LigandGated Ion Channels Molecular Biophysics 28 September 2007

Ligand-Gated Ion Channels Molecular Biophysics 28 September 2007
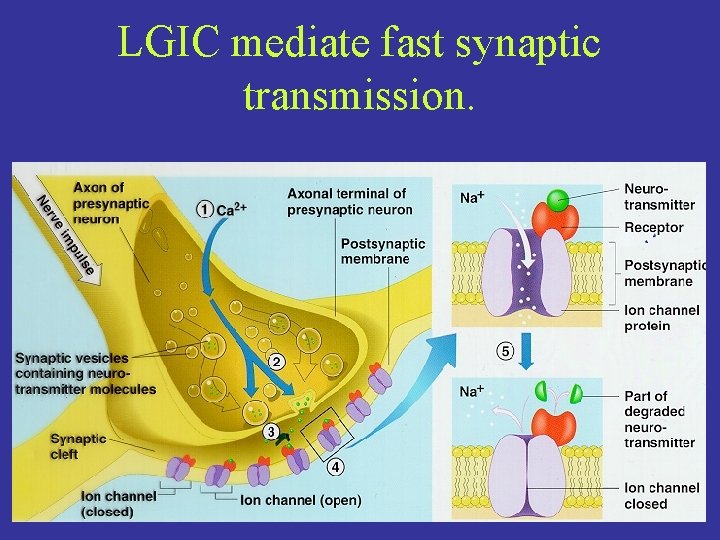
LGIC mediate fast synaptic transmission.
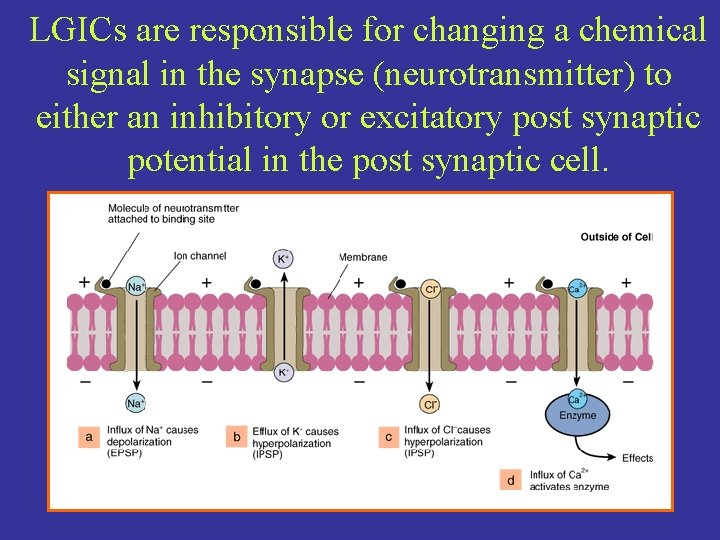
LGICs are responsible for changing a chemical signal in the synapse (neurotransmitter) to either an inhibitory or excitatory post synaptic potential in the post synaptic cell.
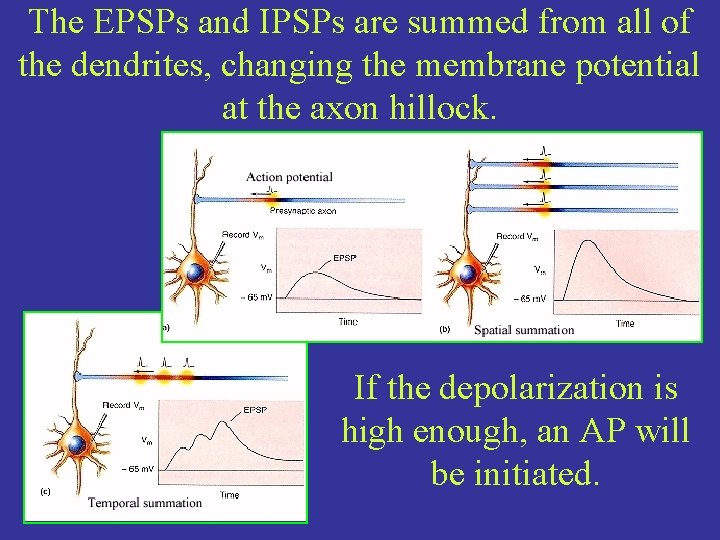
The EPSPs and IPSPs are summed from all of the dendrites, changing the membrane potential at the axon hillock. If the depolarization is high enough, an AP will be initiated.

Fast exchange of bath is needed to study ligand-gated ion channels FSU Neuroscience Website Paul Trombley

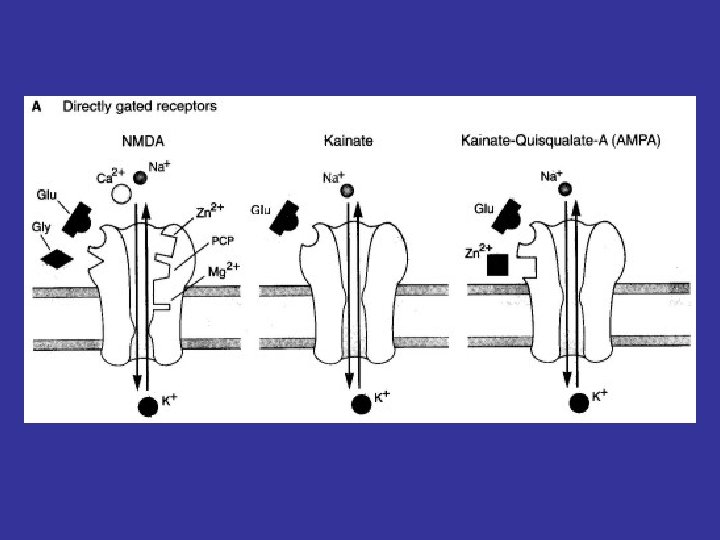
Families of Ligand-Gated Ion Channels • Cys-loop receptors – – Nicotinic Acetylcholine receptor GABAA and GABAC Receptors Glycine Receptor 5 -HT 3 Receptor • Ionotrophic Glutamate Receptors – NMDA – AMPA – Kainate • P 2 X Receptors Kandel, Schwartz & Jessel, Principles of Neural Science 4 th Ed. (2000)

