Lewis Structure Review Hybridization Objective Today I will

Lewis Structure Review Hybridization

Objective Today I will be able to: Determine the exceptions to the octet rule Predict the molecular shape of a molecule using the VSEPR theory Apply hybridization to determining the orbital shape of a molecule Evaluation/ Assessment Informal assessment – Listening to group interactions as they complete the exeptions to the octet Lewis Structure Practice Formal Assessment – Analyzing student responses to the exit ticket and the Lewis Structure Practice. Common Core Connection Make sense of problem and persevere in solving them Reason abstractly and quantitatively Use appropriate tools strategically Look for and make use of structure

Lesson Sequence Warm – Up Evaluate: Review Lewis Structure Answers Explain: Hybridization Notes Elaborate: Hybridization Practice Evaluate: Exit Ticket

Warm - Up Draw the following Lewis Structures SF 6 SF 4 ICl 3

Warm-Up What is the hybridization for a molecule of CCl 4?

Objective Today I will be able to: Predict the molecular shape of a molecule using the VSEPR theory Apply hybridization to determining the orbital shape

Homework Abstract Corrections Due Monday, December 22 STEM Fair Board Due January 9 Molecular Shapes/ Hybridization Quiz Monday, December 22

Agenda Warm – Up Review VSEPR Practice Hybridization Exit Ticket

VSEPR Theory Practice Work with the people in your row. We will review the answers as a class

Draw the Lewis Structures for the following molecules Determine the shape Determine if the molecule is polar or nonpolar CH 3 Cl CH 2 O Be. Cl 2 BCl 3 PF 5 SF 6 Xe. F 4 SF 4 ICl 3
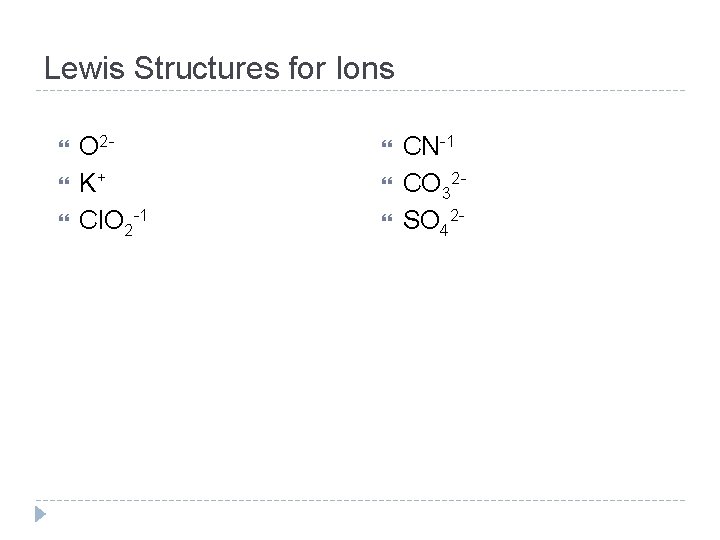
Lewis Structures for Ions O 2 K+ Cl. O 2 -1 CN-1 CO 32 SO 42 -
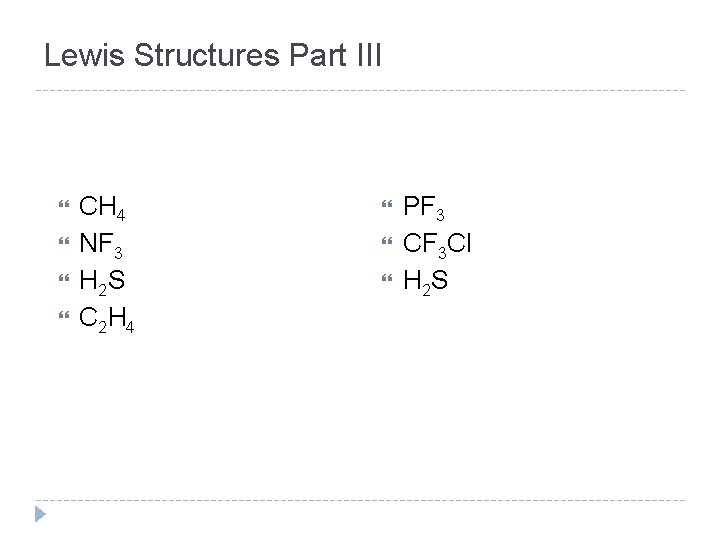
Lewis Structures Part III CH 4 NF 3 H 2 S C 2 H 4 PF 3 CF 3 Cl H 2 S

Hybridization

Hybridization

What is hybridization? Atoms use their valence electrons to form bonds. But how is it that they form bonds of equal energy when some of the electrons come from the s orbital and some come from the p orbital? ? Hybrid orbitals are orbitals of equal energy (between the energy of s & p orbitals) produced by the combination of two or more orbitals on the same atom.

