Light The Human Perception of Color Color what



















































- Slides: 65

Light The Human Perception of Color

Color: what to learn in this lesson • Color isn’t “real”, figment of our imagination it is a • Color only “exists” in the human mind • Energy is the real characteristic of every light particle • Our eyes can detect only a small range of the possible energies • Goal today is to understand this. . .

Description of LIGHT • Called the Electromagnetic spectrum • Based on the energy of the photons

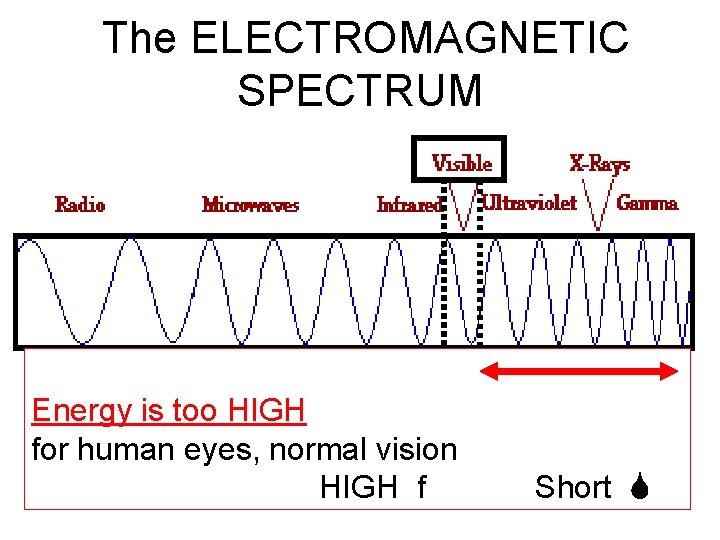
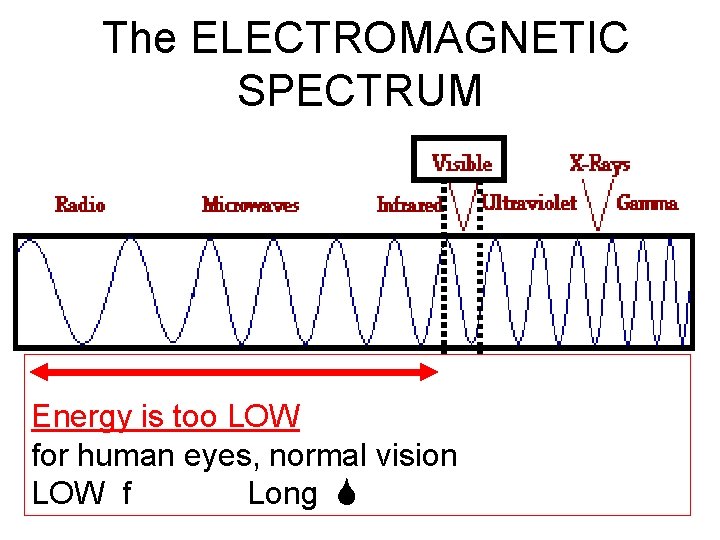
The EM spectrum • LIGHT comes in two “flavors”: 1. VISIBLE LIGHT: the very small range of energies of light which our eyes can detect. 2. There is MUCH, MUCH more light which is not “visible” to human eyes.

