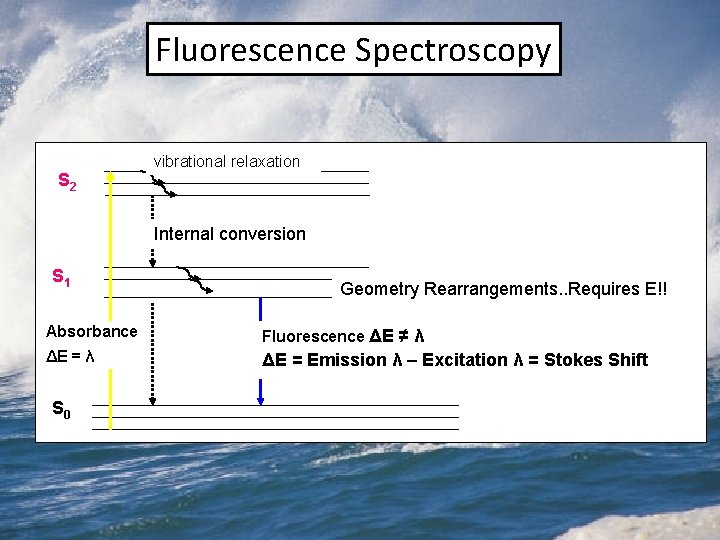
Fluorescence Spectroscopy S 2 vibrational relaxation Internal conversion S 1 Geometry Rearrangements. . Requires E!! Absorbance Fluorescence ΔE ≠ λ ΔE = Emission λ – Excitation λ = Stokes Shift S 0

Fluorescence Spectroscopy

“Q” “HQ” Brief Quinones found in. Interlude enzymes, e. g ubiquinone, and formed by lignin oxidation. For • More • Chemistry • Ubiquinone Forms of this complex are found throughout cells Important in electron transfer reactions, such as the oxidation of NADH Also known as coenzyme Q

lignin OH O Me O OH lignin O O Me O OMe O Me Quinones! O OMe HO O OH

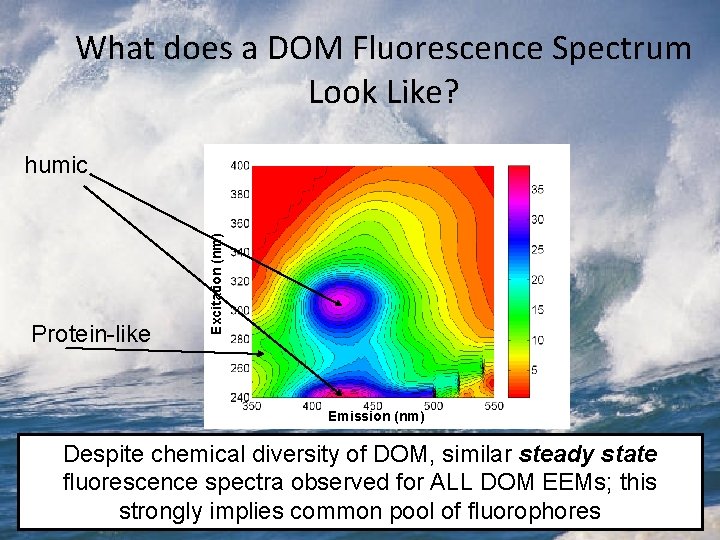
What does a DOM Fluorescence Spectrum Look Like? Protein-like Excitation (nm) humic Emission (nm) Despite chemical diversity of DOM, similar steady state fluorescence spectra observed for ALL DOM EEMs; this strongly implies common pool of fluorophores

