Ligand Field Theory Bonding Combination of Metal and
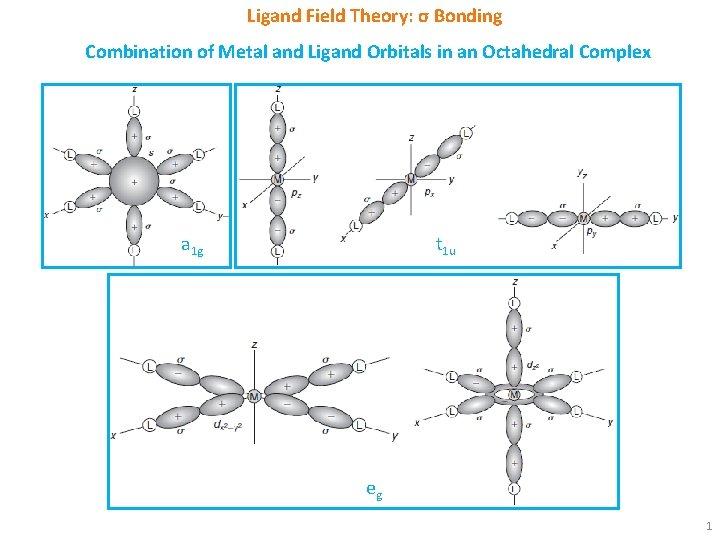
Ligand Field Theory: σ Bonding Combination of Metal and Ligand Orbitals in an Octahedral Complex a 1 g t 1 u eg 1

Ligand Field Theory: σ Bonding § Overlap of metal orbitals and linear combination of ligand group orbitals (LGOs) leads to the formation of molecular orbitals (MOs). § s orbitals transforms as a 1 g, set of p orbitals as t 1 u, whereas five d orbitals lose their degeneracy to form eg (dz 2 and dx 2 -y 2) and t 2 g (dxy, dxz and dyz). § Spherical a 1 g orbitals are capable to overlap with ligand group orbitals (LGOs) on all axes. § t 1 u and eg sets have lobes along bond directions and participate in bonding. § However, t 2 g set have lobes directed between the bonding axis and thus will yield no overlap with ligand orbitals. § Electronic configuration for [Co(NH 3)6]3+: a 1 g 2 t 1 u 6 eg 4 t 2 g 6 § Electronic configuration for [Co. F 6]3 -: a 1 g 2 t 1 u 6 eg 4 t 2 g 4 eg*2 2
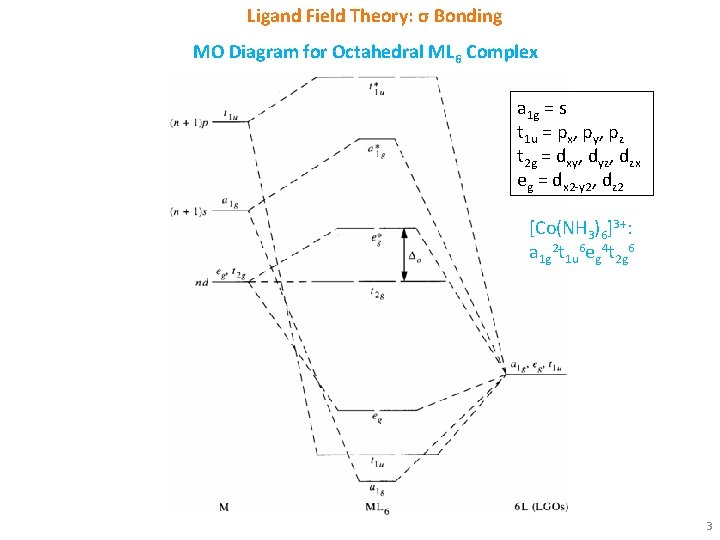
Ligand Field Theory: σ Bonding MO Diagram for Octahedral ML 6 Complex a 1 g = s t 1 u = px, py, pz t 2 g = dxy, dyz, dzx eg = dx 2 -y 2, dz 2 [Co(NH 3)6]3+: a 1 g 2 t 1 u 6 eg 4 t 2 g 6 3
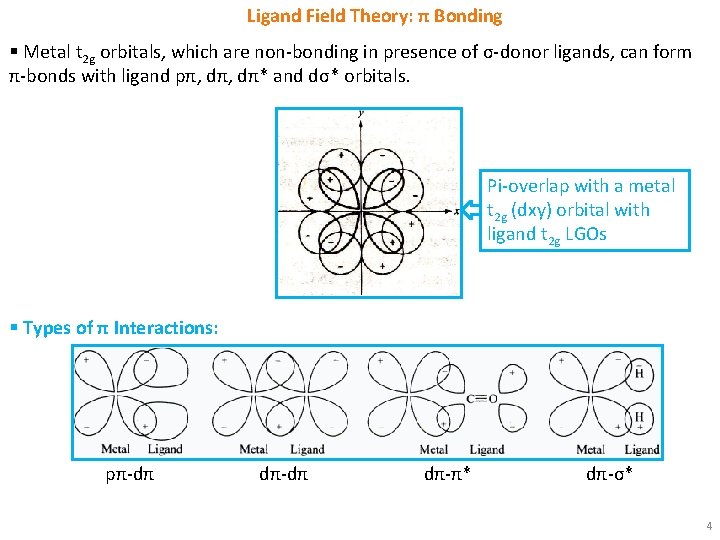
Ligand Field Theory: π Bonding § Metal t 2 g orbitals, which are non-bonding in presence of σ-donor ligands, can form π-bonds with ligand pπ, dπ* and dσ* orbitals. Pi-overlap with a metal t 2 g (dxy) orbital with ligand t 2 g LGOs § Types of π Interactions: pπ-dπ dπ-π* dπ-σ* 4
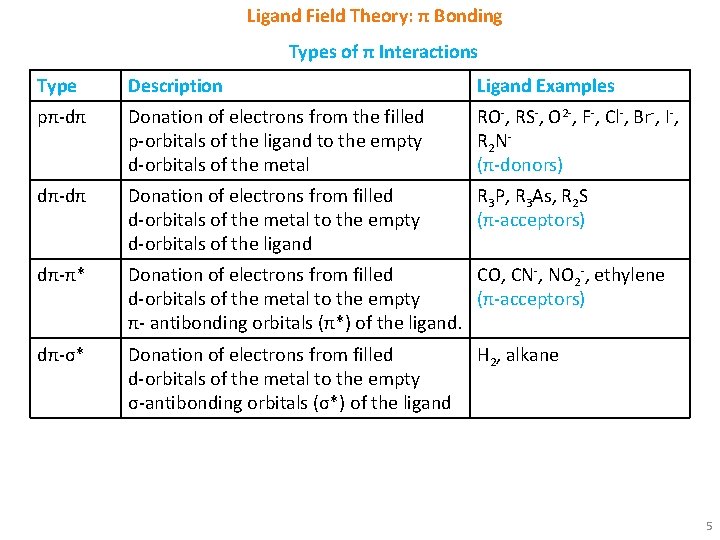
