Lewis Acids Bases Lewis acid a substance that

Lewis Acids & Bases Lewis acid = a substance that accepts an electron pair Lewis base = a substance that donates an electron pair 1
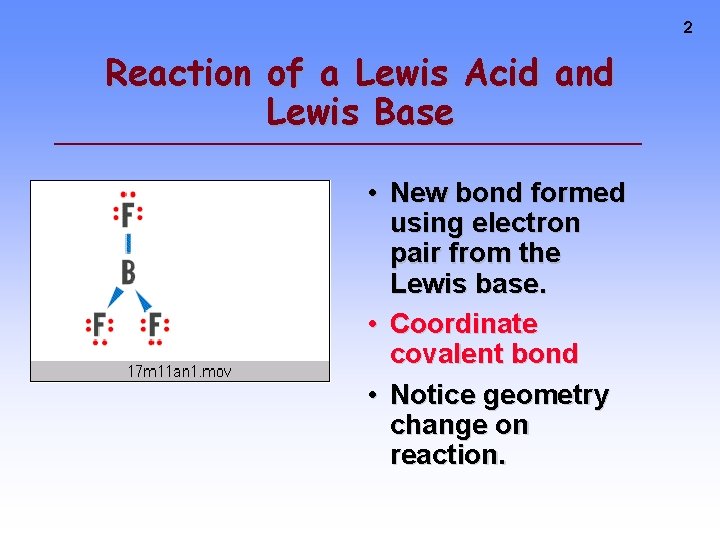
2 Reaction of a Lewis Acid and Lewis Base • New bond formed using electron pair from the Lewis base. • Coordinate covalent bond • Notice geometry change on reaction.
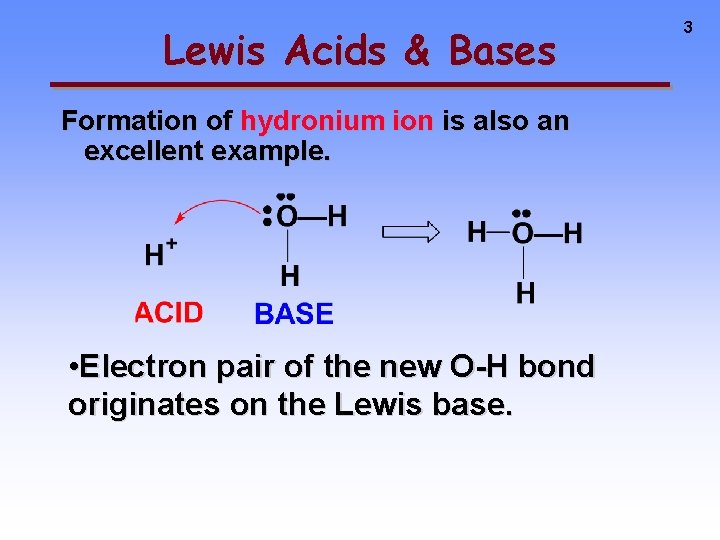
Lewis Acids & Bases Formation of hydronium ion is also an excellent example. • Electron pair of the new O-H bond originates on the Lewis base. 3
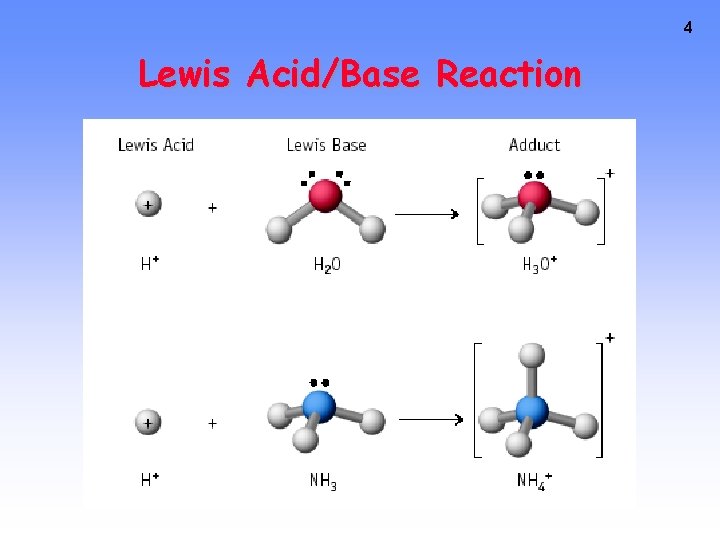
4 Lewis Acid/Base Reaction
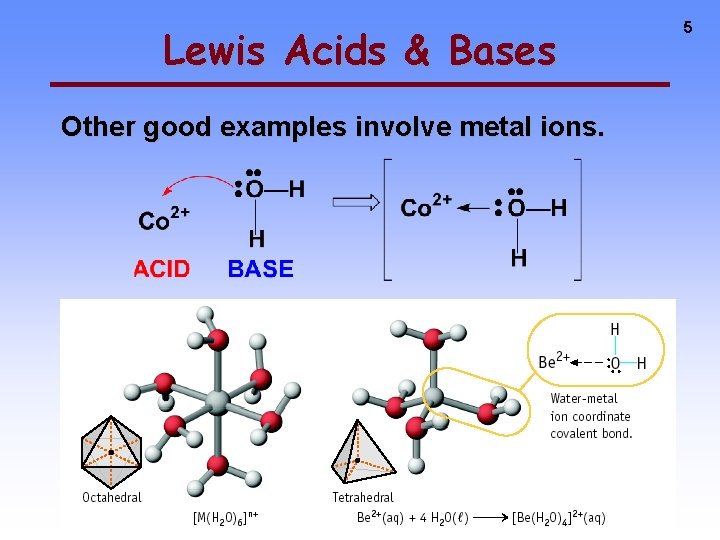
Lewis Acids & Bases Other good examples involve metal ions. 5
