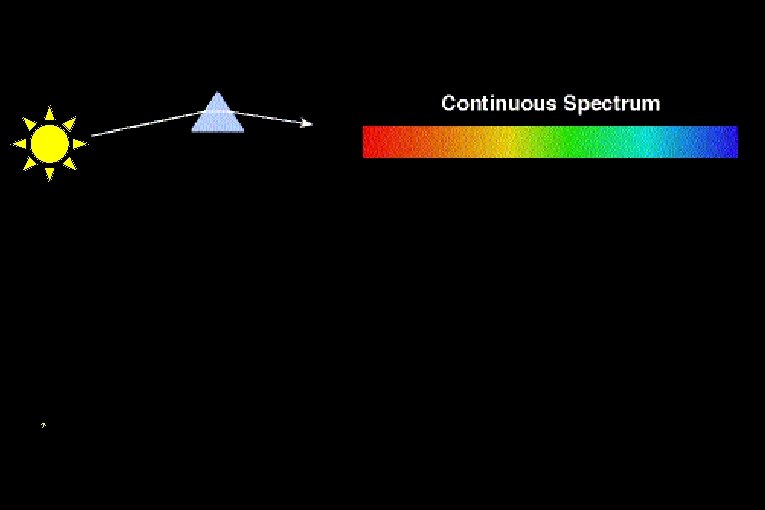
Light as Information Bearer Spectrum light separated into

Light as Information Bearer Spectrum: light separated into its different wavelengths. Spectroscopy: The quantitative analysis of spectra

Four Ways in Which Light can Interact with Matter The type of interaction between light and matter is determined by characteristics of the “matter” and by the wavelength of light.
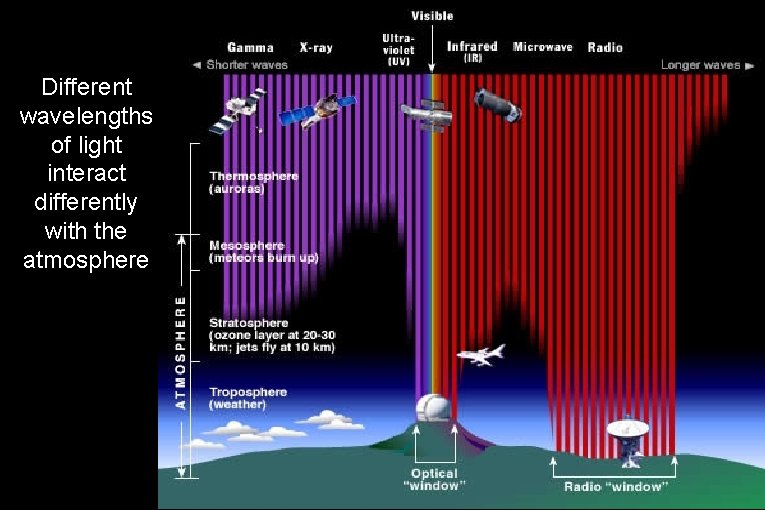
Different wavelengths of light interact differently with the atmosphere