Ligand Field Theory: π Bonding Types of π Interactions Type Description Ligand Examples pπ-dπ Donation of electrons from the filled p-orbitals of the ligand to the empty d-orbitals of the metal RO-, RS-, O 2 -, F-, Cl-, Br-, I-, R 2 N(π-donors) dπ-dπ Donation of electrons from filled d-orbitals of the metal to the empty d-orbitals of the ligand R 3 P, R 3 As, R 2 S (π-acceptors) dπ-π* Donation of electrons from filled CO, CN-, NO 2 -, ethylene d-orbitals of the metal to the empty (π-acceptors) π- antibonding orbitals (π*) of the ligand. dπ-σ* Donation of electrons from filled d-orbitals of the metal to the empty σ-antibonding orbitals (σ*) of the ligand H 2, alkane 5
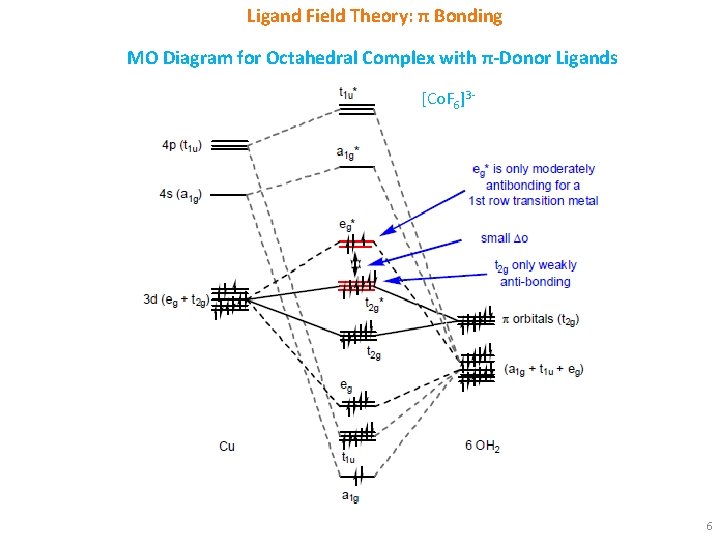
Ligand Field Theory: π Bonding MO Diagram for Octahedral Complex with π-Donor Ligands [Co. F 6]3 - 6
![Ligand Field Theory: π Bonding MO Diagram for Octahedral Complex with π-Acceptor Ligands [Cr(CO)6] Ligand Field Theory: π Bonding MO Diagram for Octahedral Complex with π-Acceptor Ligands [Cr(CO)6]](http://slidetodoc.com/presentation_image_h/937bd828051a2033bed243ac61bab432/image-7.jpg)
Ligand Field Theory: π Bonding MO Diagram for Octahedral Complex with π-Acceptor Ligands [Cr(CO)6] 7
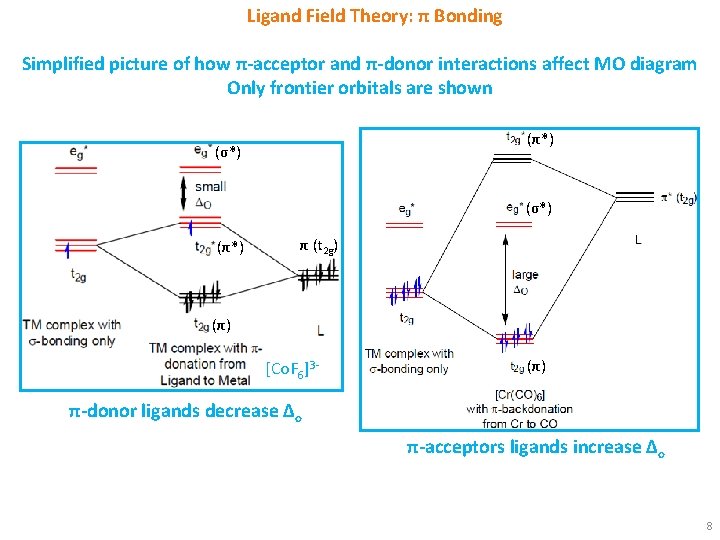
Ligand Field Theory: π Bonding Simplified picture of how π-acceptor and π-donor interactions affect MO diagram Only frontier orbitals are shown (π*) (σ*) (π*) π (t 2 g) (π) [Co. F 6]3 - (π) π-donor ligands decrease Δo π-acceptors ligands increase Δo 8