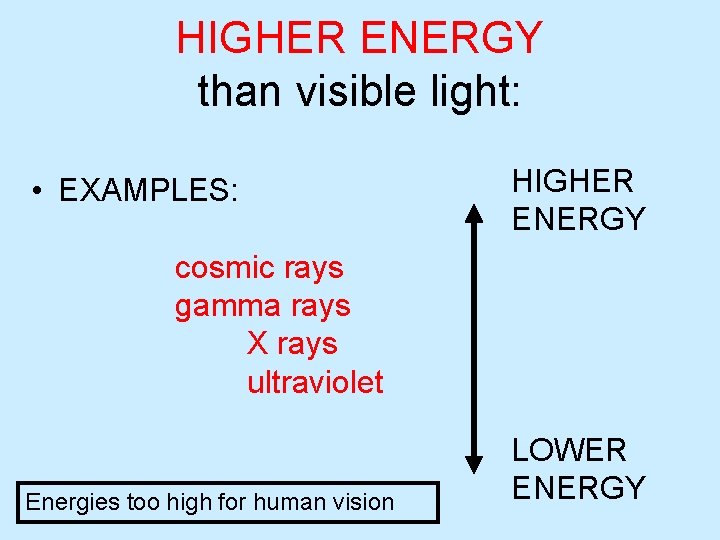
HIGHER ENERGY than visible light: • EXAMPLES: HIGHER ENERGY cosmic rays gamma rays X rays ultraviolet Energies too high for human vision LOWER ENERGY

The ELECTROMAGNETIC SPECTRUM • Energy is too HIGH for human eyes, normal vision HIGH f Short

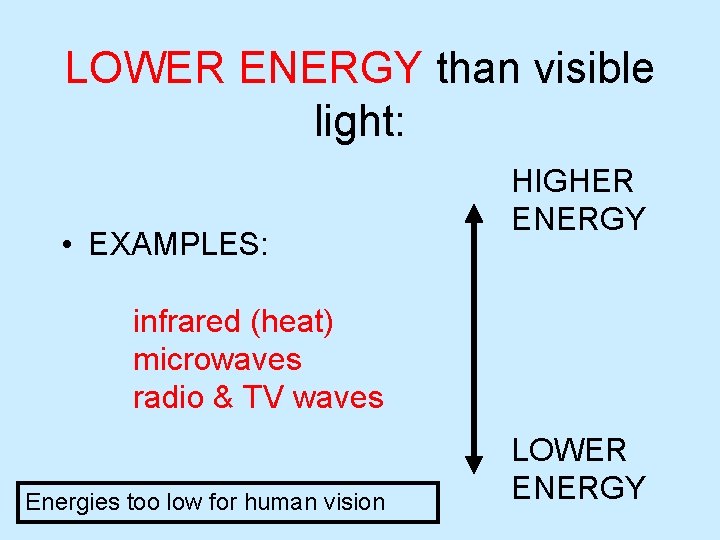
LOWER ENERGY than visible light: • EXAMPLES: HIGHER ENERGY infrared (heat) microwaves radio & TV waves Energies too low for human vision LOWER ENERGY

The ELECTROMAGNETIC SPECTRUM • Energy is too LOW for human eyes, normal vision LOW f Long

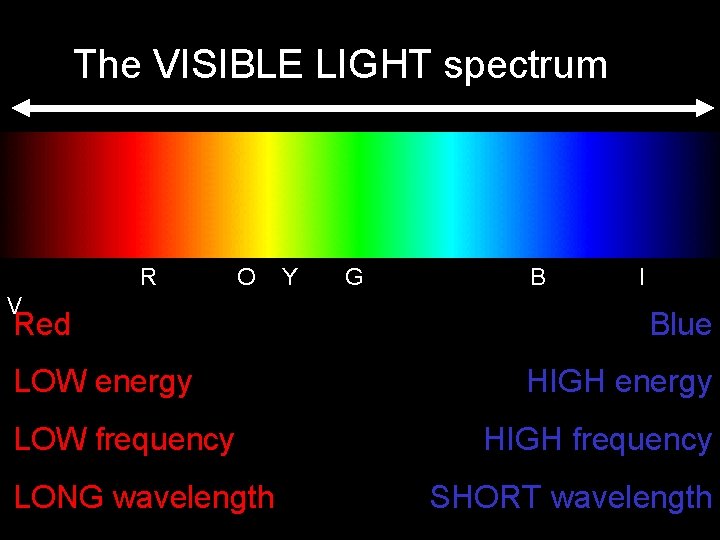
The VISIBLE LIGHT spectrum • the portion of the electromagnetic spectrum we can sense with our EYES. • Our skin can detect other portions as well. . . Leading to sunburn, cancer. . . • Both UV and IR are invisible to our eyes, but are sensed by the skin. • Some of the energies, “colors of light”, which are invisible to humans are visible to other species.

