Light Series Benchmark 5 LIGHT Speed of Light

Light & Series Benchmark 5
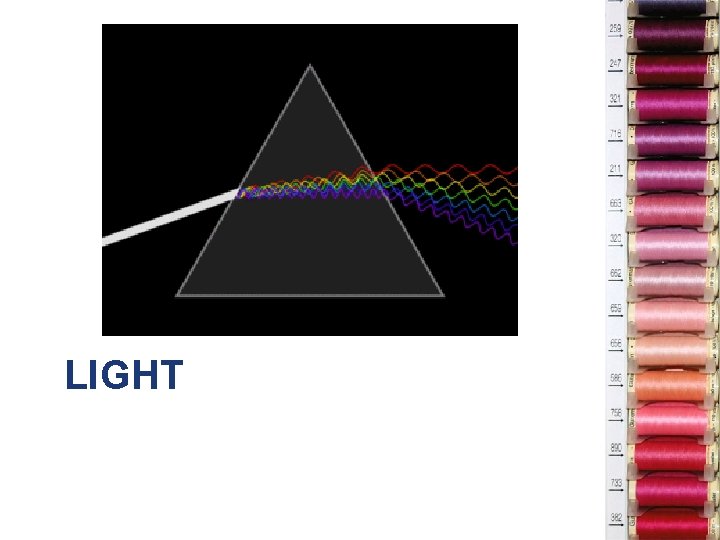
LIGHT

Speed of Light • All light travels the same speed in air according to the equation: λ • v=c λ = wavelength, usually in meters V = frequency, cycles/sec, s-1, Hertz c = speed of light 3. 00 x 108 m/s

Albert Einstein Light is quantized! • 1. Light is emitted in packages called photons. • 2. Different colors of light have different amounts of energy in their photons • 3. The energy is proportional to the frequency of light wave. • Higher v = higher E • E = hv • h = Planck’s constant • ROY G BIV
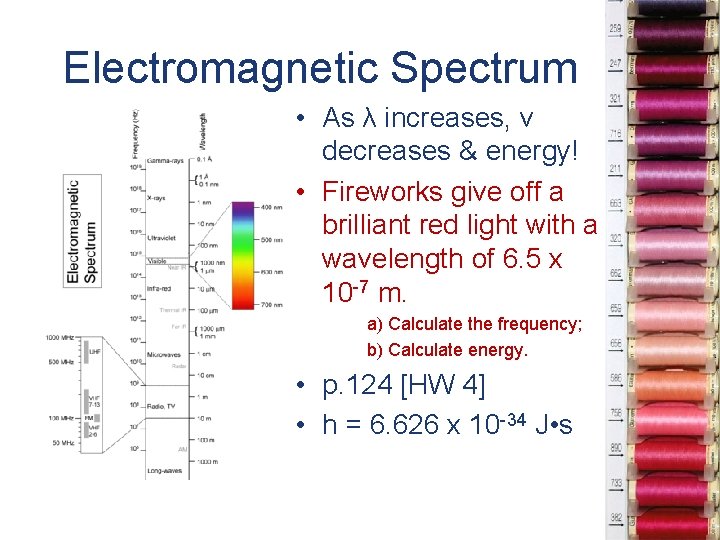
Electromagnetic Spectrum • As λ increases, v decreases & energy! • Fireworks give off a brilliant red light with a wavelength of 6. 5 x 10 -7 m. a) Calculate the frequency; b) Calculate energy. • p. 124 [HW 4] • h = 6. 626 x 10 -34 J • s

What does light have to do with the atom? SERIES & SPECTRUM

Rutherford’s Model • Solar System Model • Planetary Model BUT Classic physics – a charged particle (electron) traveling a curved path loses (radiates) energy