Cystine-Loop Superfamily of Ligand-Gated Ion Channels • Heteromeric or homomeric pentamers • Characterized by a large N-terminal loop cross-linked by cystine bridges • Each subunit is made up of 4 membrane spanning helices • The large intracellular M 3 -M 4 linker is the site for many cytoskeletal protein interactions. • M 2 lines the pore Keramidas et al. , 2004. Cys Ashcroft 2000

Cystine-Loop Superfamily of Ligand-Gated Ion Channels n. ACh. R

n. ACh. R • Activated by Acetylcholine and Nicotine – µs activation times • Blocked by curare and some general anesthetics. • Non-selective cation channel including sodium, potassium and calcium. • Isolated from Torpedo marmorata and visualized by N. Unwin and colleagues in the mid 1980’s
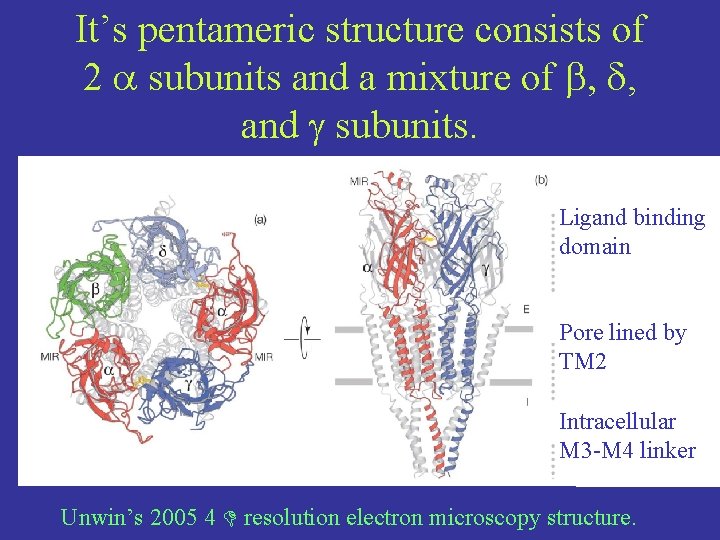
It’s pentameric structure consists of 2 subunits and a mixture of b, d, and g subunits. Ligand binding domain Pore lined by TM 2 Intracellular M 3 -M 4 linker Unwin’s 2005 4 resolution electron microscopy structure.

Open and closed state of the channel at the gate is different by 3 Van der Waal’s surface representation at the gate. Unwin 2003
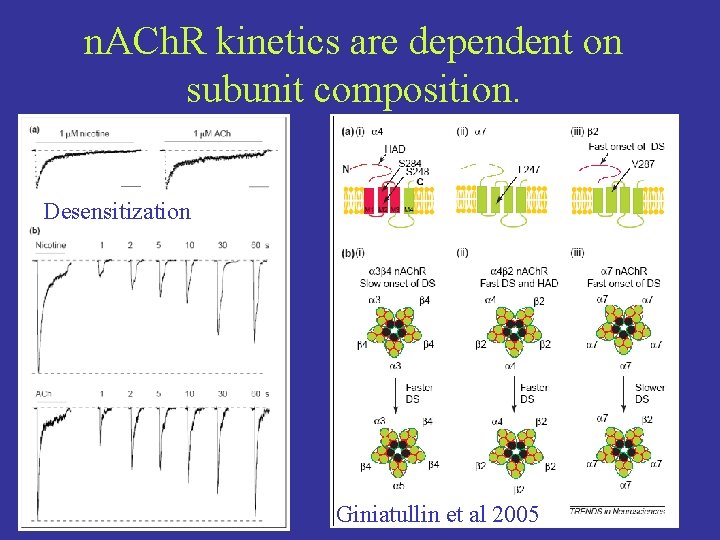
n. ACh. R kinetics are dependent on subunit composition. Desensitization Giniatullin et al 2005

Cystine-Loop Superfamily of Ligand-Gated Ion Channels 5 -HT 3 Receptor
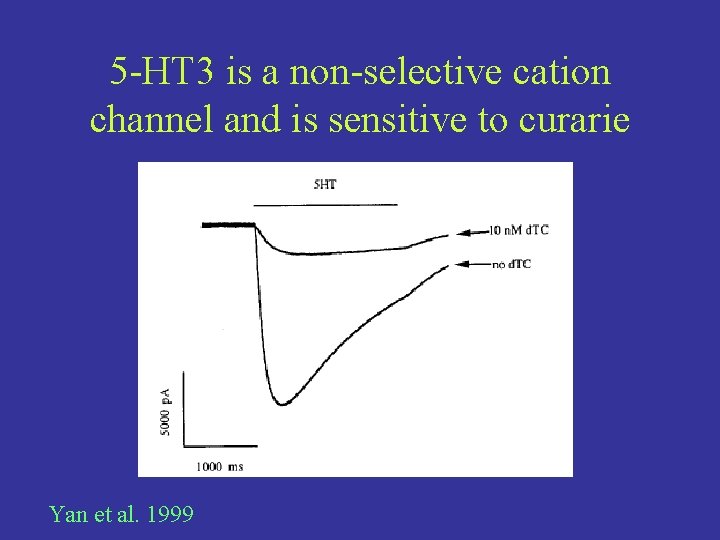
5 -HT 3 is a non-selective cation channel and is sensitive to curarie Yan et al. 1999

Homomultimers and heteromultimers of 5 HT 3 A and 5 -HT 3 B Receptor Subunits produce channels with different characteristics. Peters et al 2005
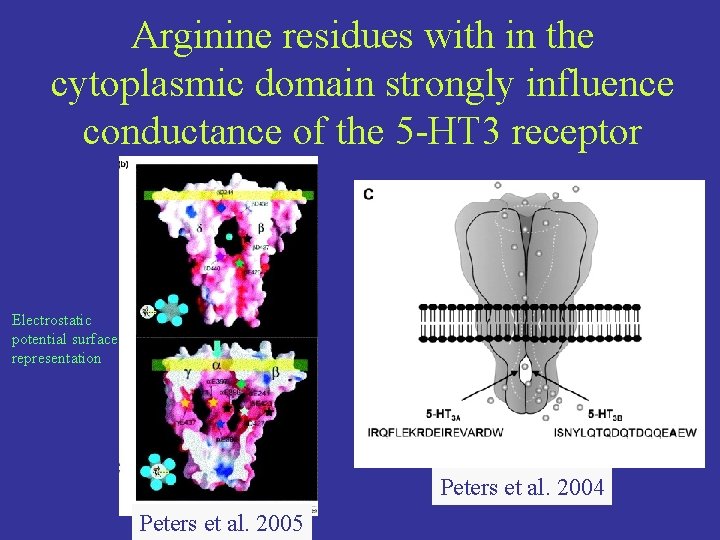
Arginine residues with in the cytoplasmic domain strongly influence conductance of the 5 -HT 3 receptor Electrostatic potential surface representation Peters et al. 2004 Peters et al. 2005

