Life depends on hydrogen bonds in water Water


Life depends on hydrogen bonds in water. • Water is the most abundant compound in living things. • Some of water’s properties that facilitate an environment for life are: • • Cohesive and adhesive behavior Ability to moderate temperature (high specific heat) Expansion upon freezing Versatility as a solvent http: //www. sumanasinc. com/webcontent/animations/content/propert iesofwater/water. html

Polarity & Hydrogen Bonding in Water • The water molecule is a polar molecule: the opposite ends have opposite charges. Slightly negative and slightly positive charge Water is polar because the oxygen atom has a slightly negative charge and the hydrogen atoms have slightly positive charges. • Water shares electrons unequally! • Polarity allows water molecules to form hydrogen bonds with each other. These are weak covalent bonds. • • Ionic > Covalent > Hydrogen

Cohesion & Adhesion Cohesion • Hydrogen bonds hold water molecules together, cohesion: • Cohesion is the attraction of molecules of like substance. • Water molecules attracted to other water molecules • Examples: Water forming beads on a car, puddles of water, tear drops Adhesion • The clinging of one substance to a different substance • Water molecules stick to other things. • Examples: Water sticking to glass mirror, water sticking to your skin

Cohesion + Adhesion = Capillary Action • Cohesion and adhesion work together to give capillarity – the ability of water to spread through fine pores or to move upward through narrow tubes against the force of gravity. • Cohesion due to hydrogen bonding contributes to the transport of water and dissolved nutrients against gravity in plants. Adhesion of water to cell walls by hydrogen bonds helps to counter the downward pull of gravity on the liquids passing through plants Significance: The process of water moving through plants against the force of gravity. • •
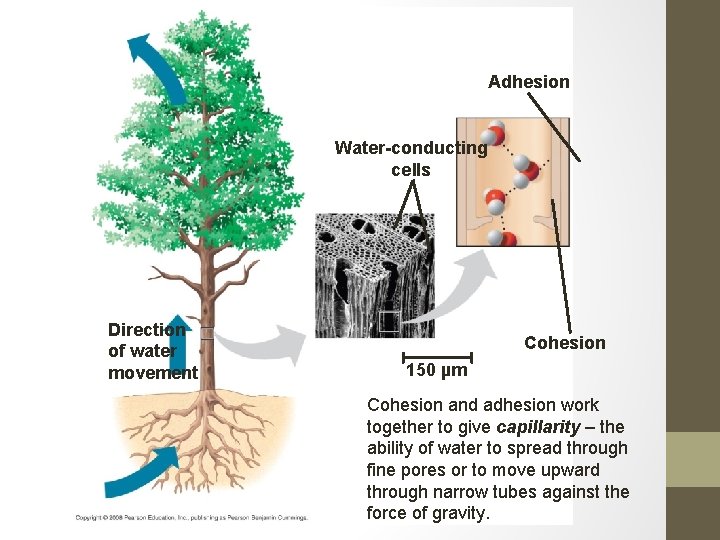
Adhesion Water-conducting cells Direction of water movement Cohesion 150 µm Cohesion and adhesion work together to give capillarity – the ability of water to spread through fine pores or to move upward through narrow tubes against the force of gravity.

Surface Tension • Surface tension is directly related to the cohesive property of water • It is a measurement of how difficult it is to stretch or break the surface of a liquid. • Example: Water Strider • Significance: allows the water strider to walk on the surface of the pond to escape a predator or find food

High Specific Heat Moderation of Temperature • Water can absorb or release a large amount of heat with only a slight change in its own temperature. • The ability of water to stabilize temperature results from its relatively high specific heat: • Water’s high specific heat can be traced to hydrogen bonding. • H-Bonds require a lot of energy to break the bonds • Significance: Helps minimize temperature fluctuations by regulating the rates of water and air temperatures causing temperatures to gradually change rather than suddenly (Season Changes)

The Density Anomaly • Ice floats in liquid water because hydrogen bonds in ice are more “ordered, ” making ice less dense. • Significance: If ice sank, all bodies of water would eventually freeze solid, making life impossible on Earth.

The Solvent of Life Universal Solvent • Water provides living systems with excellent dissolving capabilities. • Can dissolve several substances because of its electronegative charge • A solution is a liquid that is a homogeneous mixture of substances • • Solvent (dissolving agent) Greater Amount Solute (substance that is dissolved) Lesser Amount • Significance: Breakdown food, Plasma (liquid part of blood) is 95% water allowing sugar and nutrients to dissolve in the water of blood plasma because they are polar

Acids and Bases • An acid is any substance that increases the H+ concentration of a solution. • Acid p. H= below 7 ( 0 -6) • Neutral = p. H of 7 neither acidic or basic • A base is any substance that reduces the H+ concentration of a solution. • Base p. H = above 7 ( 8 – 14)
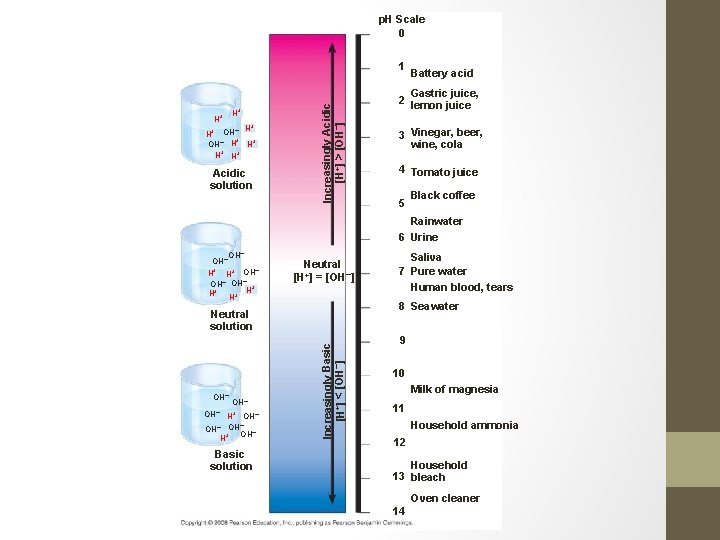
p. H Scale 0 H+ H+ + – H H+ OH– H H+ H+ H+ Acidic solution Increasingly Acidic [H+] > [OH–] 1 Battery acid Gastric juice, 2 lemon juice 3 Vinegar, beer, wine, cola 4 Tomato juice 5 Black coffee Rainwater 6 Urine OH– H+ OH– OH– + H+ H+ H Neutral [H+] = [OH–] 8 Seawater OH– H+ OH– – OH OH– + H Basic solution Increasingly Basic [H+] < [OH–] Neutral solution OH– Saliva 7 Pure water Human blood, tears 9 10 Milk of magnesia 11 Household ammonia 12 Household 13 bleach Oven cleaner 14

Water Summary Video http: //vimeo. com/37064053
- Slides: 13