Lewis Dot structures Created by Gilbert N Lewis

Lewis Dot structures Created by Gilbert N. Lewis Devised a system of representing atoms based on the number of valence electrons in the atom. These diagrams are referred as Lewis dot structures Use the main group elements in group 1, 2, 13, 14, 15, 16, 17, and 18.
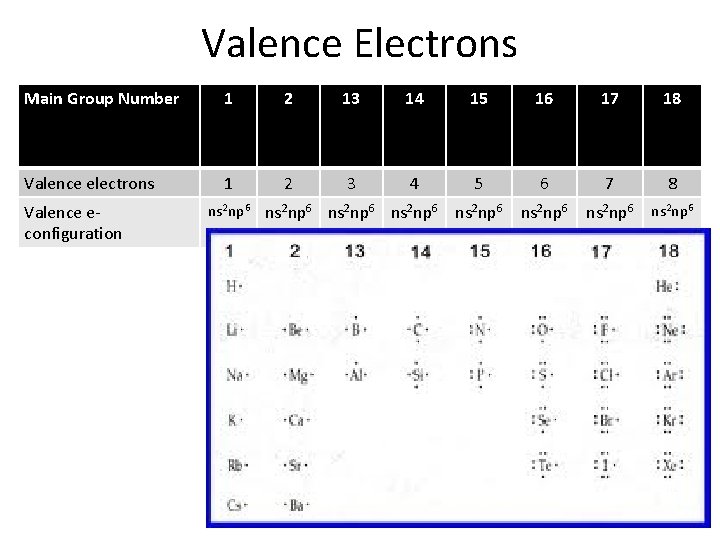
Valence Electrons Main Group Number 1 2 13 14 15 16 17 18 Valence electrons 1 2 3 4 5 6 7 8 ns 2 np 6 Valence econfiguration ns 2 np 6 ns 2 np 6

Steps for writing Lewis Dot Structures 1. Write the element symbol. 2. Place a dot around the element for each valence electron. 3. For each valence electron place one dot first at each position above, below, left and to the right of the element 4. Only after all four positions are fill you may start pairing the electrons.

Important rules: Each element in a family has the same number if valence electrons, and therefore the same number of dots. For metals the total number of dots represents the number of electrons that each atom loses forming a cation. For non-metals the number of unpaied erepresents the number of electrons they must either share or gain to make a stable octet
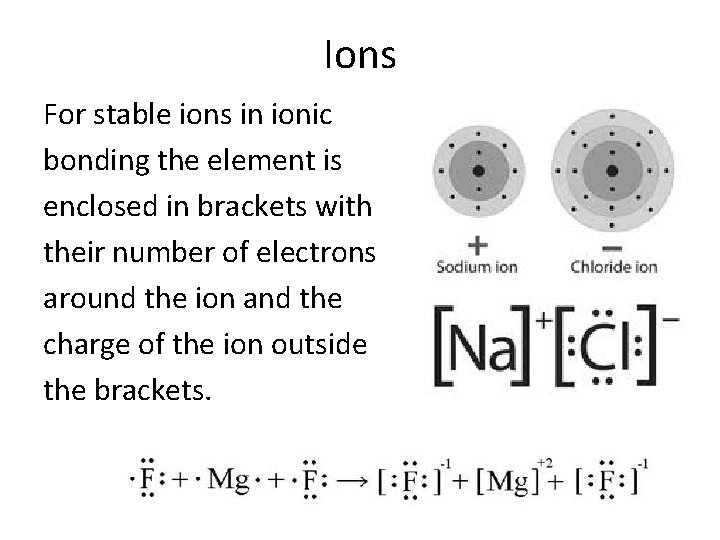
Ions For stable ions in ionic bonding the element is enclosed in brackets with their number of electrons around the ion and the charge of the ion outside the brackets.

Quick Check: What do the dots in the Lewis structure represent? In groups 1, 2, 13 what does the total number of dots tell us about the cations they’ll form? Group 15 elements have one pair and three unpaired dots. How many electrons are need to fill the octet? Practice Problems page 327, 1 -3.
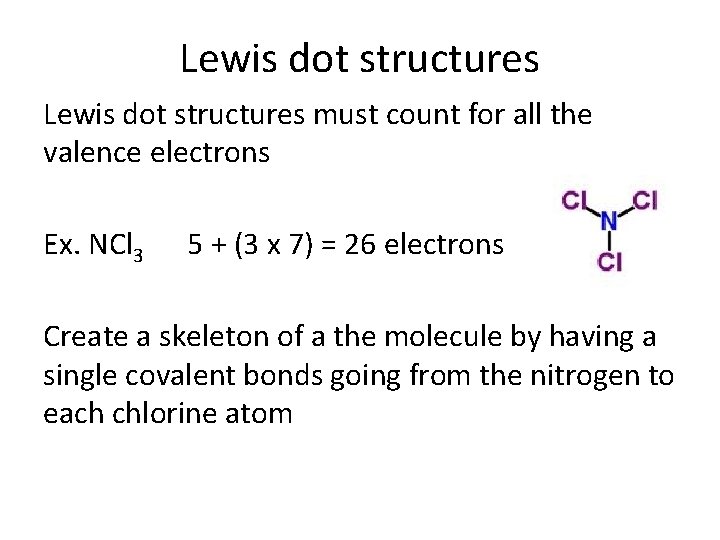
Lewis dot structures must count for all the valence electrons Ex. NCl 3 5 + (3 x 7) = 26 electrons Create a skeleton of a the molecule by having a single covalent bonds going from the nitrogen to each chlorine atom
Creating the skeleton General compound formula AXn A is generally a lower group number, having a lower electronegativity. Remember hydrogen only forms one bond Halogens typically only form one bond Oxygen forms two bonds hand rarely with itself when other atoms are present Nitrogen makes three bonds Carbon has four bond Avoid making rings when you draw structures