Cystine-Loop Superfamily of Ligand-Gated Ion Channels Glycine and GABA Receptors
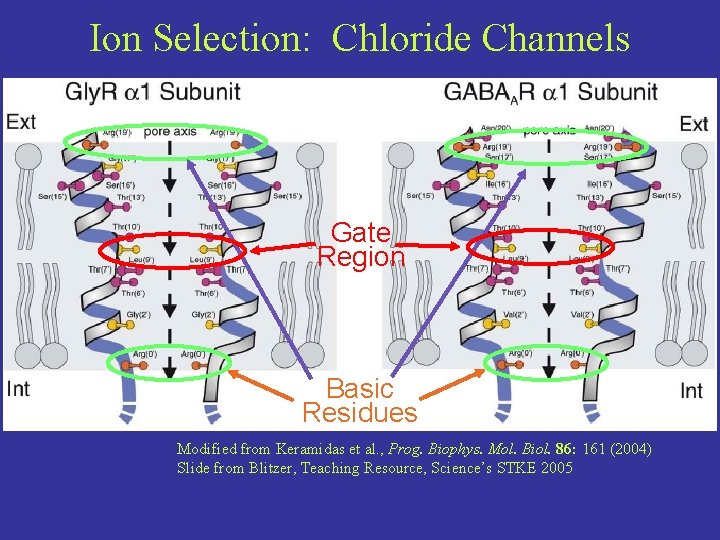
Ion Selection: Chloride Channels Gate Region Basic Residues Modified from Keramidas et al. , Prog. Biophys. Mol. Biol. 86: 161 (2004) Slide from Blitzer, Teaching Resource, Science’s STKE 2005
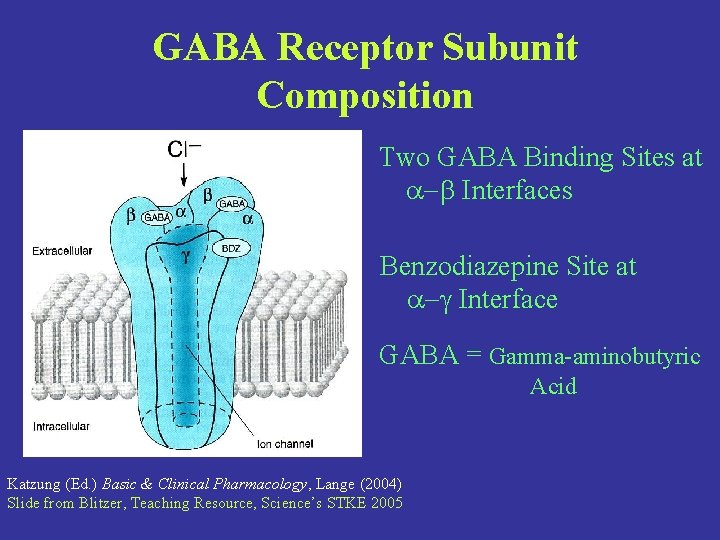
GABA Receptor Subunit Composition Two GABA Binding Sites at -b Interfaces Benzodiazepine Site at -g Interface GABA = Gamma-aminobutyric Acid Katzung (Ed. ) Basic & Clinical Pharmacology, Lange (2004) Slide from Blitzer, Teaching Resource, Science’s STKE 2005
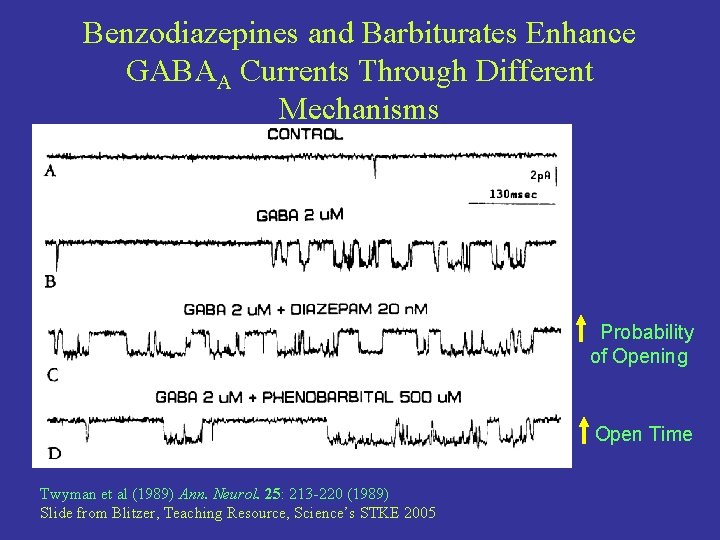
Benzodiazepines and Barbiturates Enhance GABAA Currents Through Different Mechanisms Probability of Opening Open Time Twyman et al (1989) Ann. Neurol. 25: 213 -220 (1989) Slide from Blitzer, Teaching Resource, Science’s STKE 2005
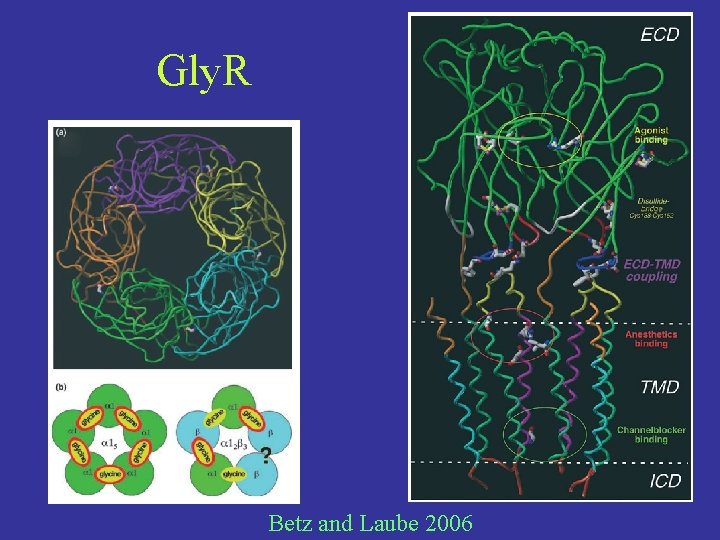
Gly. R Betz and Laube 2006