The VISIBLE LIGHT spectrum R O V Red LOW energy LOW frequency LONG wavelength Y G B I Blue HIGH energy HIGH frequency SHORT wavelength

SUMMARY: Radio & TV micro IR EM spectrum visible UV X rays long low WAVELENGTH FREQUENCY ENERGY short high low DANGER to LIFE high Know this table and “ROY G BIV” See page 521 in your text

Where does light come from? • Photons are emitted when electrons lose energy: • All atoms are surrounded by electrons • CHEMISTRY CONNECTION: these electrons “live” in certain energy levels • Electrons can change energy states in an atom: if they Absorb light: gain energy Loose energy: emit light

OUTCOME • Atoms of each element have a unique, characteristic electron configuration. • Each element has a unique, characteristic light spectrum. • A specific element can only emit and absorb particular energies of light. • We can only “see” some of those “colors.

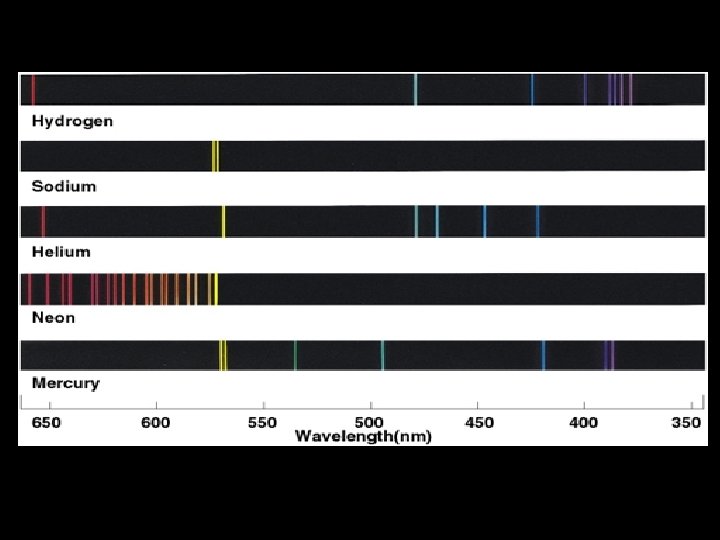
Atomic emission spectra SOURCES of light: In order, from left to right Atomic sources • Hydrogen • Helium • Neon • Argon • Krypton • Mercury Compound sources • H 2 O • CO 2

Our eyes “see” a collection of different energies. . • We normally “see” a lot of different colors SIMULTANEOUSLY. • To see what colors are really there, we have to use a diffraction grating. • Notice the differences when we look at each light, one at a time:

Hydrogen H 2 O Helium CO 2 Neon Argon Krypton Mercury


How do we see colors ? • We see when light enters the eye. • No light: DON’T SEE ANYTHING. • Our brain processes light information two ways: 1) in the “where” system 2) in the “what” system • The following examples of visual stimuli demonstrate some of the things we experience because of our color vision.

Example #1 The twinkling black spots do not actually exist in the intersections of the gray lines on the next slide. You can discover this by focusing on one particular intersection. The spots appear because of the way the cones and rods in your eyes respond to light.


This is easy to read, so you can decide to ignore it very rapidly

Very low contrast text is read by the part of the brain which locates objects, the “where” system. As long as the words are easy to recognize, you can read rather rapidly, but recondite or infrequently encountered words seem unfamiliar and have to be read letter-by-letter.

This is also hard to read. It jumps around and seems unstable because the “where” system has trouble seeing it. Advertisers use this trick to make you pay attention because you have to slow down and read each individual word.

This is also hard to read. It jumps around and seems unstable because the “where” system has trouble seeing it. Advertisers use this trick to make you pay attention because you have to slow down and read each individual word. Changing the color of the letters lot helps a

This is also hard to read. It jumps around and seems unstable because the “where” system has trouble seeing it. Advertisers use this trick to make you pay attention because you have to slow down and read each individual word. Changing the background color also helps a lot

This is hard to read even though each individual letter is easy to see, so you have to pay a lot of attention in order to read it.