Hybridization An atom in a molecule may adopt a different set of atomic orbitals (called hybrid orbitals) than those it has in the free state.

The hybridization of a particular molecule is determined by the central atom. We only need to worry about it’s valence electrons.

Your Hybridization Options: sp sp 2 sp 3 d 2 sp 3 d 3

Hybridization Be. F 2 Look at B Write the orbital diagram for B You need to have 2 e- available to bond to F Write a new orbital diagram Promote electrons
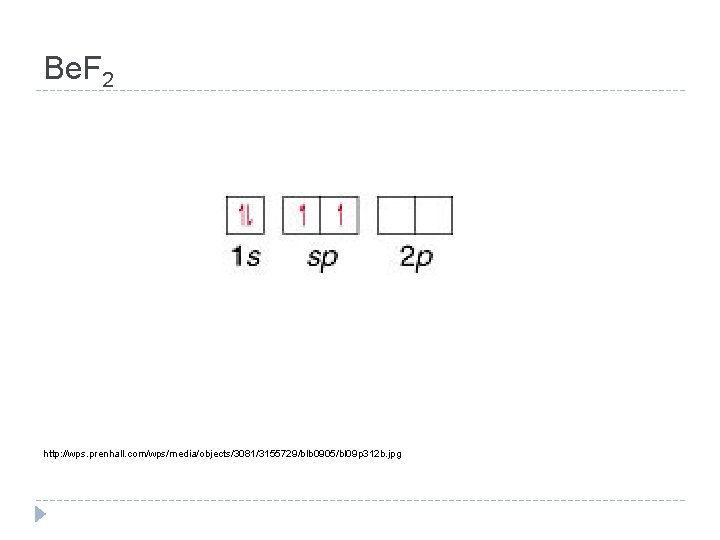
Be. F 2 http: //wps. prenhall. com/wps/media/objects/3081/3155729/blb 0905/bl 09 p 312 b. jpg

Hybridization BCl 3 Look at B Write the orbital diagram for B Promote electrons
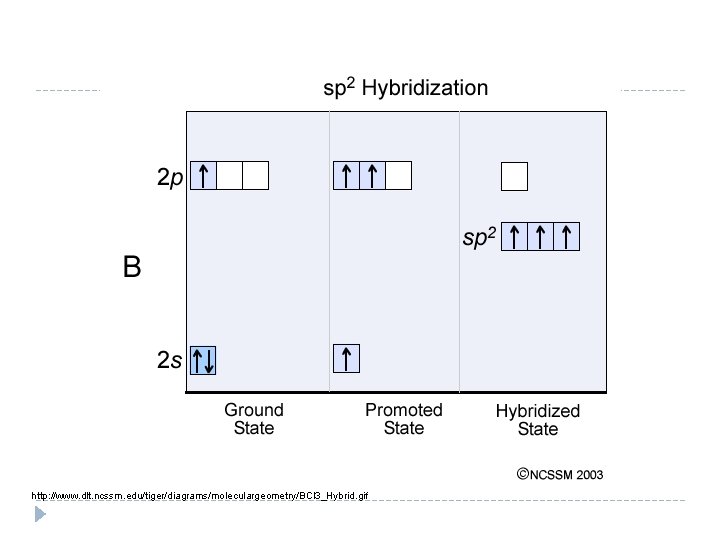
http: //www. dlt. ncssm. edu/tiger/diagrams/moleculargeometry/BCl 3_Hybrid. gif