PNAS
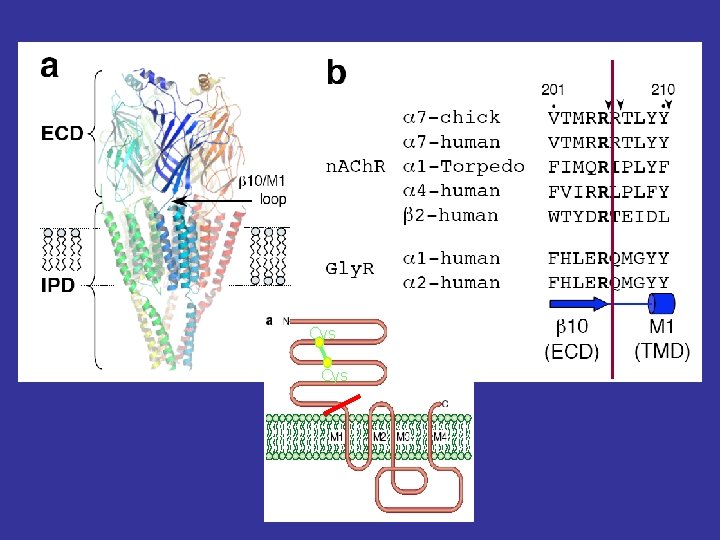
Cys
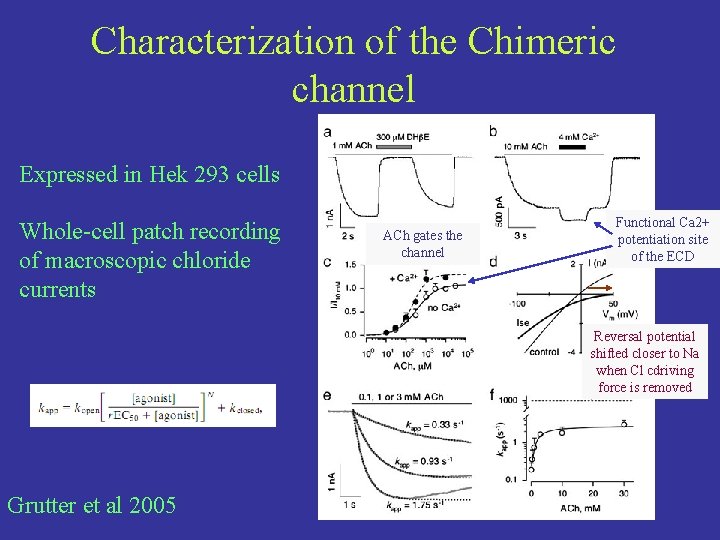
Characterization of the Chimeric channel Expressed in Hek 293 cells Whole-cell patch recording of macroscopic chloride currents ACh gates the channel Functional Ca 2+ potentiation site of the ECD Reversal potential shifted closer to Na when Cl cdriving force is removed Grutter et al 2005

Activation is slowed in the 7/Cly chimeria possibly due to mismatched interactions of the poorly conserved Cys-loop of n. ACh. R and the M 2 -M 3 linker of the Glycine receptor.
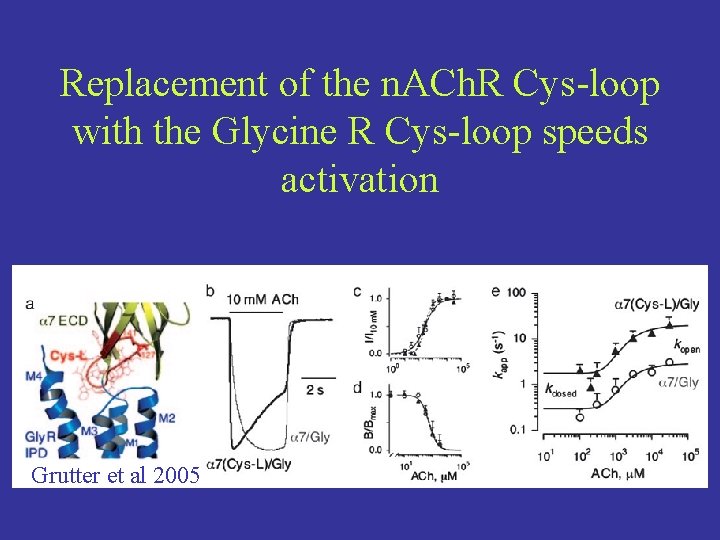
Replacement of the n. ACh. R Cys-loop with the Glycine R Cys-loop speeds activation Grutter et al 2005
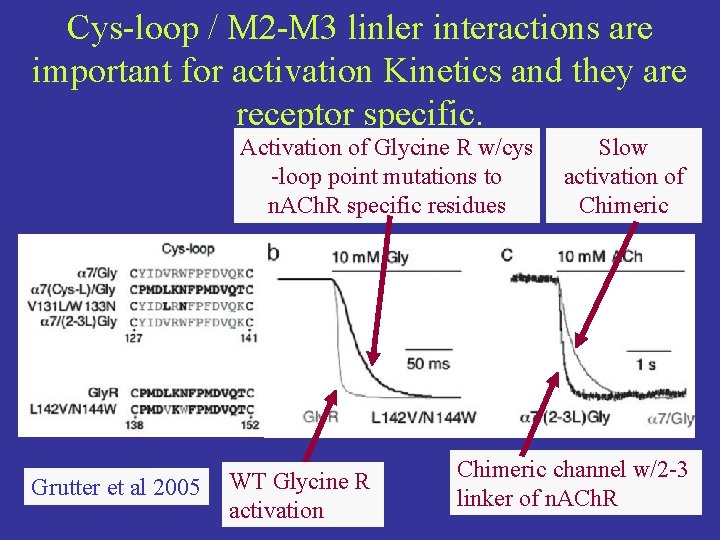
Cys-loop / M 2 -M 3 linler interactions are important for activation Kinetics and they are receptor specific. Activation of Glycine R w/cys -loop point mutations to n. ACh. R specific residues Grutter et al 2005 WT Glycine R activation Slow activation of Chimeric channel w/2 -3 linker of n. ACh. R
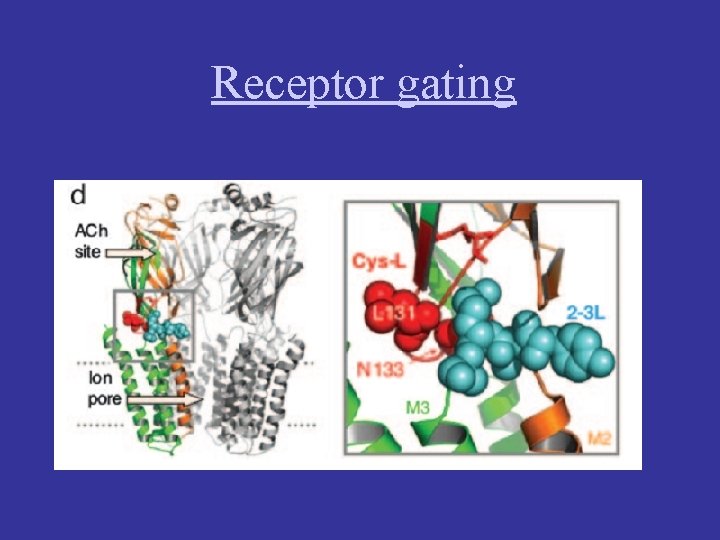
Receptor gating