Why do things like this happen ?

When light enters the eye. . • Light from some source strikes the eye • Passes through the CORNEA • Amount let in controlled by the IRIS which adjusts the opening of the PUPIL • The light is focused by the LENS, • passes through some “transparent stuff” and strikes the RETINA. • It is absorbed, creating an electrical signal which is sent by the optic nerve to the brain where it is perceived as an image.

COLOR VISION ONLY EXISTS IN THE BRAIN • Biology connection: • Color is not something “real” or “special” about the visible spectrum. • Most species do not see color. • We see color because of the way our eyes are built. . . and the way our brain interprets what we see.

How do we see the different colors? • Occurs because of the construction of the retina. • The retina is composed of millions of photoreceptor cells called “rods” and “cones”.

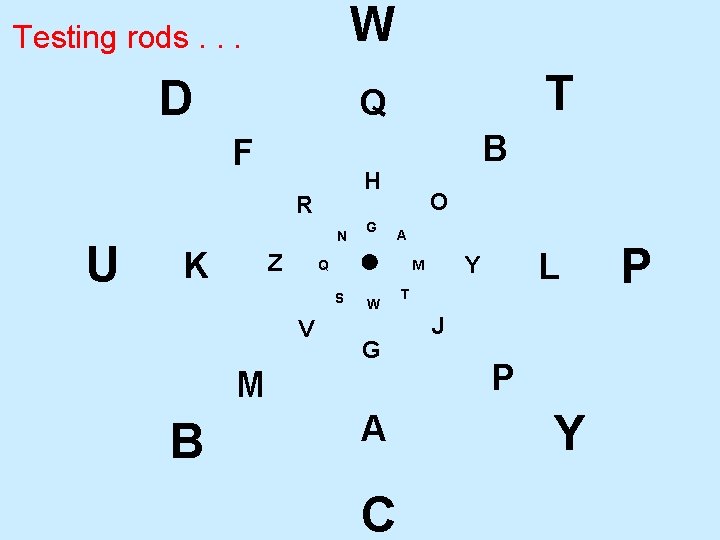
rods • Can not distinguish energies (see color) • Can only sense brightness. • Extremely sensitive to dim light. • Are use to identify object location using the “where” portion of the brain perceptual processing centers. • The way rods work are behind the checker board pattern illusion.

rods • Do not detect “edges” well: so are not very useful in reading. ( Large pixels) • Used to see at night. Helpful in looking at stars. • Quite sensitive to peripheral motion.

W Testing rods. . . D F N K Z G O A Q V W G M A C L Y M S B B H R U T Q T J P Y P

cones • Function only under bright light conditions (takes a lot of photons). • Cones can distinguish different photon energies. • Very sensitive to edges; good focus. VERY Small pixels • About 3 million packed in fovea, center part of the retina. • Fovea is where image is formed when we look at something.

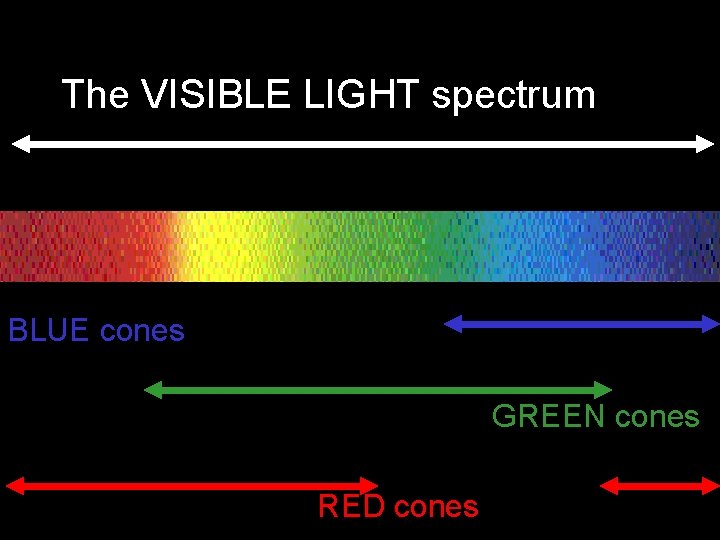
How does color vision work? • Three kinds of cones: million in the fovea. about 3 • “Blue” cones = about 1 % • The rest are “Green” and “Red” • Each type of cone is sensitive to a energies (what we call “colors”) range of