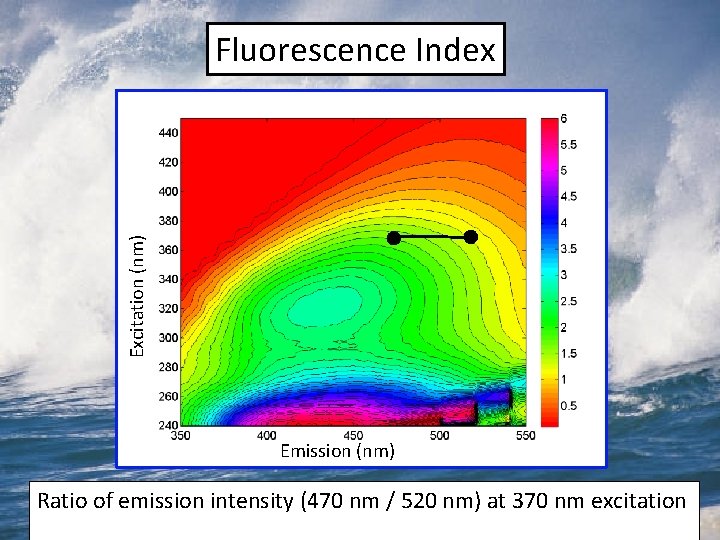
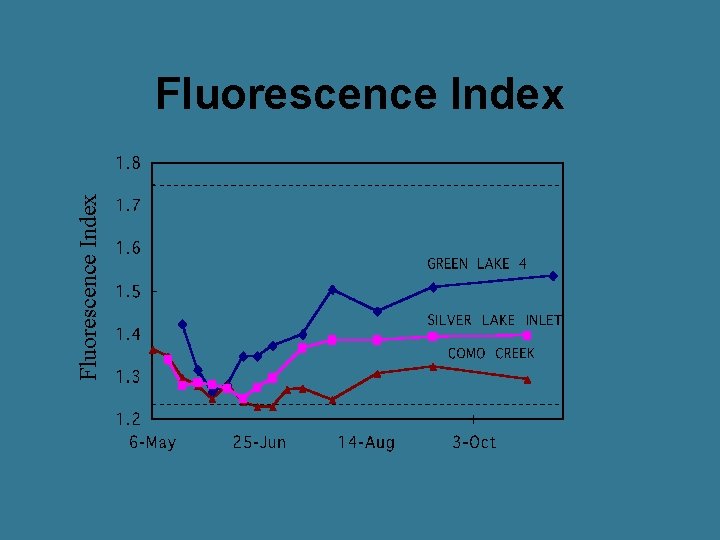
Excitation (nm) Fluorescence Index Emission (nm) Ratio of emission intensity (470 nm / 520 nm) at 370 nm excitation

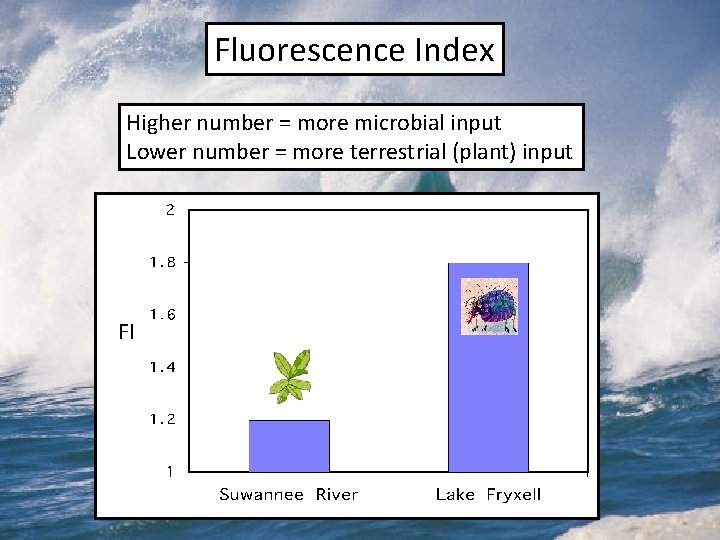
Fluorescence Index Higher number = more microbial input Lower number = more terrestrial (plant) input FI