Ionotrophic Glutamate Receptors

How many subunits make up an ionotrophic glutamate receptor?

Determination of binding sites by single channel electrophysiology • AMPA receptor composed of Glu. R 6/Glu. R 3 chimeric channel expressed in HEK 293 cells – Form homomultimers – No desensitization • The assumptions – # of binding sites = # of subunits – Binding sites must be equivalent
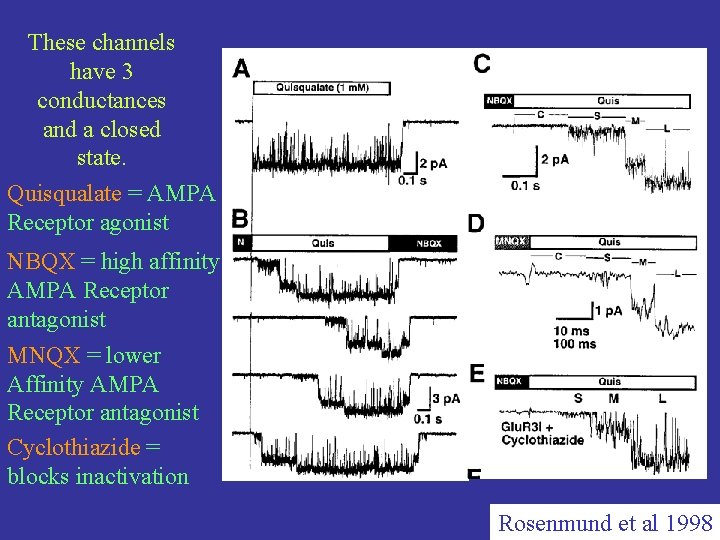
These channels have 3 conductances and a closed state. Quisqualate = AMPA Receptor agonist NBQX = high affinity AMPA Receptor antagonist MNQX = lower Affinity AMPA Receptor antagonist Cyclothiazide = blocks inactivation Rosenmund et al 1998
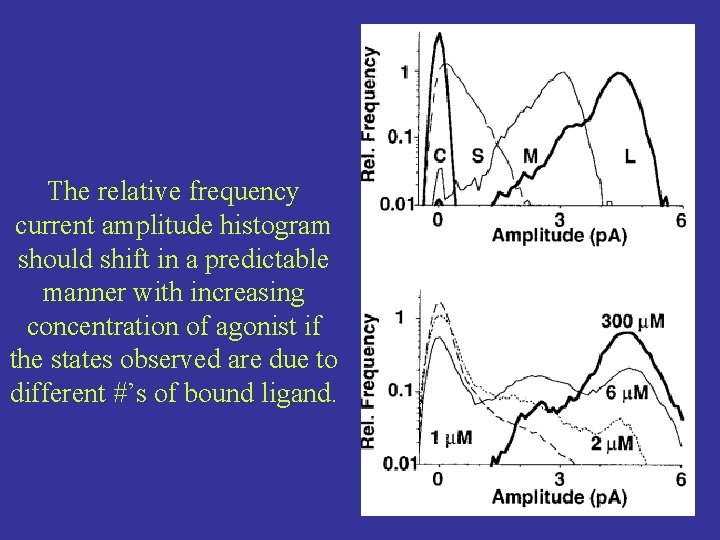
The relative frequency current amplitude histogram should shift in a predictable manner with increasing concentration of agonist if the states observed are due to different #’s of bound ligand.
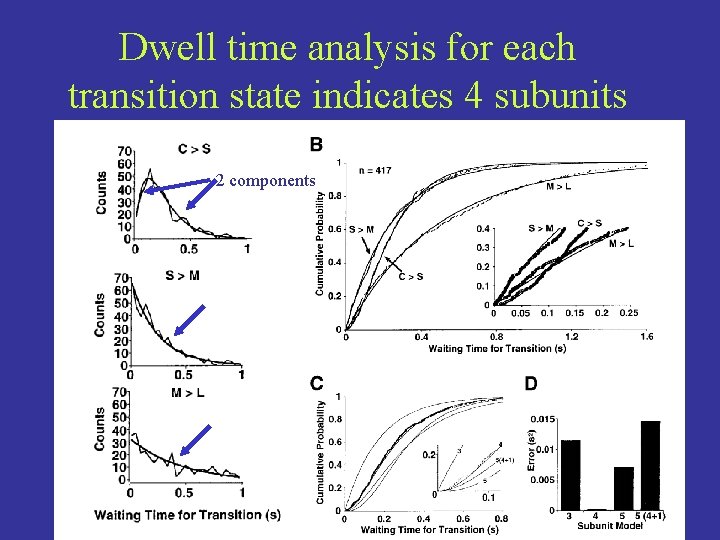
Dwell time analysis for each transition state indicates 4 subunits 2 components