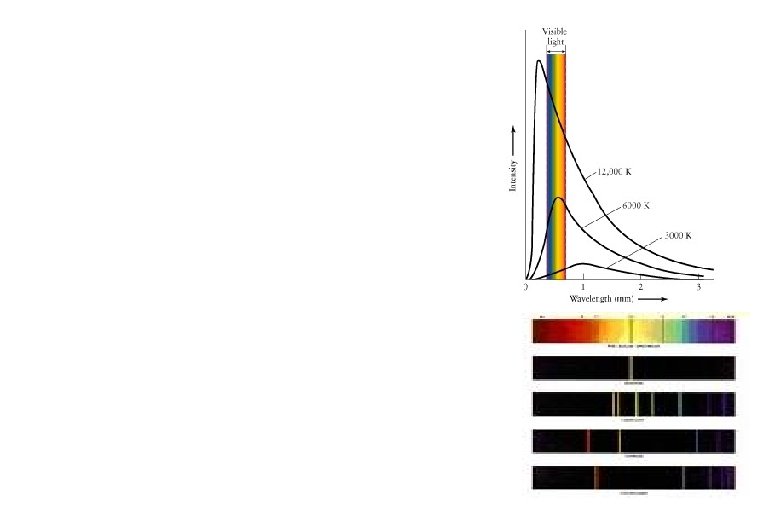

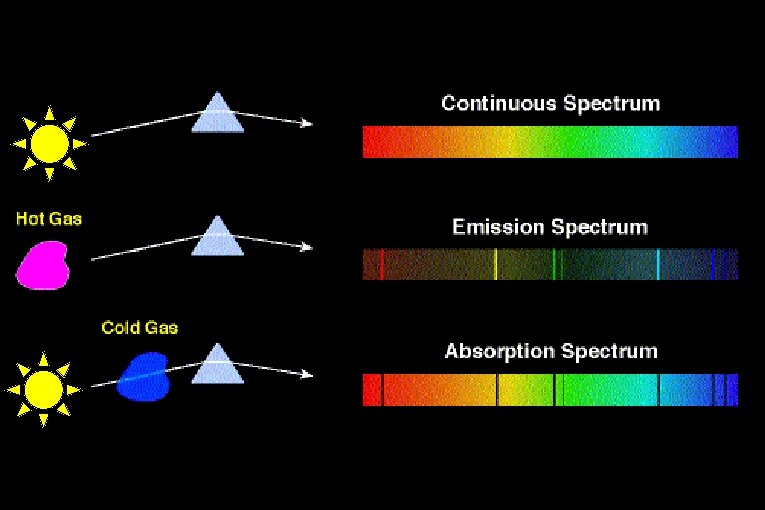
Thermal Emission A hot, dense glowing object (solid or gas) emits a continuous spectrum.
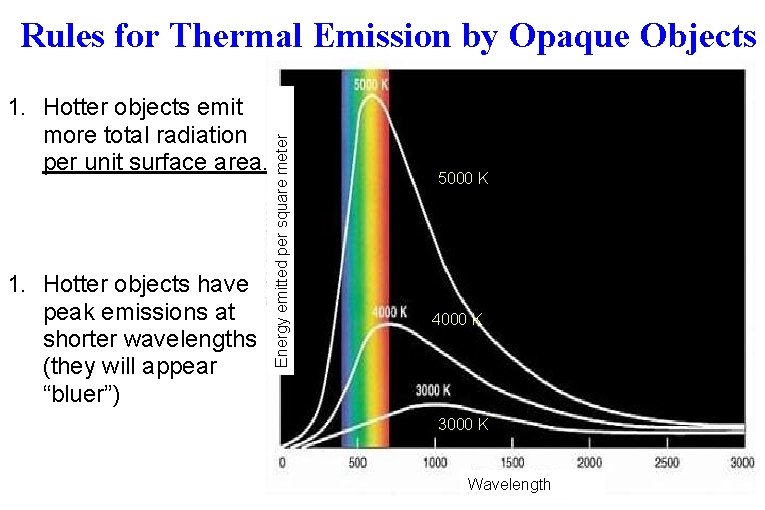
1. Hotter objects emit more total radiation per unit surface area. 1. Hotter objects have peak emissions at shorter wavelengths (they will appear “bluer”) Energy emitted per square meter Rules for Thermal Emission by Opaque Objects 5000 K 4000 K 3000 K Wavelength
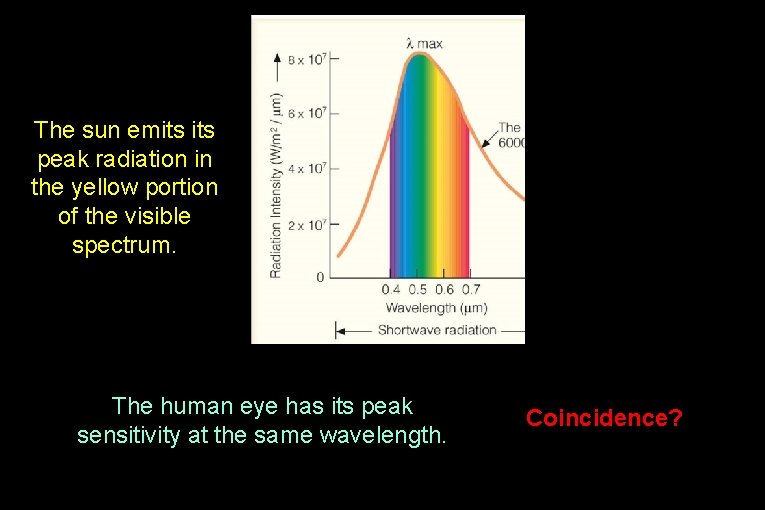
The sun emits peak radiation in the yellow portion of the visible spectrum. The human eye has its peak sensitivity at the same wavelength. Coincidence?