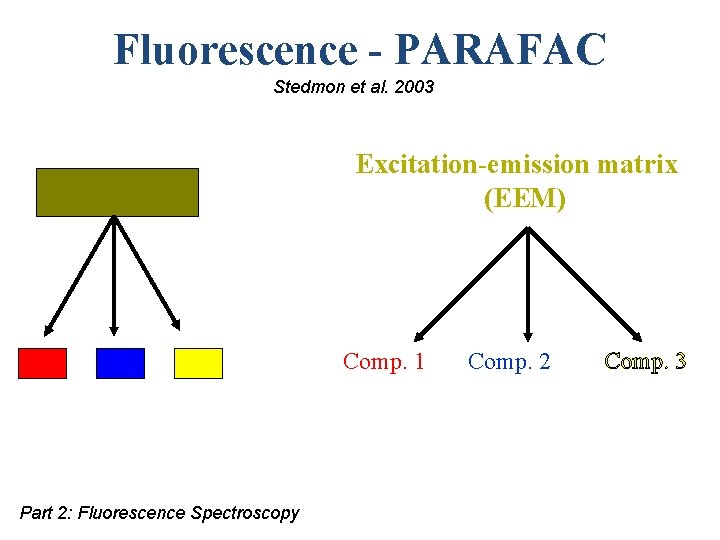
Fluorescence - PARAFAC Stedmon et al. 2003 Excitation-emission matrix (EEM) Comp. 1 Part 2: Fluorescence Spectroscopy Comp. 2 Comp. 3

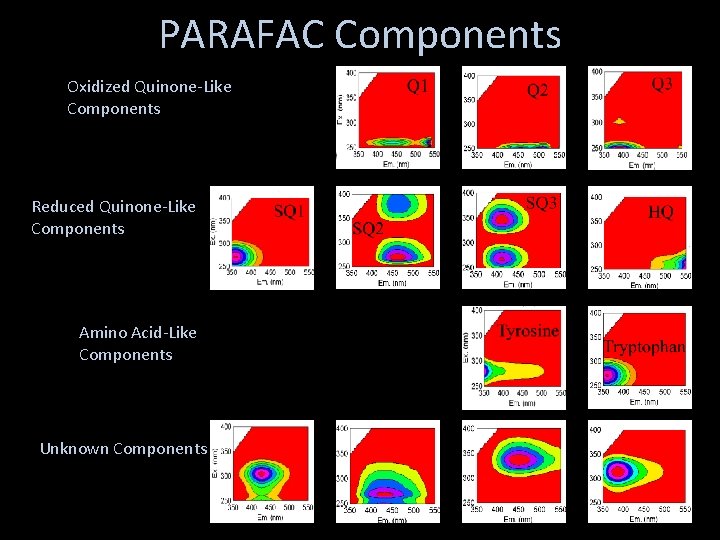
PARAFAC Components Oxidized Quinone-Like Components Reduced Quinone-Like Components Amino Acid-Like Components Unknown Components

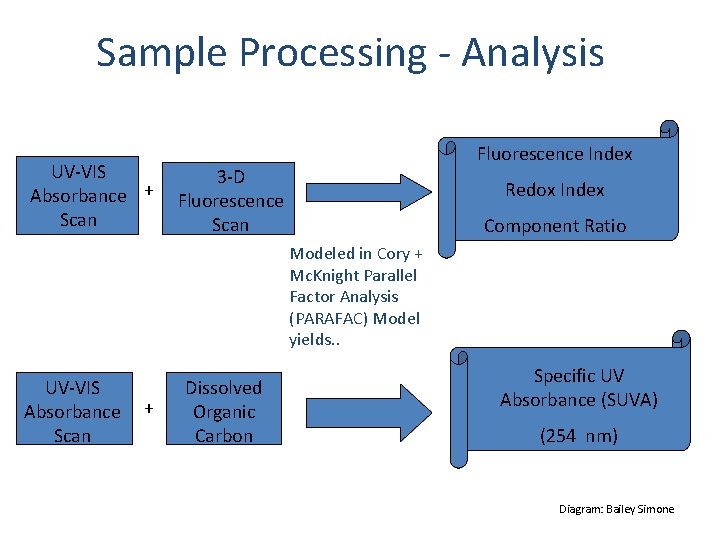
Sample Processing - Analysis UV-VIS Absorbance + Scan Fluorescence Index 3 -D Fluorescence Scan Redox Index Component Ratio Modeled in Cory + Mc. Knight Parallel Factor Analysis (PARAFAC) Model yields. . UV-VIS Absorbance Scan + Dissolved Organic Carbon Specific UV Absorbance (SUVA) (254 nm) Diagram: Bailey Simone