The VISIBLE LIGHT spectrum BLUE cones GREEN cones RED cones

Basic understanding All of the “colors” of the rainbow exist in an energy sense. actually But because of the way our eyes work : 1. we can see yellow when we see yellow or 2. we can “see yellow” when only the colors red and green are both present at the same time. .


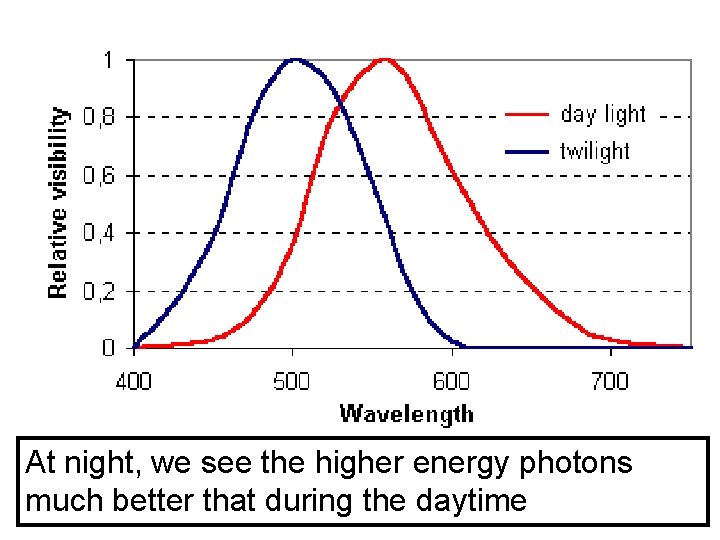
At night, we see the higher energy photons much better that during the daytime

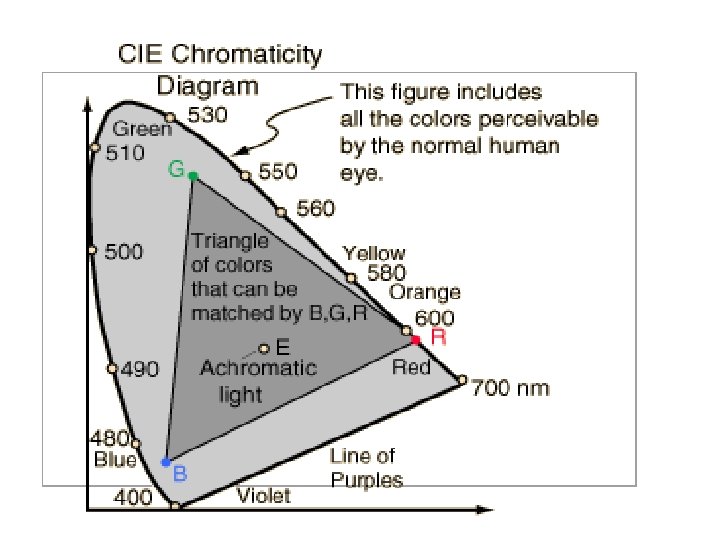
What are the PRIMARY COLORS ? • Based on the way the human eye works we synthesize “colors” in our brain. • The human mind creates our color palate. • In practice, it only takes three colors: BLUE Primary colors GREEN of light RED to “see” all of the colors known to humans.


Examples of 3 color process: • Called “color by addition”: adding colors of light. • Television sets and computer monitors: all are “RGB” • Works for all SOURCES OF LIGHT. • Remember that each type of cone “SEES” more than a single shade of color.