Subtract the number of valence electrons use to create the skeleton to determine the number remaining. Ex. NCl 3 26 - 6 = 20 electrons remaining Assume all atoms except hydrogen obey the octet rule, fill in the electrons around the chlorine and nitrogen atoms. Now count the number of electrons you added to the atoms to fill their octet and if they equal the remaining you have a viable lewis dot structure

DO NOT violate the octet rule!!! Exception: Be, B, and Al often have incomplete octets, resulting in fewer electrons. Hypervalency- to have more than 8 electrons by using d orbitals. Phosphorus and Sulphur are capable of this

Quick Check Q. 1 -3 Practice problems Q. 1 -3 Page 330 -331

Multiple Bonds When more then one pair of electrons are shared between two electrons. If two pairs of electrons are shared between two electron it is called a triple bond. What’s stronger multiple bond or single bond?

Bond strength is measured by the amount of energy required to break the bond. They have a greater attraction towards each other in multiple bonded atoms draws them closer together. So their bonds are much shorter.

Lewis Dot Structure CO 2 Example page 332 - 333
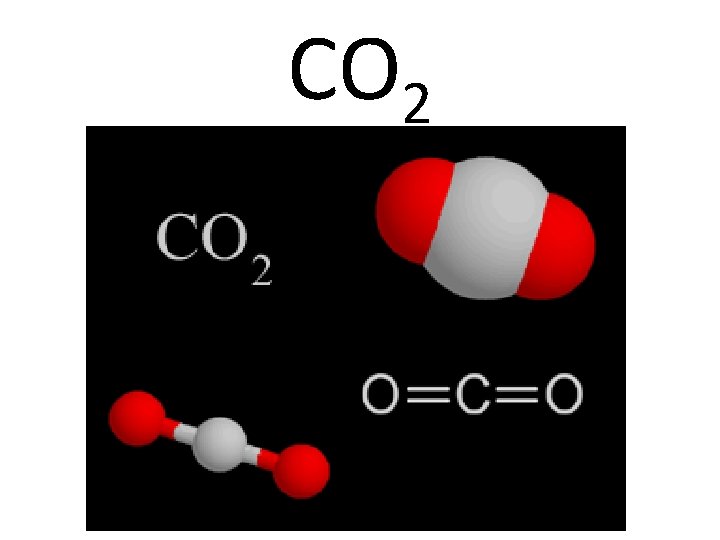
CO 2
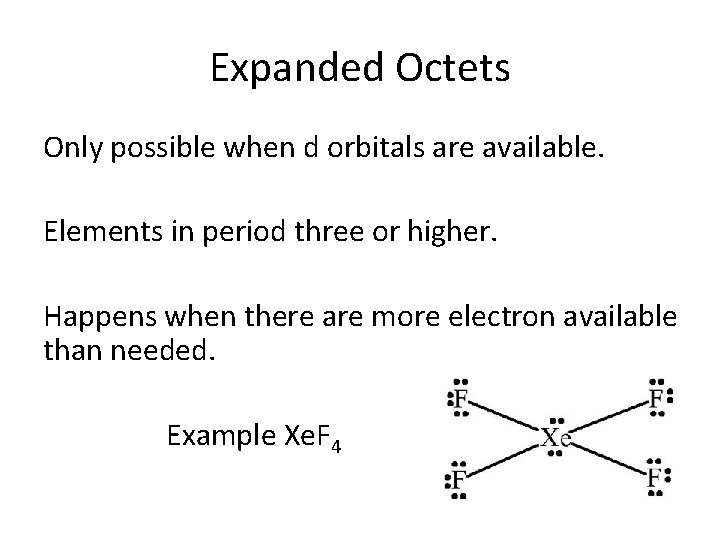
Expanded Octets Only possible when d orbitals are available. Elements in period three or higher. Happens when there are more electron available than needed. Example Xe. F 4