infrared At “room temperature”, or “body-temperature”, objects emit their peak radiation in the infrared. The surface of the Earth emits radiation in the infrared. visible Extremely hot objects will emit most of their radiation in the ultraviolet, x-ray or even the gamma ray portion of the spectrum
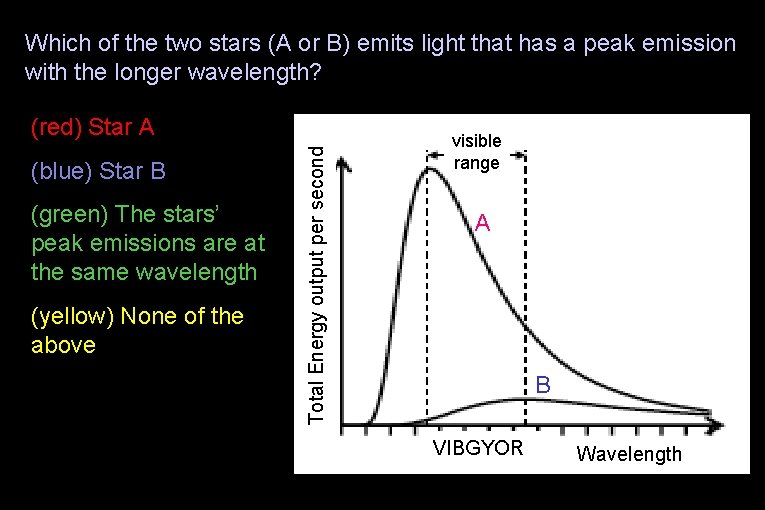
Which of the two stars (A or B) emits light that has a peak emission with the longer wavelength? (blue) Star B (green) The stars’ peak emissions are at the same wavelength (yellow) None of the above Total Energy output per second (red) Star A visible range A B VIBGYOR Wavelength
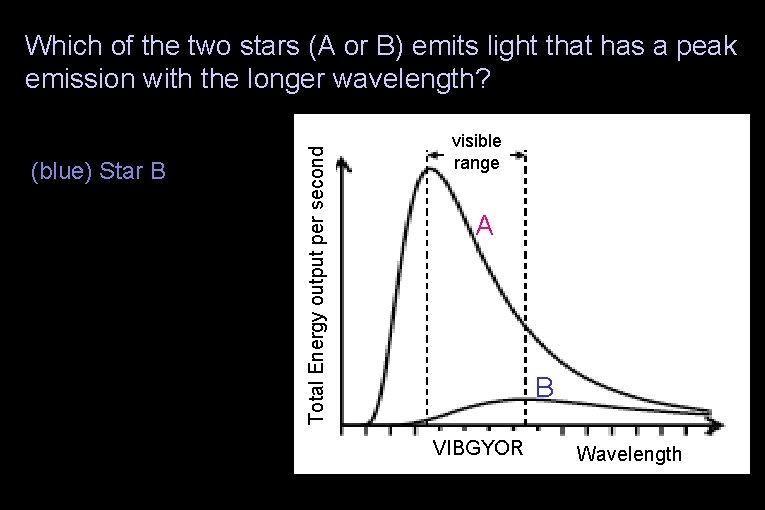
Which of the two stars (A or B) emits light that has a peak emission with the longer wavelength? (blue) Star B (green) The stars’ peak emissions are at the same wavelength (yellow) None of the above Total Energy output per second (red) Star A visible range A B VIBGYOR Wavelength
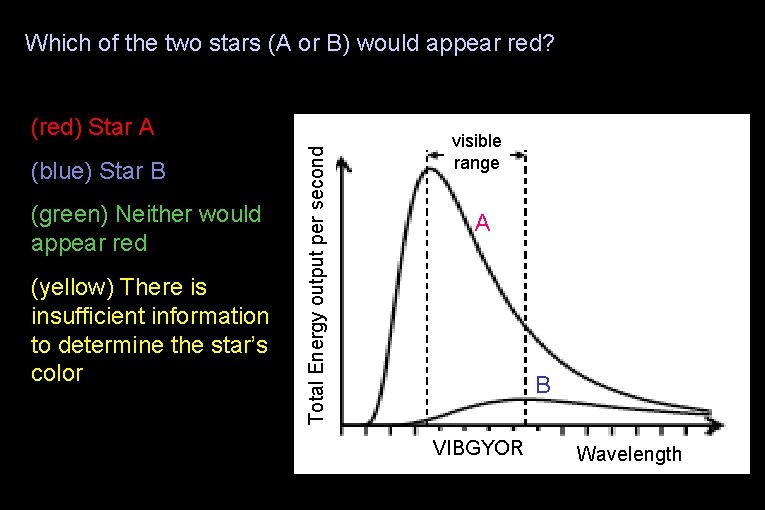
Which of the two stars (A or B) would appear red? (blue) Star B (green) Neither would appear red (yellow) There is insufficient information to determine the star’s color Total Energy output per second (red) Star A visible range A B VIBGYOR Wavelength
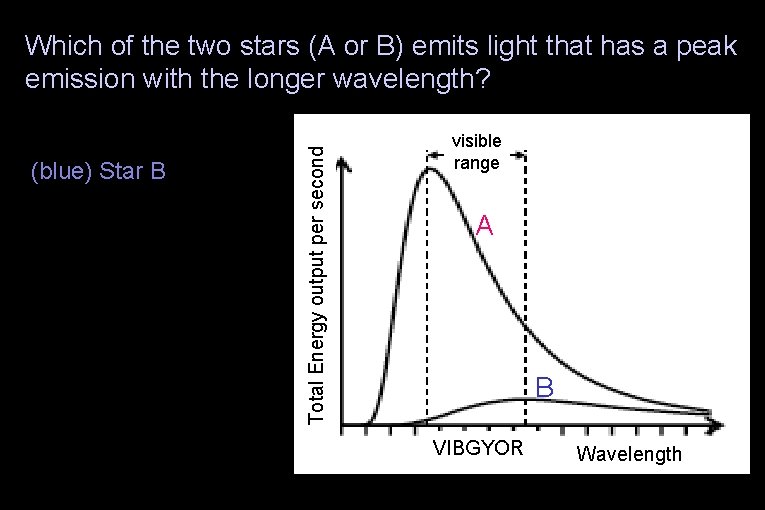
Which of the two stars (A or B) emits light that has a peak emission with the longer wavelength? (blue) Star B (green) The stars’ peak emissions are at the same wavelength (yellow) None of the above Total Energy output per second (red) Star A visible range A B VIBGYOR Wavelength
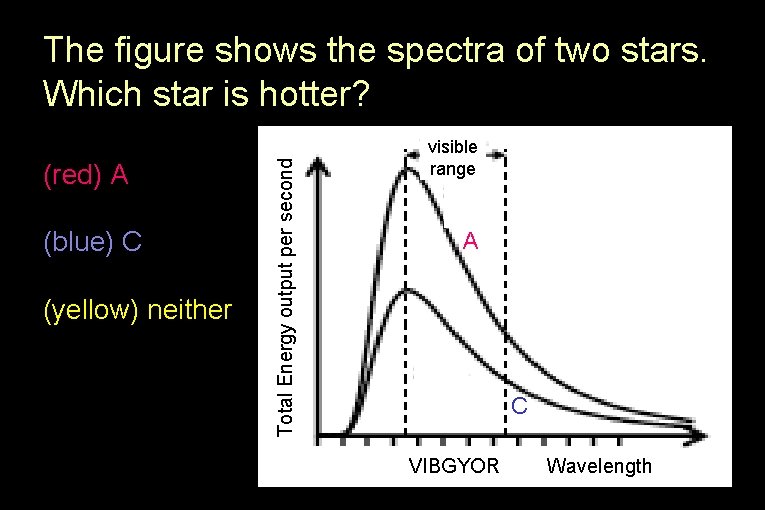
(red) A (blue) C (yellow) neither Total Energy output per second The figure shows the spectra of two stars. Which star is hotter? visible range A C VIBGYOR Wavelength
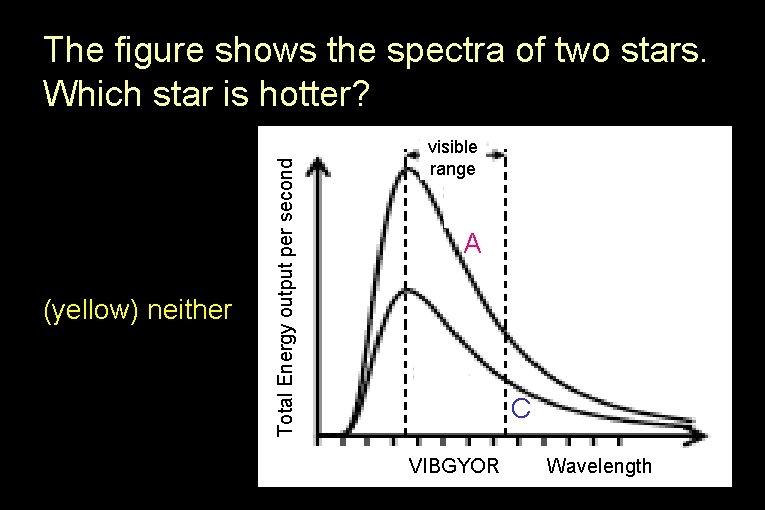
(red) A (blue) C (yellow) neither Total Energy output per second The figure shows the spectra of two stars. Which star is hotter? visible range A C VIBGYOR Wavelength
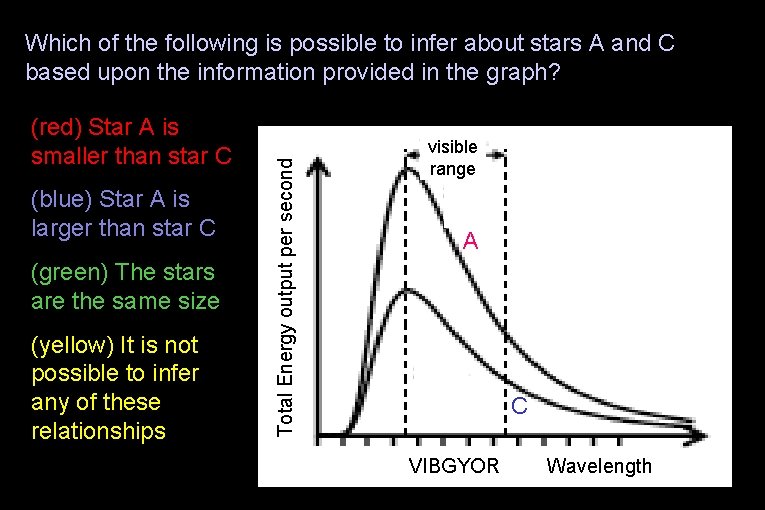
(red) Star A is smaller than star C (blue) Star A is larger than star C (green) The stars are the same size (yellow) It is not possible to infer any of these relationships Total Energy output per second Which of the following is possible to infer about stars A and C based upon the information provided in the graph? visible range A C VIBGYOR Wavelength
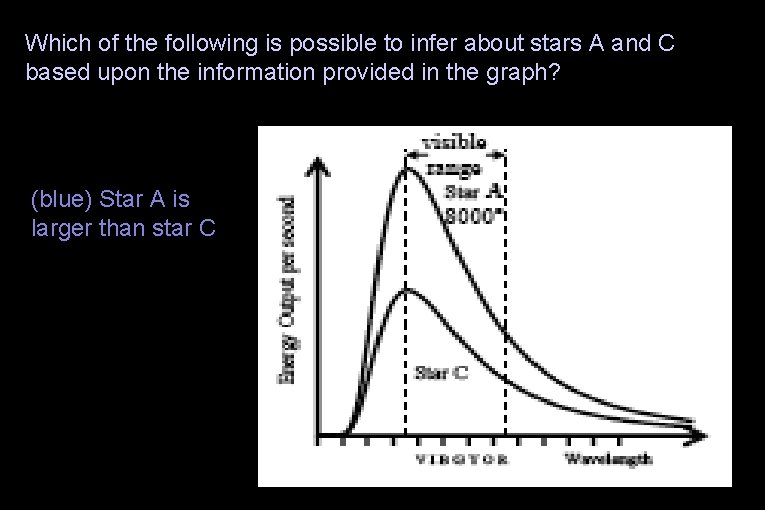
Which of the following is possible to infer about stars A and C based upon the information provided in the graph? (red) Star A is smaller than star C (blue) Star A is larger than star C (green) The stars are the same size (yellow) It is not possible to infer any of these relationships