DOM Fractionation


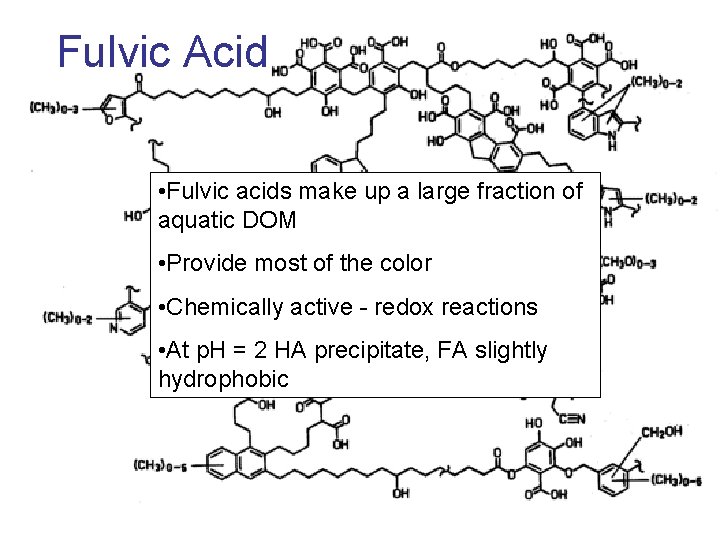
Fulvic Acid • Fulvic acids make up a large fraction of aquatic DOM • Provide most of the color • Chemically active - redox reactions • At p. H = 2 HA precipitate, FA slightly hydrophobic

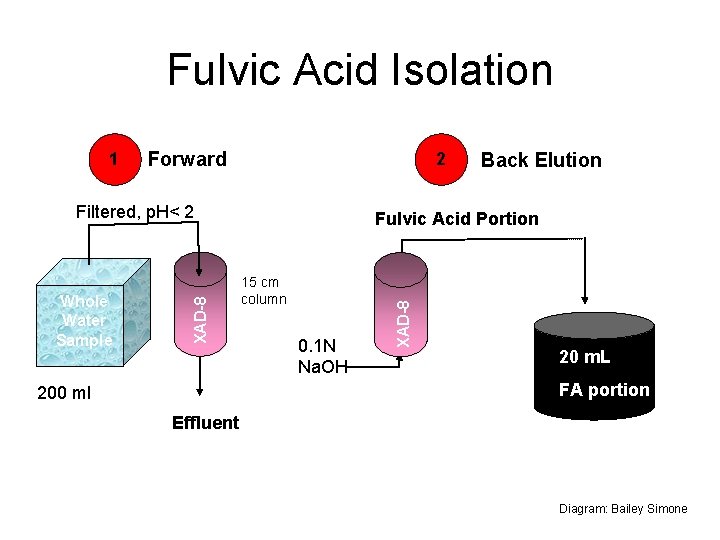
Fulvic Acid Isolation Forward 2 Whole Water Sample XAD-8 Filtered, p. H< 2 Back Elution Fulvic Acid Portion 15 cm column 0. 1 N Na. OH XAD-8 1 20 m. L FA portion 200 ml Effluent Diagram: Bailey Simone

XAD-8 Columns

So We Can Know… • DOC & DON concentrations • SUVA 254 • Degree of Aromaticity • Fluorescence Index • Microbial vs. Terrestrial Input • PARAFAC • Component Ratio • Degree of oxidation/reduction All for both whole water and fulvic acid fractions …. So What?