Bohr’s Model (1913) • Like Rutherford’s Model • Except: – Orbitals are different energy levels – Each orbital has a maximum amount of electrons that can occupy it
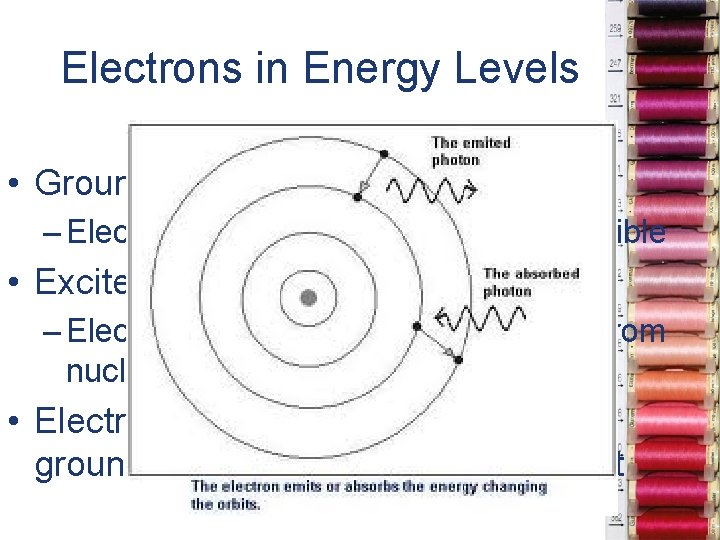
Electrons in Energy Levels • Ground state – Electrons as close to nucleus as possible • Excited state – Electron absorbs E and jumps away from nucleus • Electron traveling from excited to ground state releases energy as light
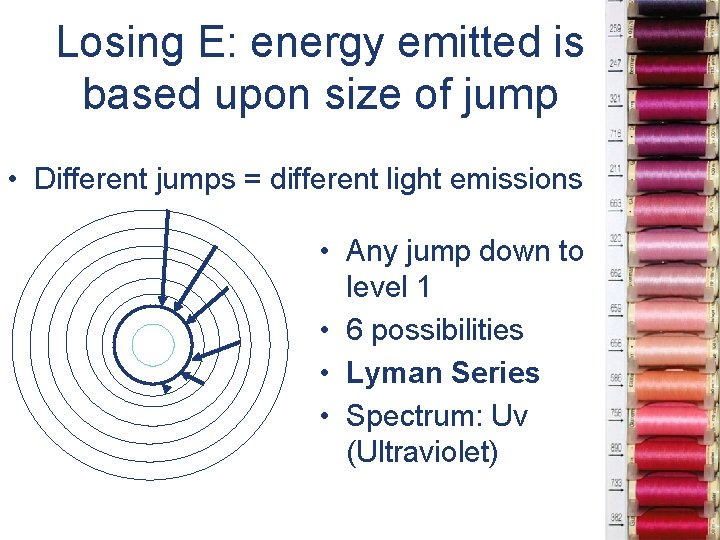
Losing E: energy emitted is based upon size of jump • Different jumps = different light emissions • Any jump down to level 1 • 6 possibilities • Lyman Series • Spectrum: Uv (Ultraviolet)

• Any jump down to level 2 • 5 possibilities • Balmer Series • Spectrum: Visible Light

• Any jump down to level 3 • 4 possibilities • Paschen Series • Spectrum: IR (infrared)
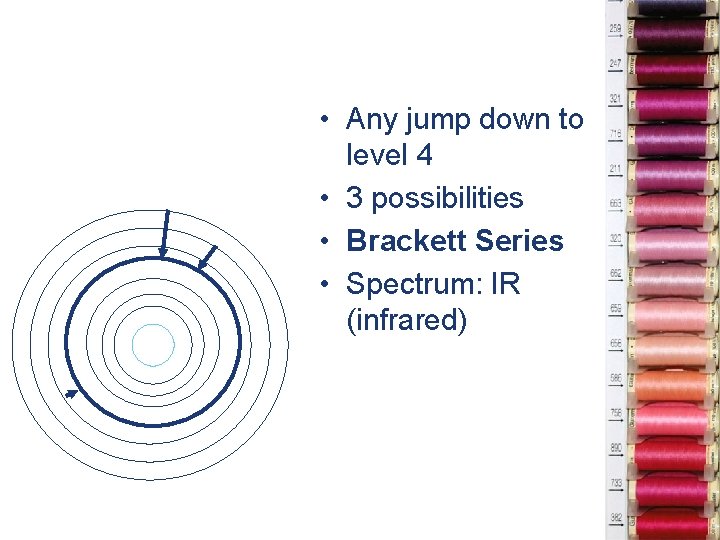
• Any jump down to level 4 • 3 possibilities • Brackett Series • Spectrum: IR (infrared)

• Any jump down to level 5 • 2 possibilities • Pfund Series • Spectrum: IR (infrared)

• Any jump down to level 6 • 1 possibility • IR 4 Series • Spectrum: IR (infrared)
- Slides: 16