A Brief Review of “Matter”
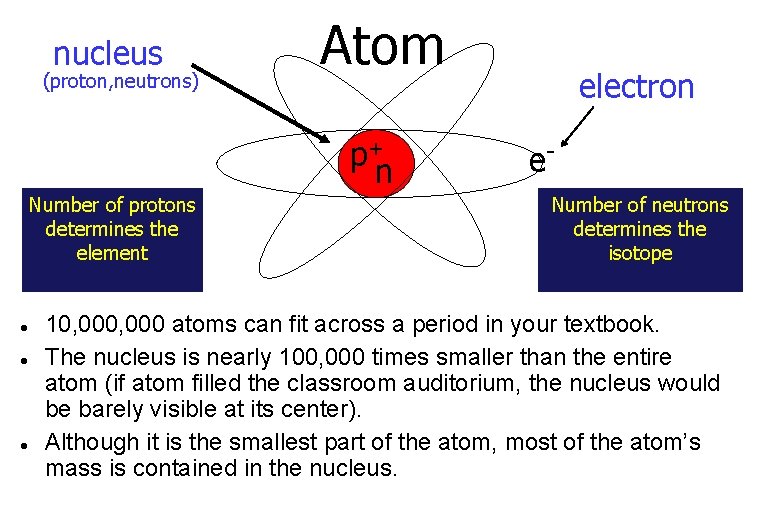
nucleus (proton, neutrons) Atom p+ n Number of protons determines the element electron e. Number of neutrons determines the isotope 10, 000 atoms can fit across a period in your textbook. The nucleus is nearly 100, 000 times smaller than the entire atom (if atom filled the classroom auditorium, the nucleus would be barely visible at its center). Although it is the smallest part of the atom, most of the atom’s mass is contained in the nucleus.

Electrons do not “orbit” the nucleus; they are “smeared out” in a cloud which give the atom its size. Incorrect view better view

