Lewis Structures visualize molecular structure assume most atoms






- Slides: 8

Lewis Structures visualize molecular structure assume most atoms will have octet of electrons 1. Determine number of valence electrons 2. Place least electronegative atom at center 3. Using e- pairs, bond central atom to surrounding atoms 4. Complete octets on surrounding atoms 5. Place remaining e- on central atom. Use multiple bonds to complete octet, if necessary

nitrogen trifluoride NF 3 valence e- 5 + (3 x 7)= 26 e- N= 5 F= 7 : : : : F : N : F : : : : : : F N F: : F : : central atom = N bond F to N complete octets surrounding atoms complete octet central atom include lone pairs 6 e 24 e 26 e-

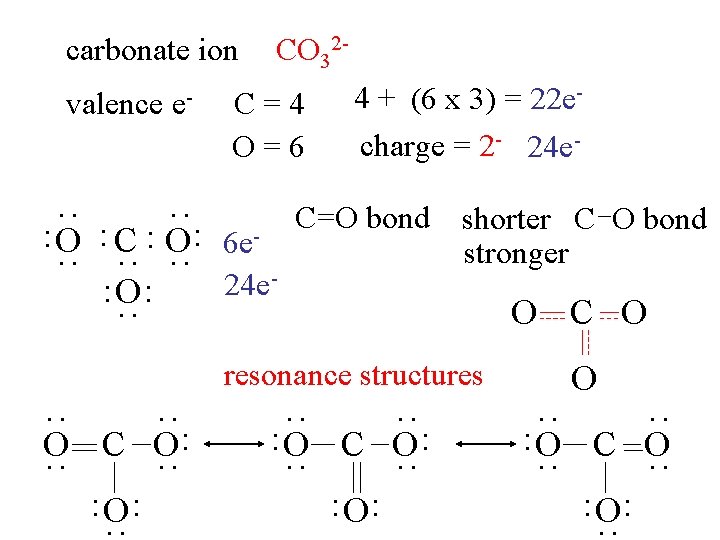
carbonate ion valence e- CO 32 - C=4 O=6 : : : O : C : O: 24 e : O: 6 e- 4 + (6 x 3) = 22 echarge = 2 - 24 e- C=O bond shorter C O bond stronger : O C O resonance structures : : : O C O : : : O: : : O C O: : : O C O: O

formaldehyde CH 2 O 4 + 6 + (2 x 1) = 12 e 4 - 0 - 4 = 0 which is best structure? H : C: : O: H formal charge 6 - 2 - 3= 1 compare e- in isolated atom to : : : : H : C: H : O : 4 - 2 - 3 = -1 : 6 - 4 - 2 = 0 e- in Lewis structure formal charge = valence e- - lone pair e- - ½ shared eformal charge = 0 preferred . . C. . . N. . . O. . .

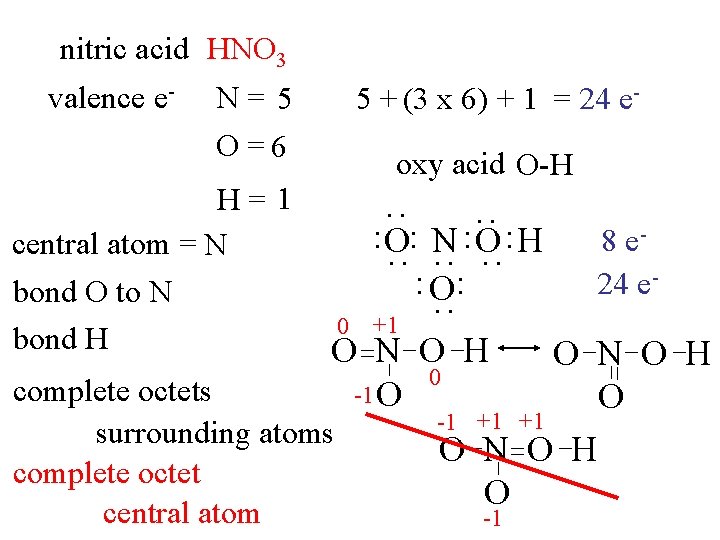
nitric acid HNO 3 valence e- N= 5 O =6 oxy acid O-H : : O: N : O : H : O: : : 0 +1 O N O H 0 -1 O complete octets surrounding atoms complete octet central atom 8 e 24 e- : H= 1 central atom = N bond O to N bond H 5 + (3 x 6) + 1 = 24 e- O N O H O -1 +1 +1 O N O H O -1

Exceptions to octet rule 1. Molecules with odd number of electrons Lewis structures electron pairs unpaired e- free radical : N: : O : : : N: O : start with 12 e- : : 5+6= : : NO 11 e- 5 - 4 - 2 = -1 5 - 3 - 2 = 0 6 - 3 - 2 = +1 6 - 4 - 2 = 0

Exceptions to octet rule 2. Incomplete octets (fewer than 8 e-) B Be (Al) B : F: : F : : : boron trifluoride BF 3 F: 3 + (3 x 7)= 24 e-

Exceptions to octet rule : : Cl : : : P : : 3. Expanded octet more than 8 eonly Period 3 and above must have d orbitals phosphorous pentachloride PCl 5 5 + (5 x 7)= 40 e- : Cl Cl :