Multiple Bonds Hydrogen Cyanide – HCN

Multiple Bonds Hydrogen Cyanide – HCN Practice Problems page 335 Q. 1 -3
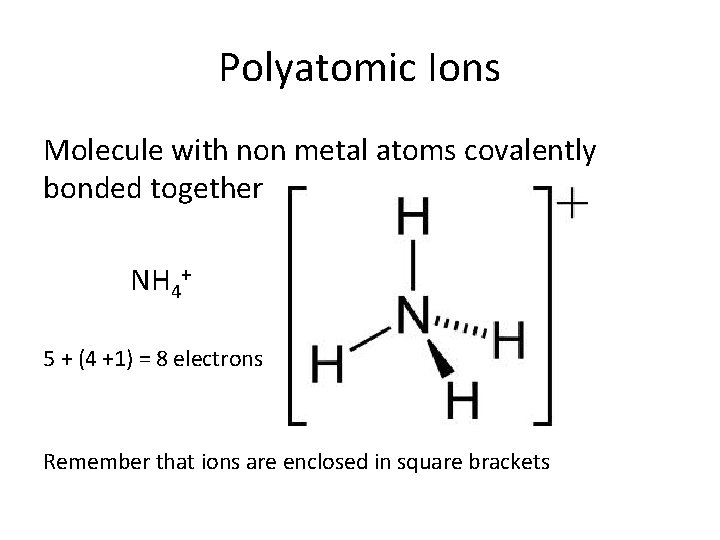
Polyatomic Ions Molecule with non metal atoms covalently bonded together NH 4+ 5 + (4 +1) = 8 electrons Remember that ions are enclosed in square brackets
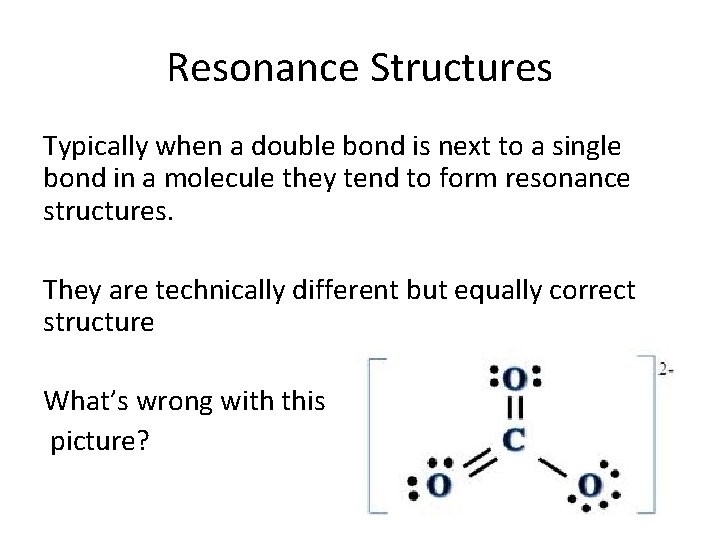
Resonance Structures Typically when a double bond is next to a single bond in a molecule they tend to form resonance structures. They are technically different but equally correct structure What’s wrong with this picture?

Ten electrons around the carbon atom. The double bond can delocalize and change between all three oxygen atoms. The bond strength and distance is the same for all three bonds

More than one central atom Formic acid: HCOOH Take out a piece of and try to draw it out. Make sure to include lone pairs of electrons.
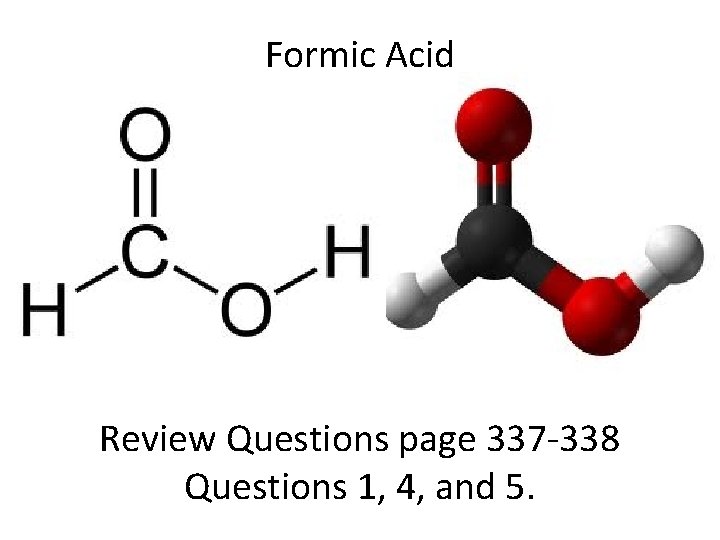
Formic Acid Review Questions page 337 -338 Questions 1, 4, and 5.

Homework: Page 339 -340. Questions 1 -9.
- Slides: 24