The number of protons determines the type of element Atomic Number 1 2 3 4 5 6 7 8 Element Hydrogen (H) Helium (He) Lithium (Li) Beryllium (Be) Boron (B) Carbon (C) Nitrogen (N) Oxygen (O)
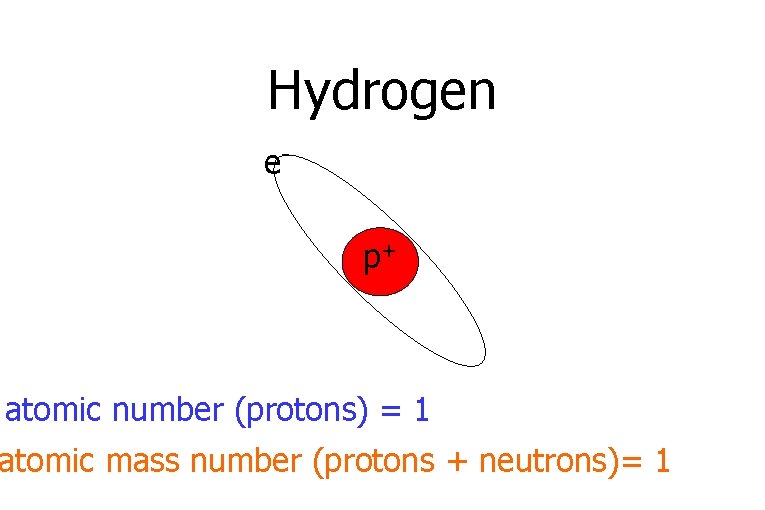
Hydrogen ep+ atomic number (protons) = 1 atomic mass number (protons + neutrons)= 1
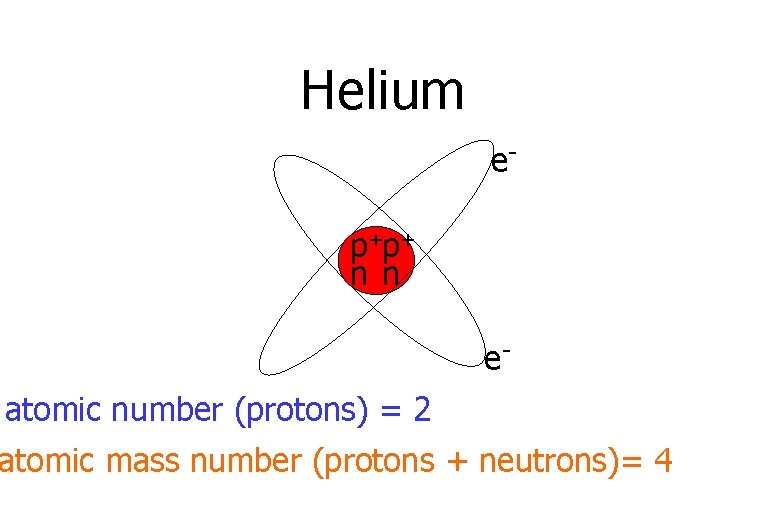
Helium ep+p+ n n eatomic number (protons) = 2 atomic mass number (protons + neutrons)= 4
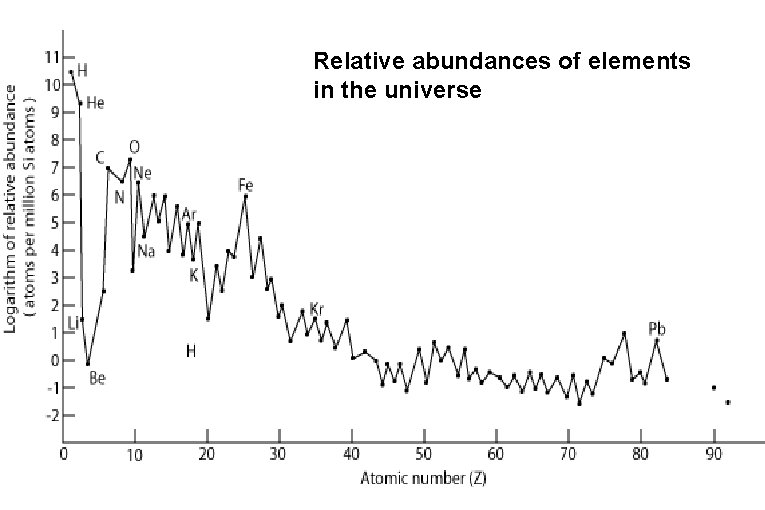
Relative abundances of elements in the universe
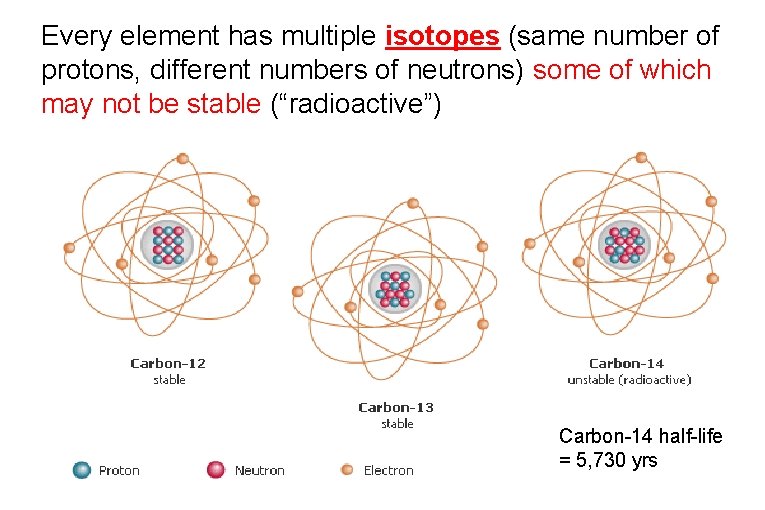
Every element has multiple isotopes (same number of protons, different numbers of neutrons) some of which may not be stable (“radioactive”) Carbon-14 half-life = 5, 730 yrs

Unstable isotopes and radioactivity Unstable (“radioactive”) isotopes “decay”, producing a new type of atom, i. e. , an atom of a different element OR a different isotope of the original element. One half of the atoms of an unstable isotope decay in one “half-life” of that isotope.