Ionotrophic Glutamate Receptors NMDA Receptors

NMDA Receptors • NMDA = N-methyl-D-aspartic acid • Made up of at least 1 NR 1 subunit and a combination of NR 2 A-D and NR 3 A-B • Permeable to K+, Na+, Ca 2+ • High conductance • Activate slowly • Desensitize slowly & incompletely – Prolonged Ca 2+ influx in the face of sustained glutamate release
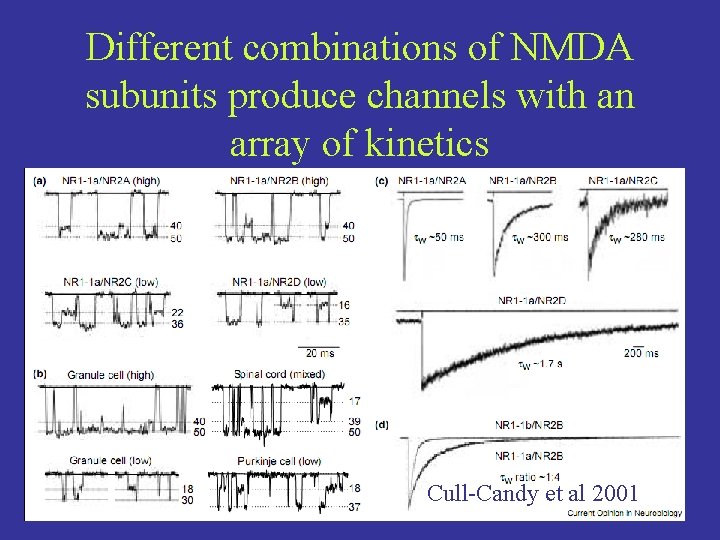
Different combinations of NMDA subunits produce channels with an array of kinetics Cull-Candy et al 2001
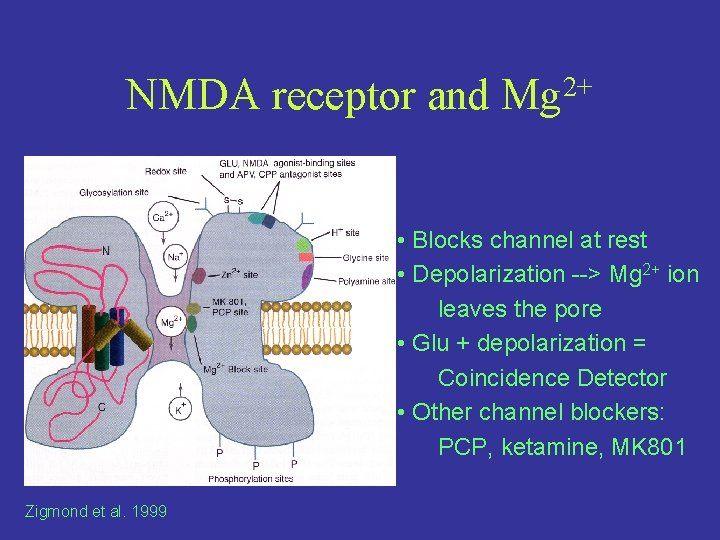
NMDA receptor and 2+ Mg • Blocks channel at rest • Depolarization --> Mg 2+ ion leaves the pore • Glu + depolarization = Coincidence Detector • Other channel blockers: PCP, ketamine, MK 801 Zigmond et al. 1999

Ionotrophic Glutamate Receptors AMPA and Kainate Receptors • • Activate rapidly Desensitize within a few milliseconds Kainate – Glu. R 5 -7, KA 1 -2 AMPA – Glu. R 1 -4 – With Glu. R 2 subunit: permeable only to K+ and Na+ – Without Glu. R 2 subunit: Ca 2+-permeable – AMPA = alpha-amino-3 -hydroxy-5 -methyl-4 isoxazolepropionic acid
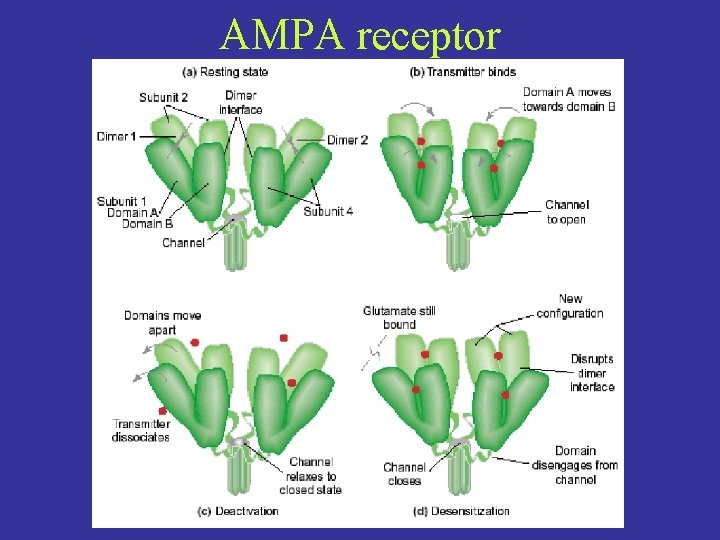
AMPA receptor
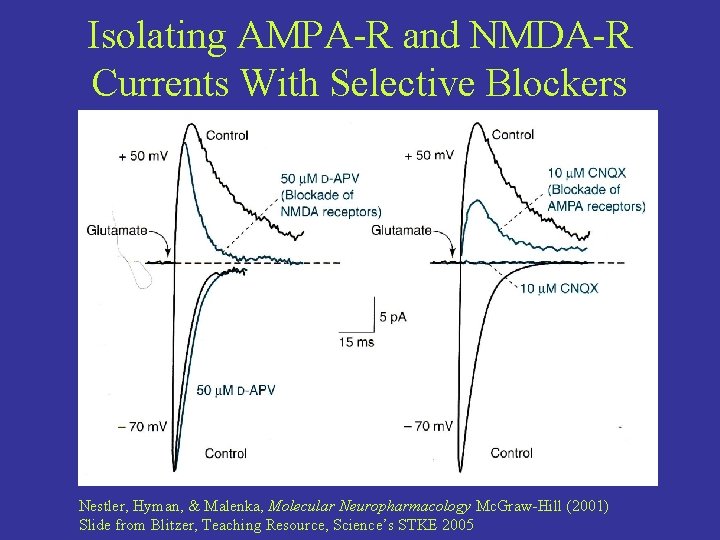
Isolating AMPA-R and NMDA-R Currents With Selective Blockers Nestler, Hyman, & Malenka, Molecular Neuropharmacology Mc. Graw-Hill (2001) Slide from Blitzer, Teaching Resource, Science’s STKE 2005
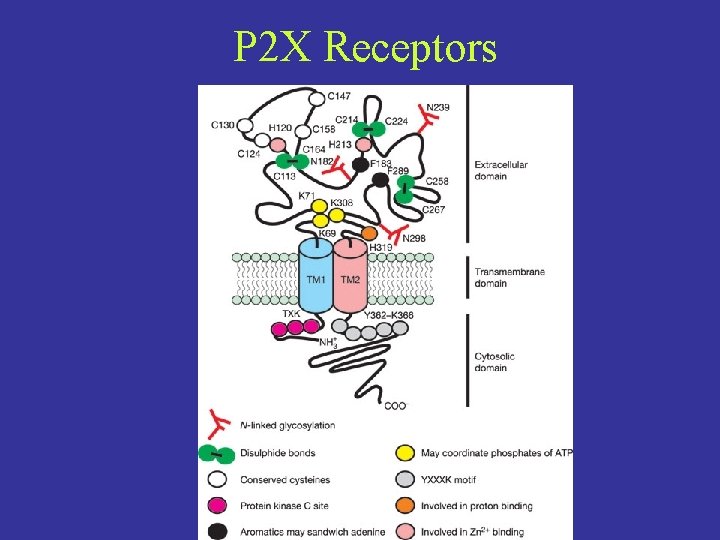
P 2 X Receptors