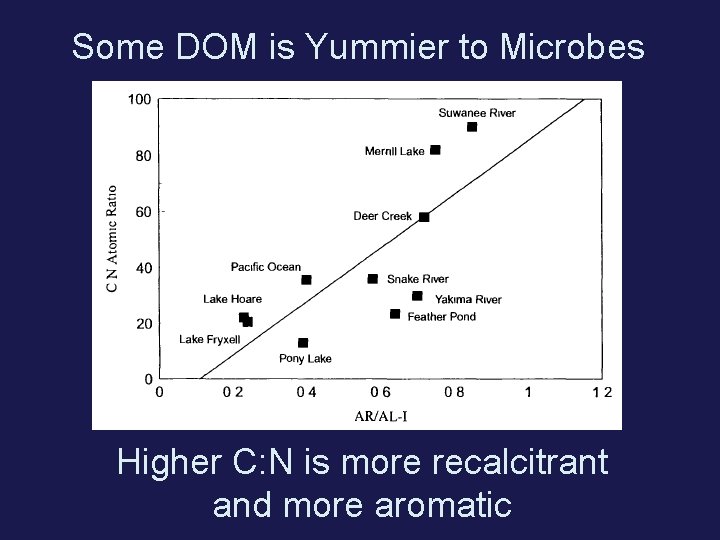
Some DOM is Yummier to Microbes Higher C: N is more recalcitrant and more aromatic

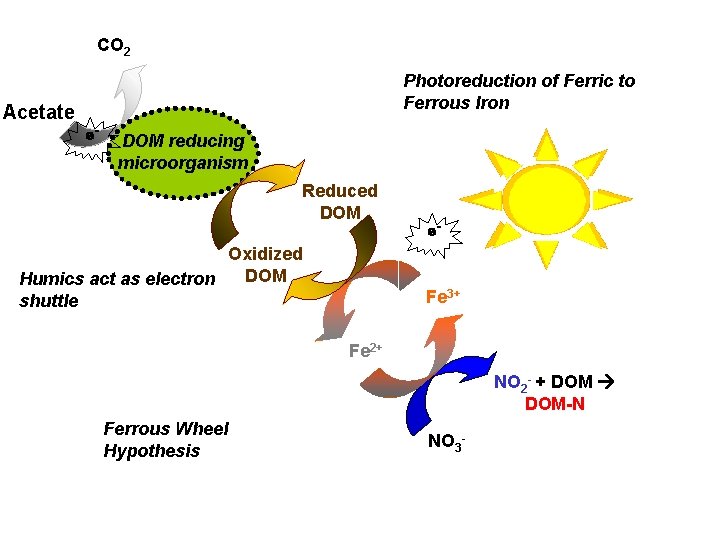
CO 2 Photoreduction of Ferric to Ferrous Iron Acetate e- DOM reducing microorganism Reduced DOM Oxidized DOM Humics act as electron shuttle e- Fe 3+ Fe 2+ NO 2 - + DOM-N Ferrous Wheel Hypothesis NO 3 -

CO 2 Electron shuttling affects: Acetate e- Photoreduction of Ferric to Ferrous Iron DOM reducing • microorganism Bioavailability of metals • Bioavailability of. Reduced certain nutrients • Degradation of persistent organic DOM epollutants Oxidized • Microbial respiration rates DOM Humics act as electron Fe 3+ shuttle And the ability depends on reduced/oxidized 2+ nature of DOM as well. Feas the source of the NO 2 - + DOM-N Ferrous Wheel Hypothesis NO 3 -