Isotopes of Hydrogen ISOTOPE PROTONS NEUTRONS ABUNDANCE HALF LIFE protium 1 0 99. 985% stable deuterium 1 1 0. 015% stable tritium 1 2 ~ 10 -14% 12. 32 yr
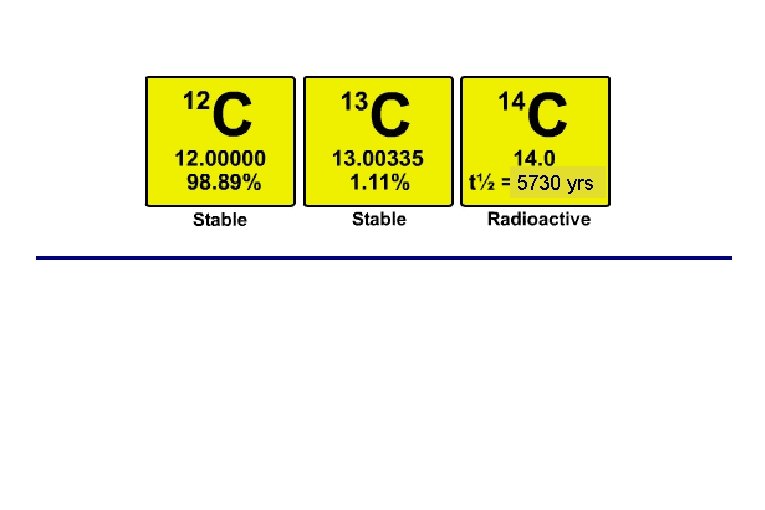
5730 yrs
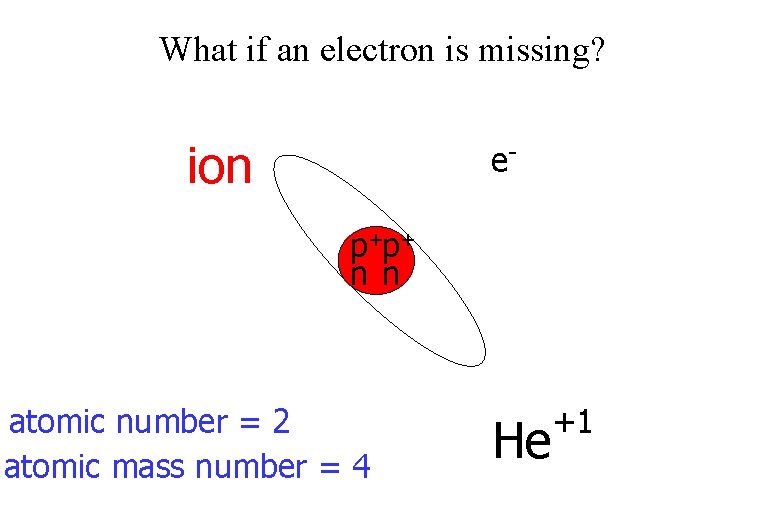
What if an electron is missing? ion ep+p+ n n atomic number = 2 atomic mass number = 4 He +1

What if two or more atoms combine to form a particle? molecule H 2 O (water) p+ Sharing of electrons (chemistry) is involved in the construction of molecules 8 p+ 8 n p+
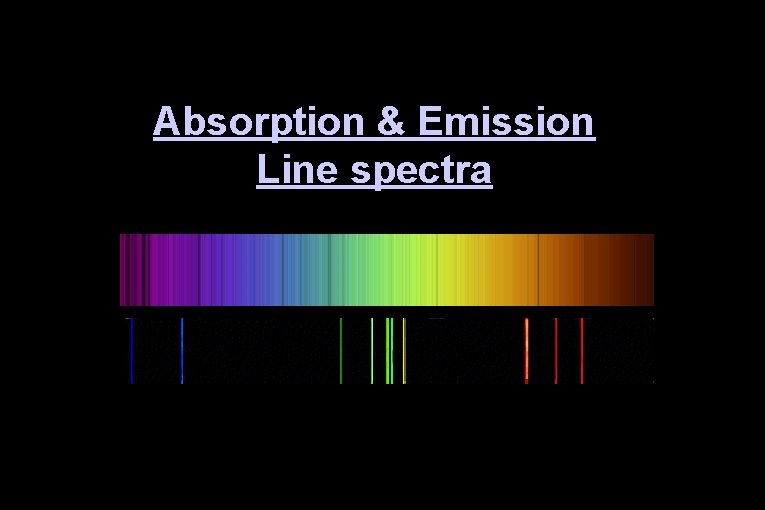
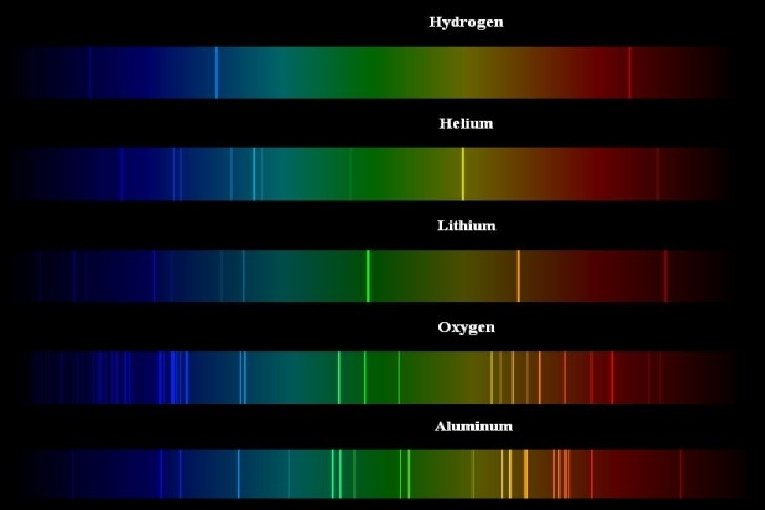
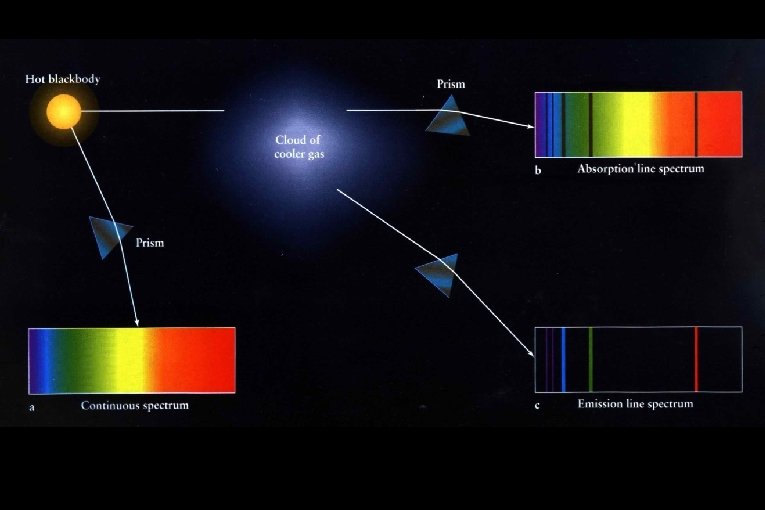
Absorption & Emission Line spectra

Interactive displays of the spectra of most elements can be seen at http: //jersey. uoregon. edu/vlab/elements/Elements. html

Electron Energy Levels Electrons in atoms cannot have just any energy while orbiting the nucleus. Only certain energy values are allowed (like the floors of an aprtment building). Electrons may only gain or lose certain specific amounts of energy (equal to differences in energy levels).