P 2 X Receptors • Gated by extracellular ATP • Trimeric arrangement determined by crosslinking and agonist binding studies • 7 subtypes, heteromultimers produce a variety of kinetic outcomes • Do not contain common ATP consensus motifs (ie. Walker motif) • M 1 is involved in gating, M 2 lines the pore • Intracellular N and C termini are important for protein-protein interactions
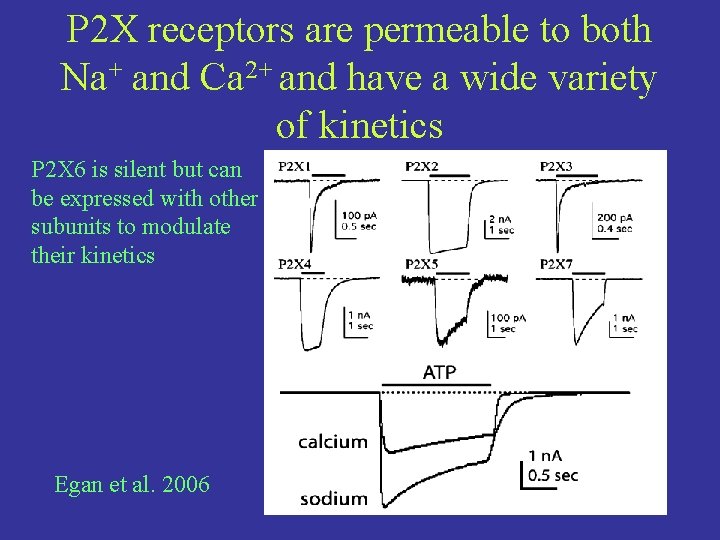
P 2 X receptors are permeable to both Na+ and Ca 2+ and have a wide variety of kinetics P 2 X 6 is silent but can be expressed with other subunits to modulate their kinetics Egan et al. 2006

• • h. P 2 X 1 Cystine point mutations of S 286 -I 329 Oocyte expression, two-electrode voltage-clamp Hek 293 expression, whole-cell patch-clamp
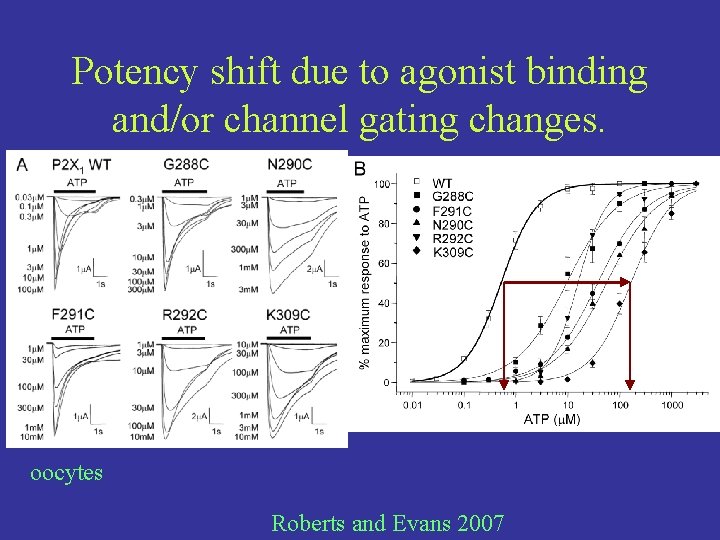
Potency shift due to agonist binding and/or channel gating changes. oocytes Roberts and Evans 2007
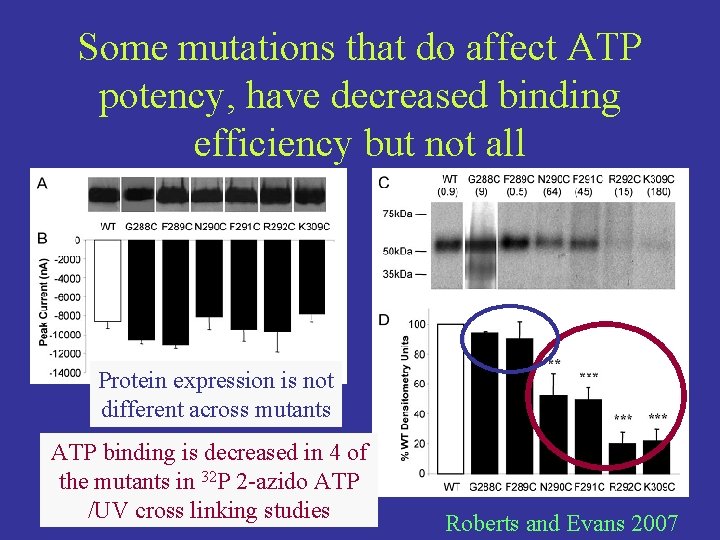
Some mutations that do affect ATP potency, have decreased binding efficiency but not all Protein expression is not different across mutants ATP binding is decreased in 4 of the mutants in 32 P 2 -azido ATP /UV cross linking studies Roberts and Evans 2007
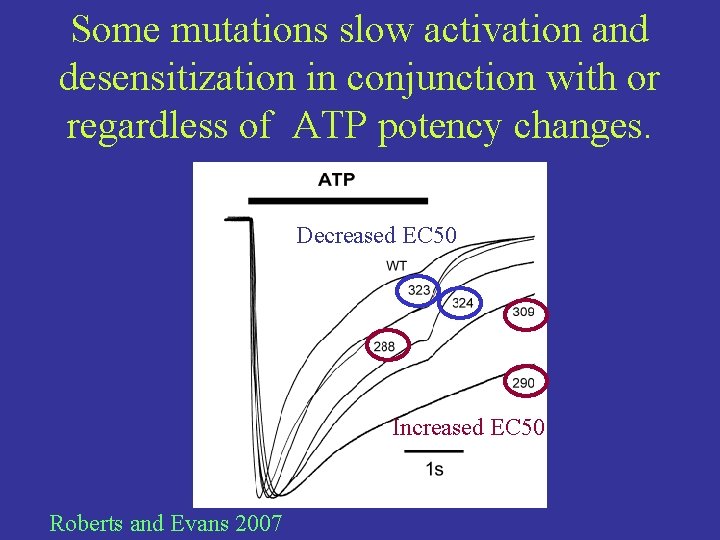
Some mutations slow activation and desensitization in conjunction with or regardless of ATP potency changes. Decreased EC 50 Increased EC 50 Roberts and Evans 2007