Primary colors of light BLUE RED GREEN

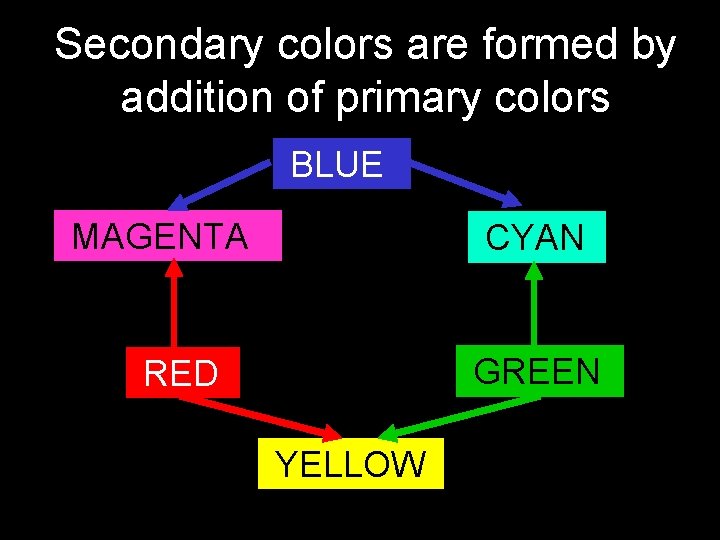
Secondary colors are formed by addition of primary colors BLUE MAGENTA CYAN GREEN RED YELLOW

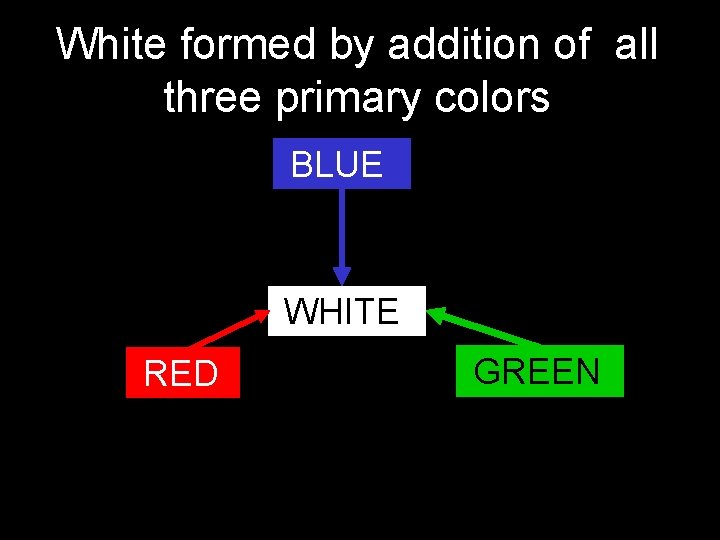
White formed by addition of all three primary colors BLUE WHITE RED GREEN

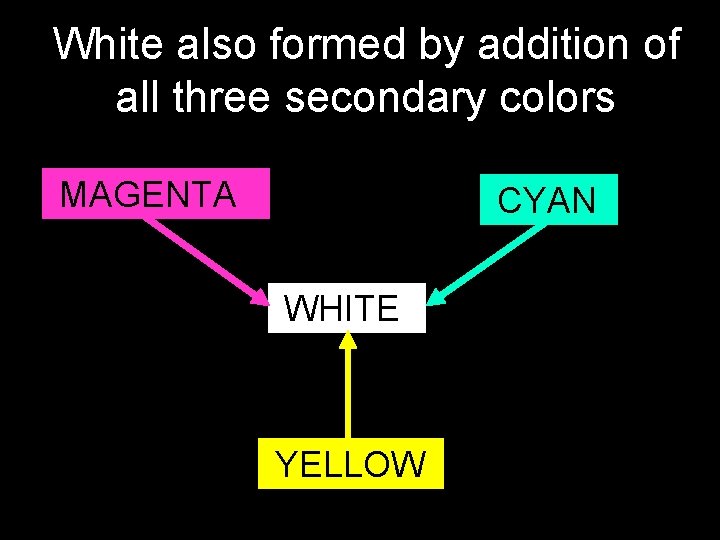
White also formed by addition of all three secondary colors MAGENTA CYAN WHITE YELLOW