Electron Orbits / Absorption & Emission Electrons can gain or lose energy while they orbit the nucleus. When electrons have the lowest energy possible, we say the atom is in the ground state. When electrons have more energy than this, we say the atom is in an excited state. When electrons gain enough energy to escape the nucleus, we say the atom is ionized. Since energy must be conserved, transitions between energy levels involve the absorption or emission of energy

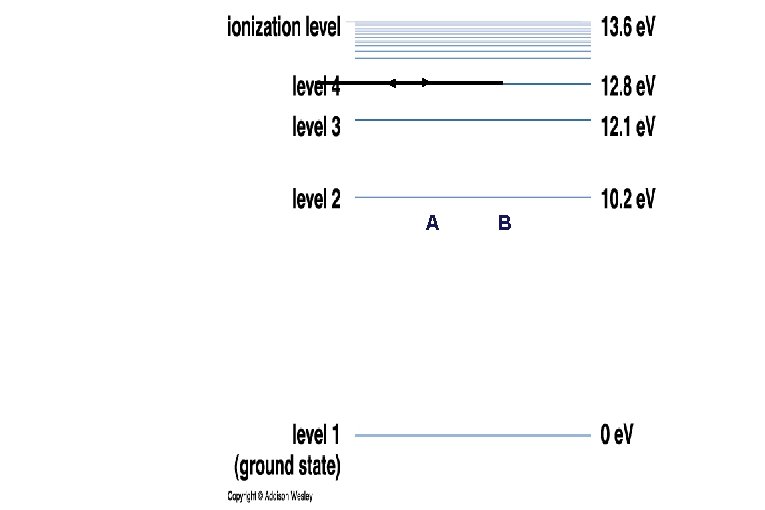
• Each element has its own distinctive set of energy levels for its electrons. • This diagram depicts the energy levels of Hydrogen. 1 e. V = 1. 60 x 10 -19 joules

Emission/Absorption Spectra Hydrogen • Each electron is only allowed to have certain energies in an atom. • Electrons can absorb light and gain energy or emit light when they lose energy. • Only photons whose energies (colors) match the “jump” in electron energy levels can be emitted or absorbed.
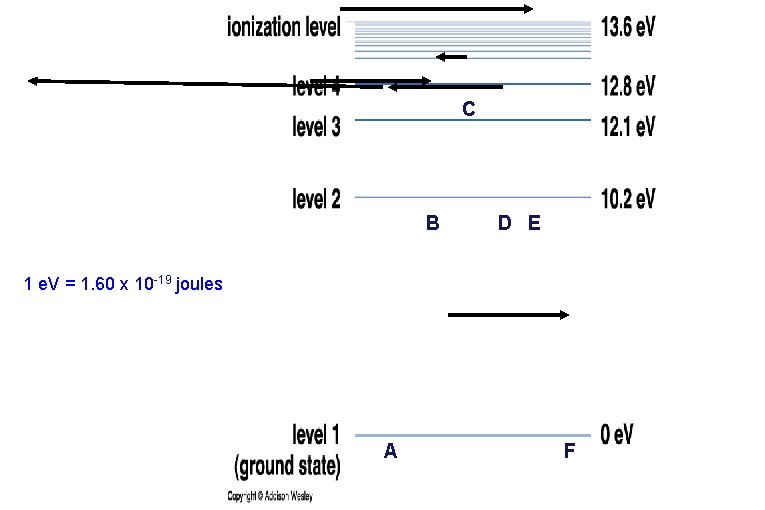
C B D E 1 e. V = 1. 60 x 10 -19 joules A F
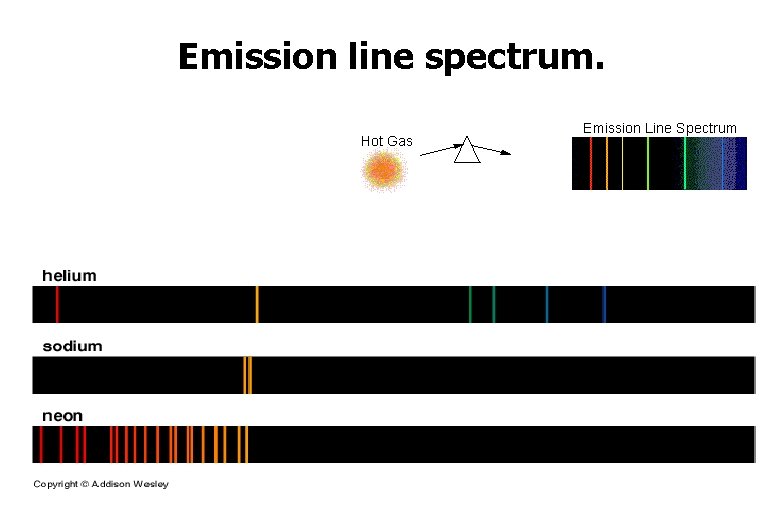
Emission line spectrum.
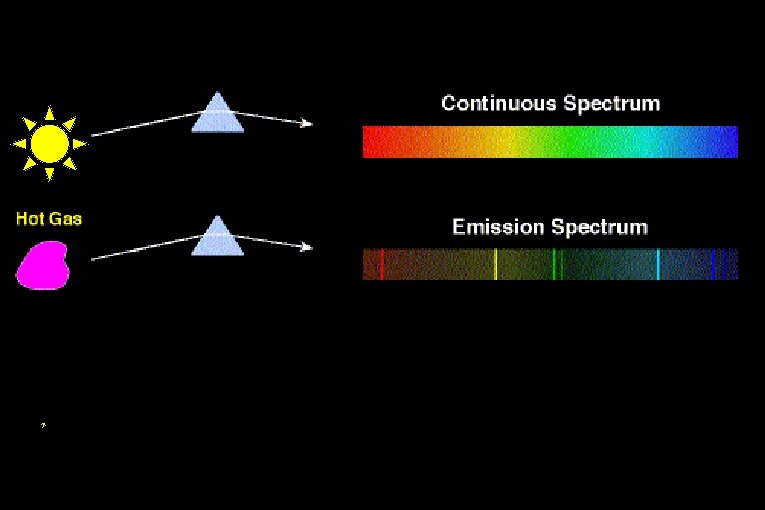
A B
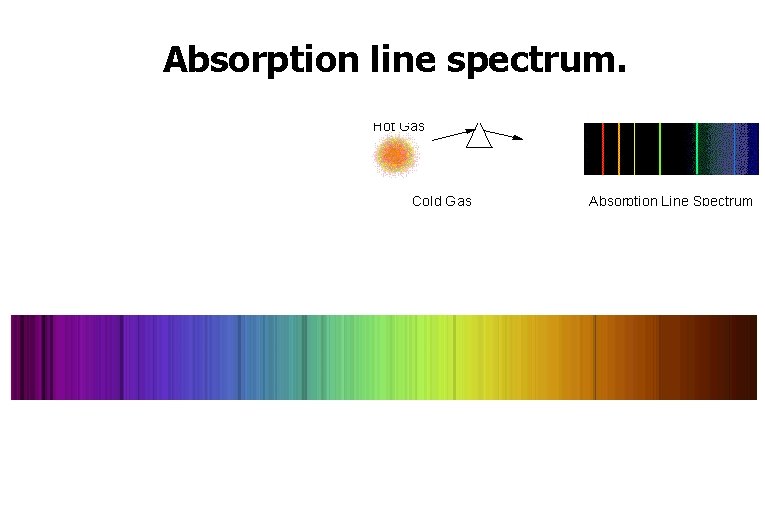
Absorption line spectrum.
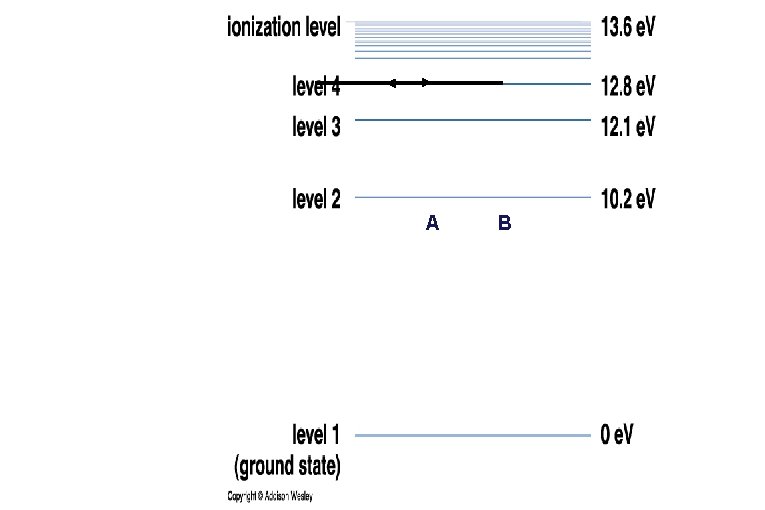
A B