Addition of a charge to some mutated residues modulate peak current magnitude. MTS compounds forms disulfide bonds with the side chain of cystine when exposed MTSEA adds a positive charge WT P 2 X 1 does not have an exposed cystine in the region in question MTSES adds a negative charge
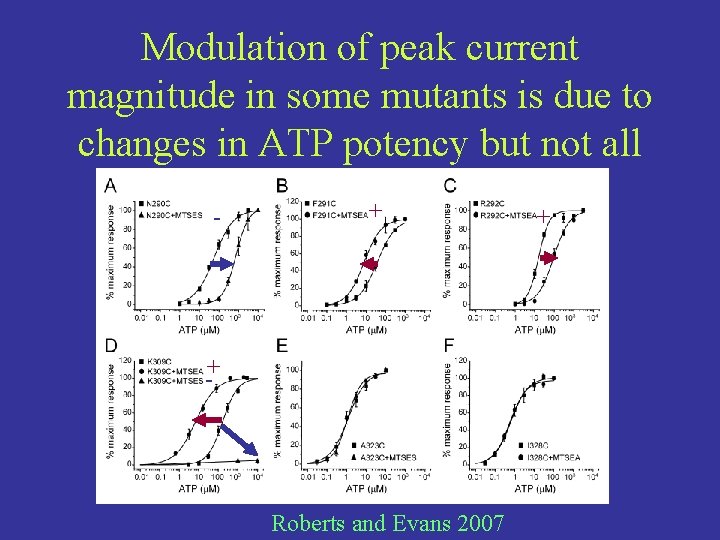
Modulation of peak current magnitude in some mutants is due to changes in ATP potency but not all - + -+ Roberts and Evans 2007 +
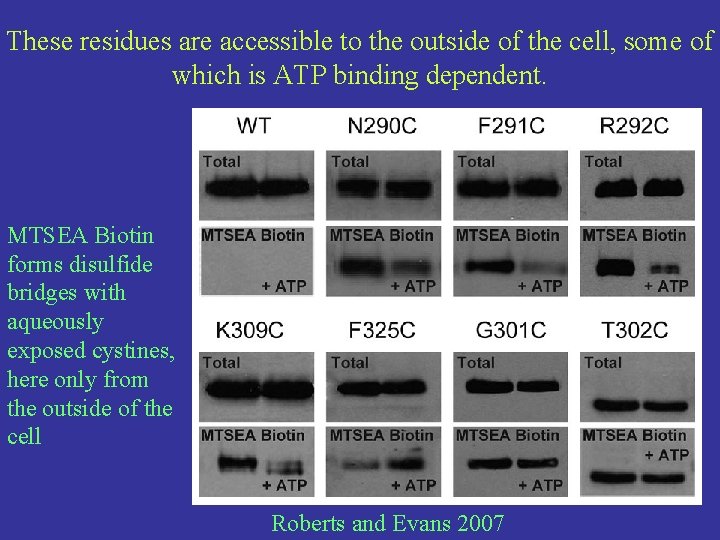
These residues are accessible to the outside of the cell, some of which is ATP binding dependent. MTSEA Biotin forms disulfide bridges with aqueously exposed cystines, here only from the outside of the cell Roberts and Evans 2007
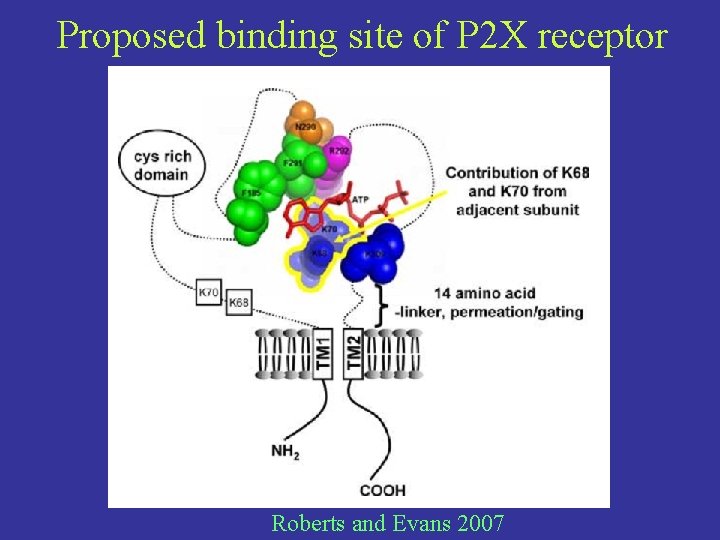
Proposed binding site of P 2 X receptor Roberts and Evans 2007
- Slides: 53