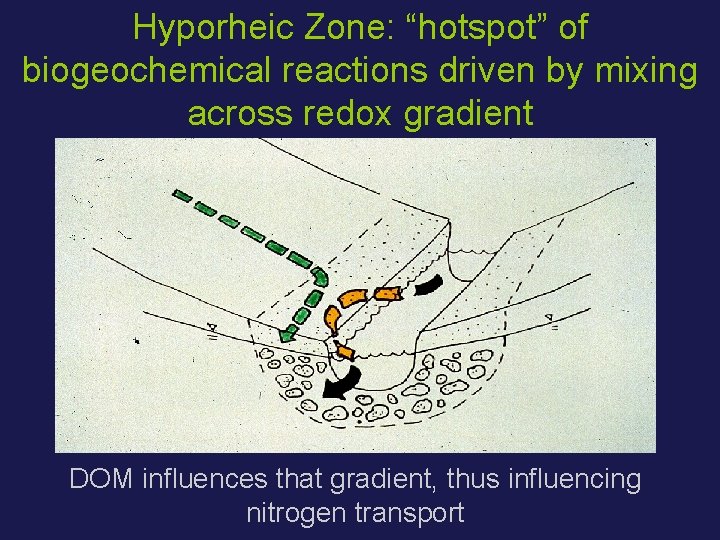
Hyporheic Zone: “hotspot” of biogeochemical reactions driven by mixing across redox gradient DOM influences that gradient, thus influencing nitrogen transport

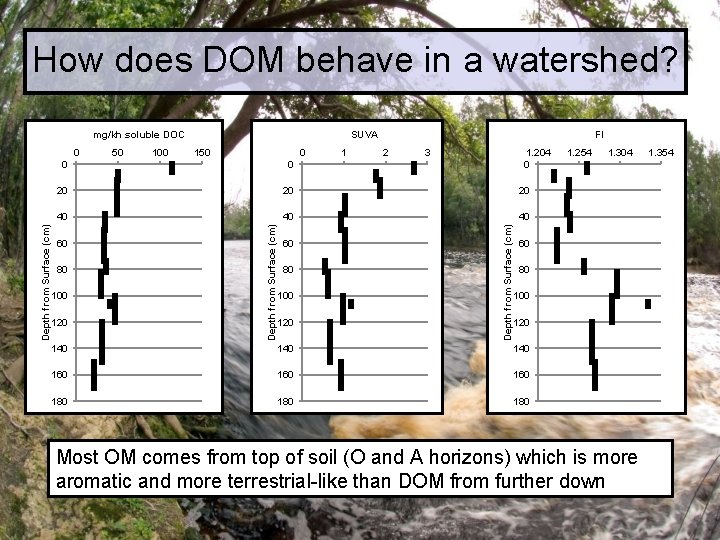
How does DOM behave in a watershed? mg/kh soluble DOC 50 100 150 0 1 FI 2 3 1. 204 0 0 20 20 20 40 40 40 60 80 100 120 Depth from Surface (cm) 0 SUVA 1. 254 1. 304 60 80 100 120 140 140 160 160 180 180 Most OM comes from top of soil (O and A horizons) which is more aromatic and more terrestrial-like than DOM from further down 1. 354

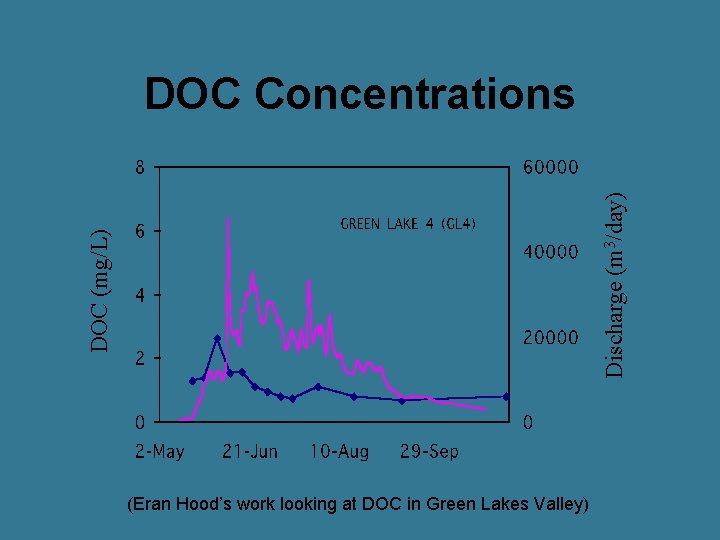
DOC (mg/L) Discharge (m 3/day) DOC Concentrations (Eran Hood’s work looking at DOC in Green Lakes Valley)

