Lewis Dot Structures Covalent Bonding Gilbert Newton Lewis

Lewis Dot Structures & Covalent Bonding Gilbert Newton Lewis (surrounded by pairs of electrons)

Lewis-Dot Structures • Find out which group (column) your element is in. • This will tell you the number of valence electrons your element has. • You will only draw the valence electrons.
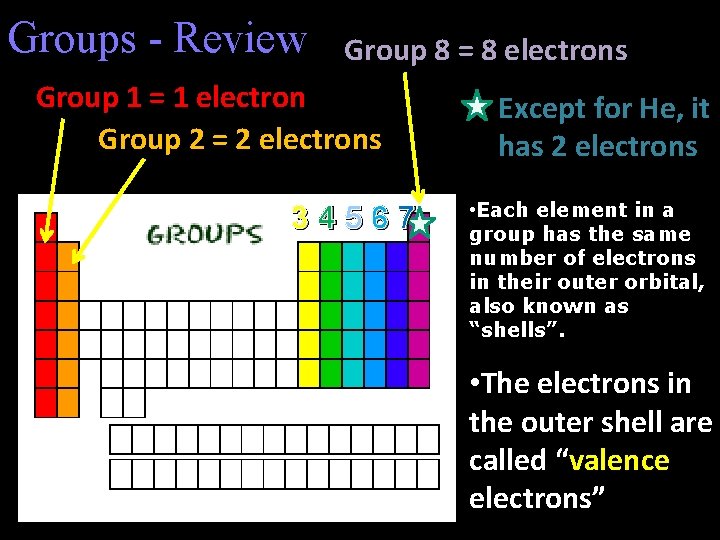
Groups - Review Group 8 = 8 electrons Group 1 = 1 electron Group 2 = 2 electrons 34567 Except for He, it has 2 electrons • Each element in a group has the same number of electrons in their outer orbital, also known as “shells”. • The electrons in the outer shell are called “valence electrons”

Lewis-Dot Structures C 1) Write the element symbol. 2) Carbon is in the 4 th group, so it has 4 valence electrons. 3) Starting at the right, draw 4 electrons, or dots, counterclockwise around the element symbol.

Lewis-Dot Structures H Try these elements on your own: a) H b) P c) Ca d) Ar
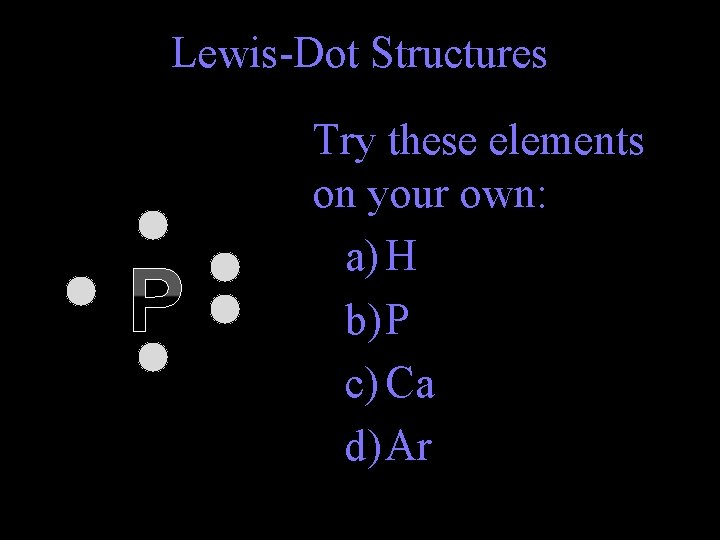
Lewis-Dot Structures P Try these elements on your own: a) H b) P c) Ca d) Ar

Lewis-Dot Structures Ca Try these elements on your own: a) H b) P c) Ca d) Ar

Lewis-Dot Structures Ar Try these elements on your own: a) H b) P c) Ca d) Ar

Lewis-Dot Structures Symbols of atoms with dots to represent the valence-shell electrons 1 2 13 14 15 16 17 H 18 He: Li Be B Na Mg Al C N Si P : O : F : Ne : : S : Cl : Ar :

In an IONIC bond, electrons are lost or gained, resulting in the formation of IONS in ionic compounds. K F
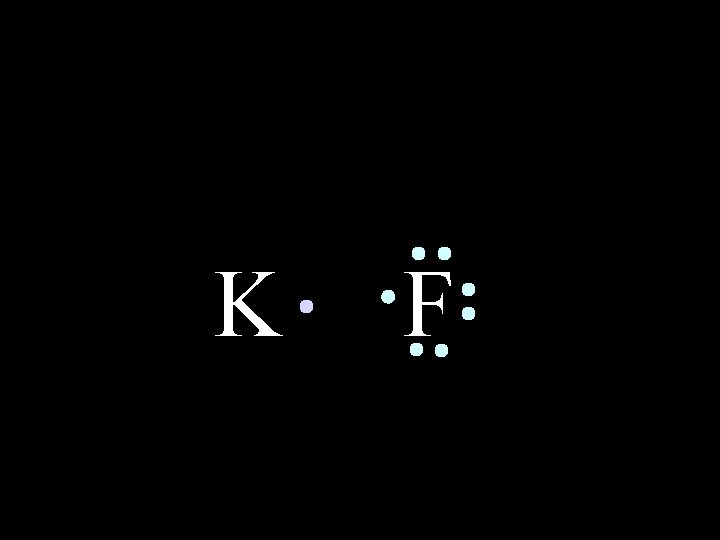
K F