Absorption Spectra • If light shines through a gas, each element will absorb those photons whose energy match their electron energy levels. The resulting absorption line spectrum has all colors minus those that were absorbed.
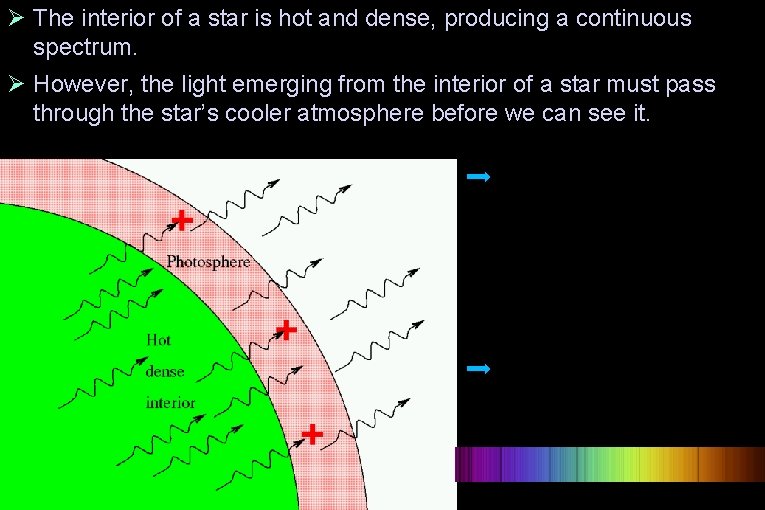
The interior of a star is hot and dense, producing a continuous spectrum. However, the light emerging from the interior of a star must pass through the star’s cooler atmosphere before we can see it.

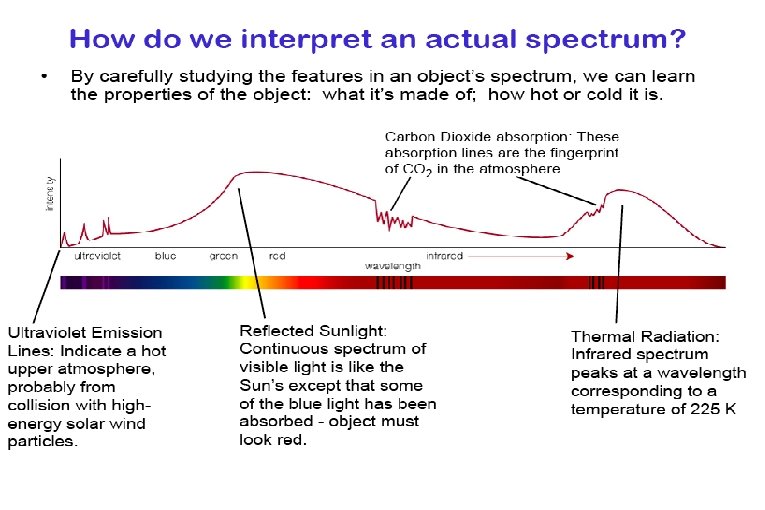
Molecules have rotational & vibrational energy levels. These levels are less energetic than electron energy levels. Therefore energy level transitions involve photons with less energy that visible photons. Energies correspond with photons in the infrared, microwave, and radio portion of the electromagnetic spectrum.
- Slides: 50