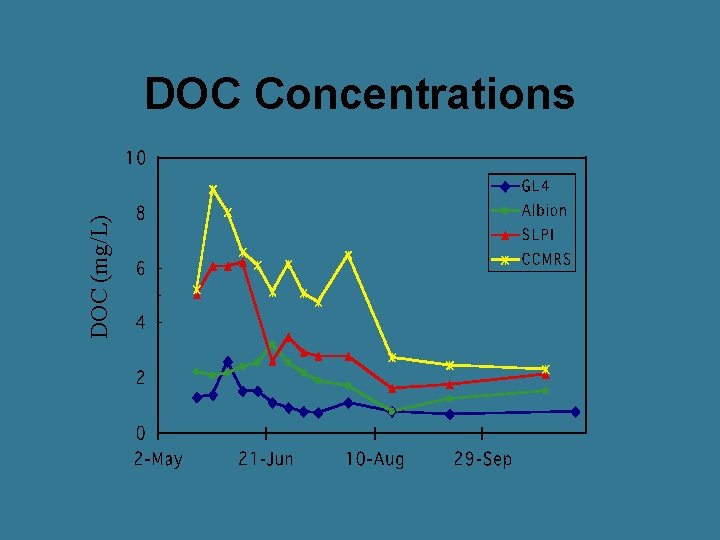
DOC (mg/L) DOC Concentrations

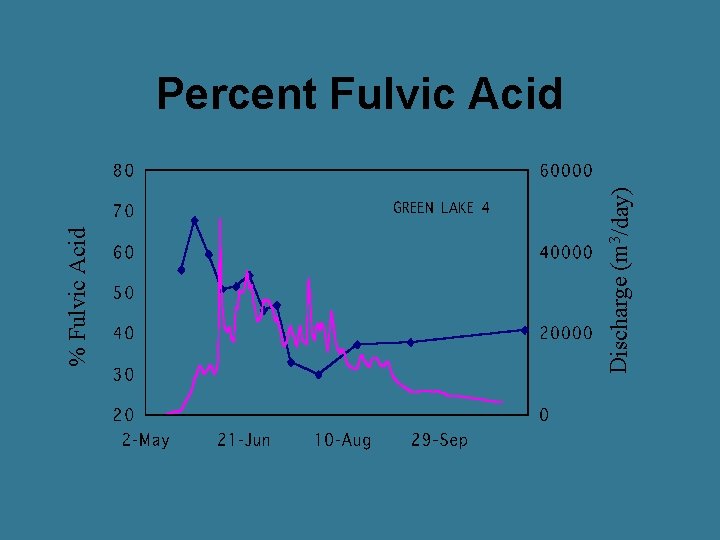
Discharge (m 3/day) % Fulvic Acid Percent Fulvic Acid

Fluorescence Index

General Trends in an Alpine System • DOC peaks just before peak discharge • Fulvic acids highest on rising limb • Terrestrial production main source of DOM during snowmelt • Terrestrial sources decrease on recession limb • On recession limb, alpine lakes have greater proportion of aquatic sources

lignin Dissolved Organic Matter lignin OH O Me O OH lignin OH O O Me O OMe HO O O Me lignin OH O OMe