K F
K F
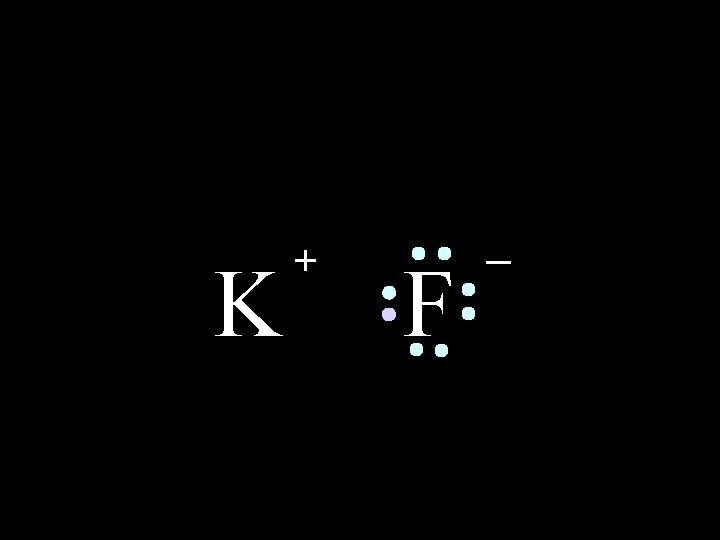

K + F _ The compound potassium fluoride consists of potassium (K+) ions and fluoride (F-) ions

K + F _ The ionic bond is the attraction between the positive K+ ion and the negative F- ion

In covalent bonding, atoms still want to be like a noble gas (the octet rule). But rather than losing or gaining electrons, atoms now share an electron pair.
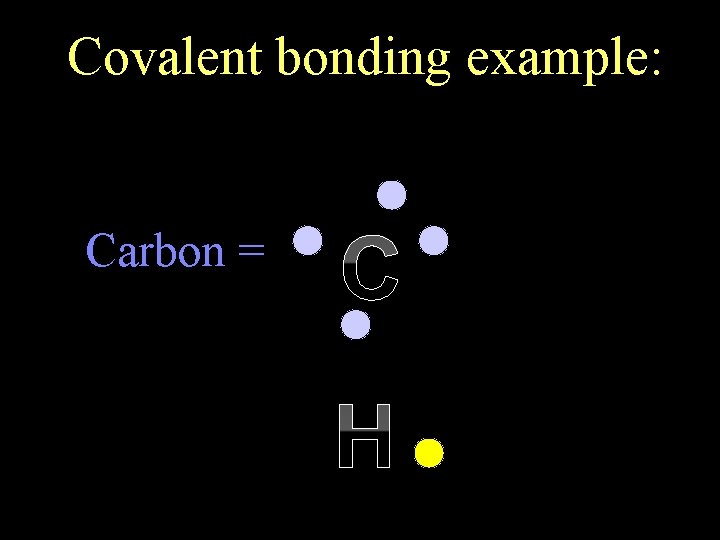
Covalent bonding example: Carbon = C H
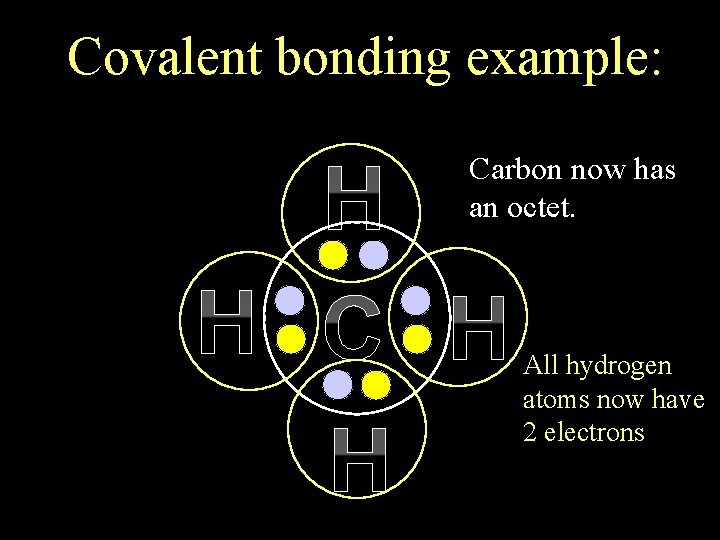
Covalent bonding example: H H Carbon now has an octet. All hydrogen atoms now have 2 electrons
- Slides: 19