Ligand Field Theory: π Bonding § π-Donor Ligands: These ligands (e. g. , F-) are more electronegative than metal and has filled p orbitals. Hence, the orbitals are lower in energy than the metal d orbitals. The 3 t 2 g metal orbitals and 3 low-lying, filled ligand orbitals form 3 bonding MOs and 3 antibonding MOs. § The electrons from the ligand p orbitals will fill the bonding π-orbitals. The electrons from the metal d orbitals will be present in the π* orbitals. The eg* orbitals are not affected. The previously nonbonding metal t 2 g orbitals become antibonding and hence are raised in energy. As a result, Δo decreases. This is the reason for halides being weak ligands in spectrochemical series in spite of their negative charges. § π-Acceptor Ligands: These ligands are less electronegative than metal and has empty orbitals (d orbitals for PR 3, π* orbitals for CO). Hence, the orbitals are higher in energy than the metal d orbitals. The 3 t 2 g metal orbitals and 3 high-lying, empty ligand orbitals form 3 bonding MOs and 3 antibonding MOs. § The electrons from the metal d orbitals will fill the bonding π-orbitals. The e g* orbitals remain empty and they are not affected. The previously nonbonding metal t 2 g orbitals become bonding and hence are lowered in energy. As a result, Δo increases. This is the reason for PR 3 and CO being strong ligands in spectrochemical series, although they are neutral ligands. 9
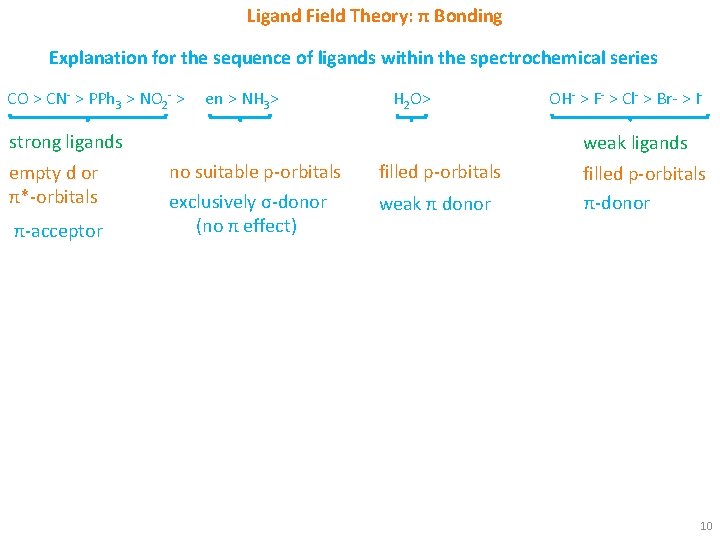
Ligand Field Theory: π Bonding Explanation for the sequence of ligands within the spectrochemical series CO > CN- > PPh 3 > NO 2 - > en > NH 3> H 2 O> strong ligands empty d or π*-orbitals π-acceptor OH- > F- > Cl- > Br- > I- weak ligands no suitable p-orbitals exclusively σ-donor (no π effect) filled p-orbitals weak π donor filled p-orbitals π-donor 10
- Slides: 10