Consider CH 4 Carbon has 4 valence electrons 1 s 2 2 p 2 2 of the electrons are in the s orbital and 2 are in the p orbital. s & p have different shapes and different amounts of energy.
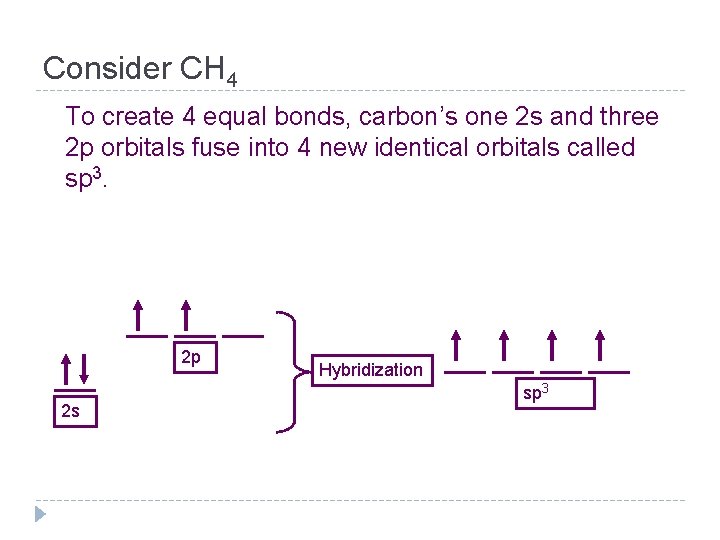
Consider CH 4 To create 4 equal bonds, carbon’s one 2 s and three 2 p orbitals fuse into 4 new identical orbitals called sp 3. 2 p 2 s Hybridization sp 3
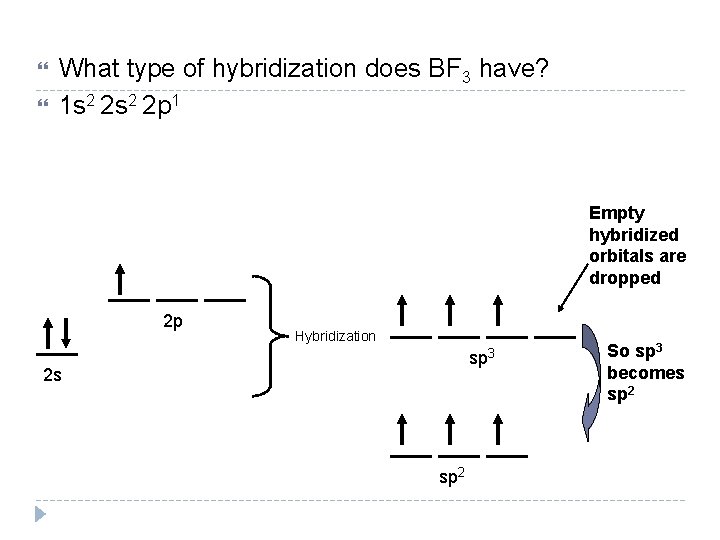
What type of hybridization does BF 3 have? 1 s 2 2 p 1 Empty hybridized orbitals are dropped 2 p Hybridization sp 3 2 s sp 2 So sp 3 becomes sp 2

How do you include lone pairs of e- in hybridization? Each lone pair of electrons has it’s own hybridized orbital. See next slide.
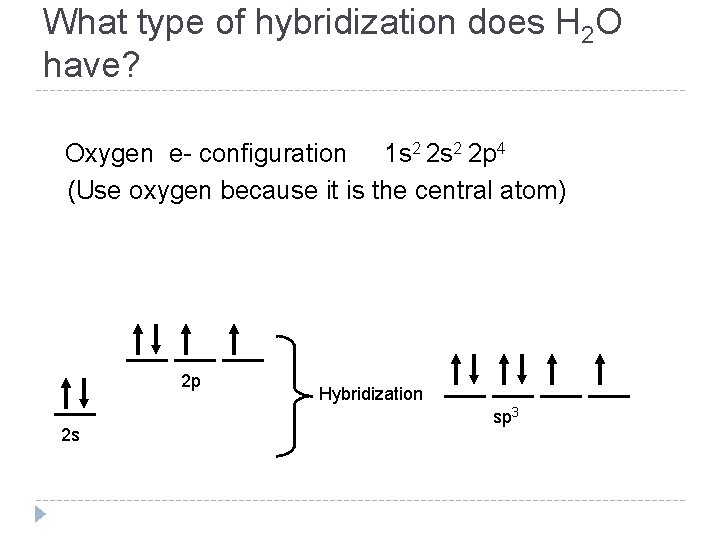
What type of hybridization does H 2 O have? Oxygen e- configuration 1 s 2 2 p 4 (Use oxygen because it is the central atom) 2 p 2 s Hybridization sp 3

Draw the hybridization orbital diagram for phosphorus in PCl 3

Try this hybridization animation http: //www. mhhe. com/physsci/chemistry/essentialche mistry/flash/hybrv 18. swf

Exit Ticket Determine the hybridization of the following molecules Xe. F 4 PCl 5 NH 3

Exit Ticket – Mol Monday # 7 How many molecules of diphosphorous pentoxide are in 5. 0 grams?
- Slides: 31