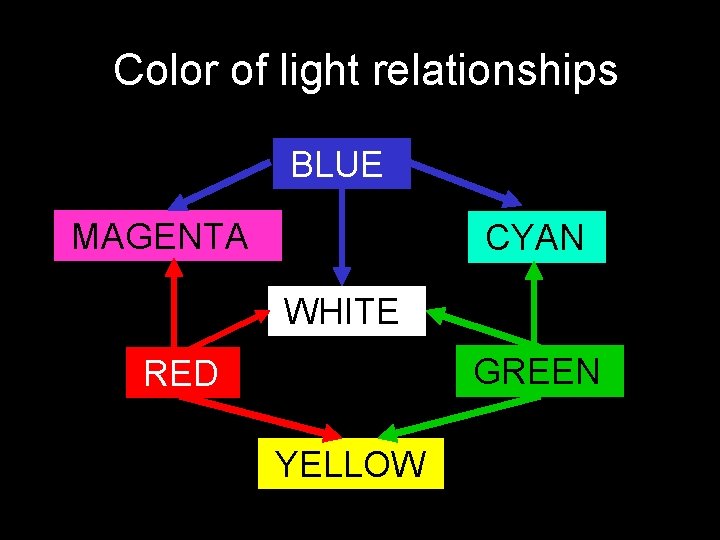
Color of light relationships BLUE MAGENTA CYAN WHITE GREEN RED YELLOW


PRIMARY and SECONDARY colors for pigments Works for all materials which color by absorption Seeing REFLECTED light. Seeing TRANSMITTED light. control

Pigments (paint, ink, filters) • DEFINE: Primary colors of pigment • Absorb only one primary color of light. • YELLOW absorbs only • CYAN absorbs only • MAGENTA absorbs only BLUE RED GREEN

Pigments (paint, ink, filters) • DEFINE: Secondary colors of pigment • Absorb two primary colors of light. • BLUE absorbs both RED and GREEN • RED absorbs both BLUE and GREEN • GREEN absorbs both RED and BLUE

What is IMPORTANT: • ASK: What colors are from the light source ? eliminated • When any light hits a page, some of the light is absorbed. • The rest is reflected into your eye. • The color you see based on the reflected colors that enter your eye. is

Comparison of PRIMARY colors • EMISSION BLUE RED GREEN • REFLECTION or TRANSMISSION YELLOW MAGENTA CYAN

Comparison of SECONDARY colors • EMISSION YELLOW MAGENTA CYAN • REFLECTION OR TRANSMISSION BLUE RED GREEN

Filters and slides • Theater filters are used to eliminate colors from white. • Slide projectors and movie film is designed to eliminate colors from white. • Some glasses are designed to eliminate colors from white. • Colored glass also eliminates colors from white.


EXAMPLES of color by subtraction • COLOR SLIDES • Photographic printing, magazines, computer printers, newspapers.

Three basic ways to see color: • Color by emission : the object emits colors of light. • Color by transmission: a filter absorbs some colors while letting others pass through • Color by reflection: pigment absorbs some colors while letting others reflect off.

IMPORTANT CONCLUSION In order to observe all “human” colors, must have a light source”. yo “whit

What you THINK you see vs. what is REALLY THERE

EXAMPLE: You can “see” yellow two different ways: 1. When only YELLOW LIGHT is present: it stimulates both red and green cones. • ONLY YELLOW LIGHT is really present. 2. When the RIGHT AMOUNT of both RED and GREEN are present • It APPEARS to be YELLOW but only RED and GREEN are actually being seen.

Key question: • What color(s) of light are actually entering your eye? • To see a particular color, the right colors of light have to (1) be present and (2) entering the eye to stimulate the cones by the right amount. • You CAN NOT see a color that isn’t present in the light SOURCE.

One More Reason Why Physics is Better Than Drugs Sunset on Maui