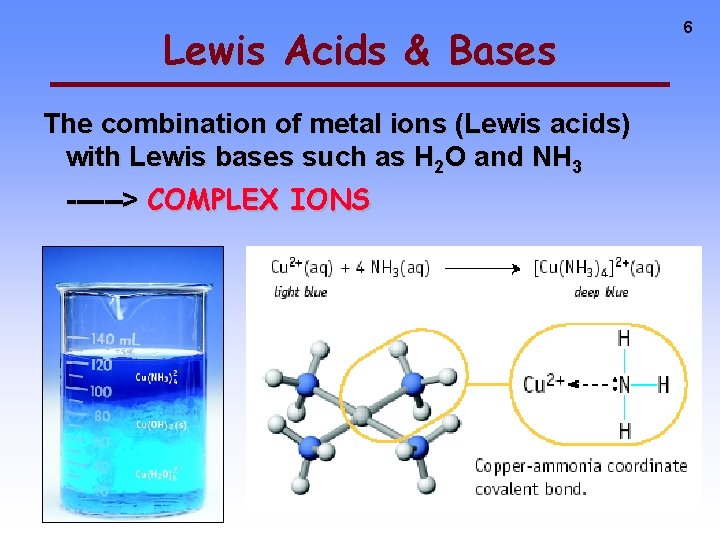
Lewis Acids & Bases The combination of metal ions (Lewis acids) with Lewis bases such as H 2 O and NH 3 ------> COMPLEX IONS 6
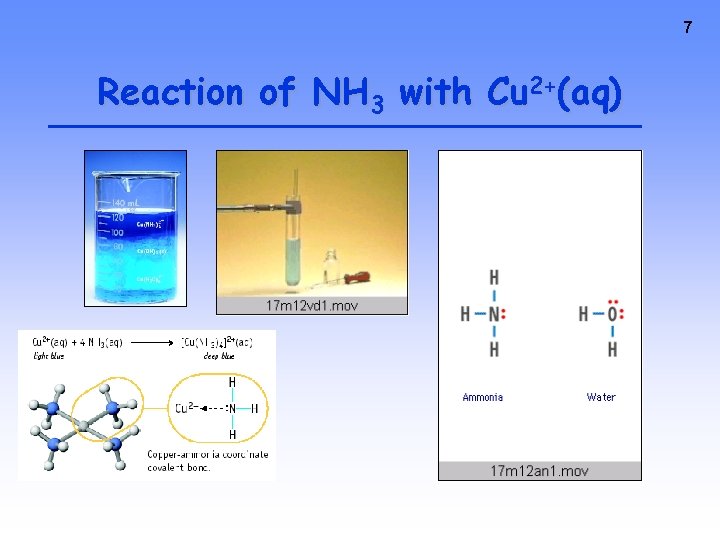
7 Reaction of NH 3 with Cu 2+(aq)
![8 Lewis Acids & Bases [Ni(H 2 O)6]2+ + 6 NH 3 ---> [Ni(NH 8 Lewis Acids & Bases [Ni(H 2 O)6]2+ + 6 NH 3 ---> [Ni(NH](http://slidetodoc.com/presentation_image/13b8614dc0d835f482807a40d315f551/image-8.jpg)
8 Lewis Acids & Bases [Ni(H 2 O)6]2+ + 6 NH 3 ---> [Ni(NH 3)6]2+ + DMG See page 924
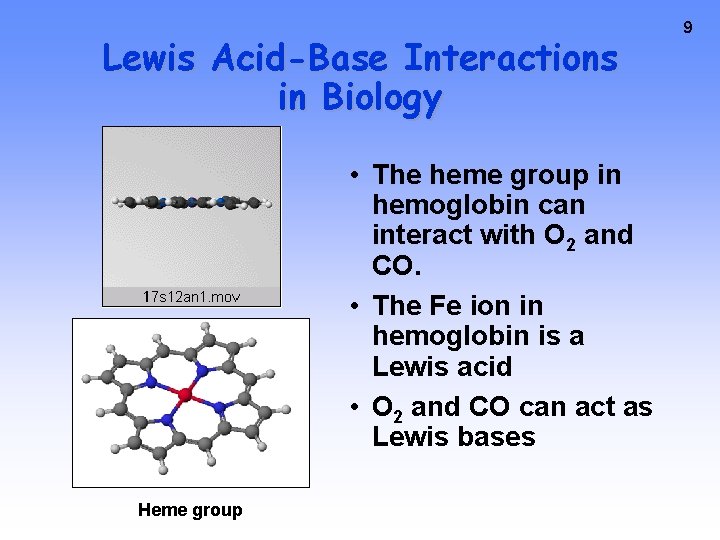
Lewis Acid-Base Interactions in Biology • The heme group in hemoglobin can interact with O 2 and CO. • The Fe ion in hemoglobin is a Lewis acid • O 2 and CO can act as Lewis bases Heme group 9
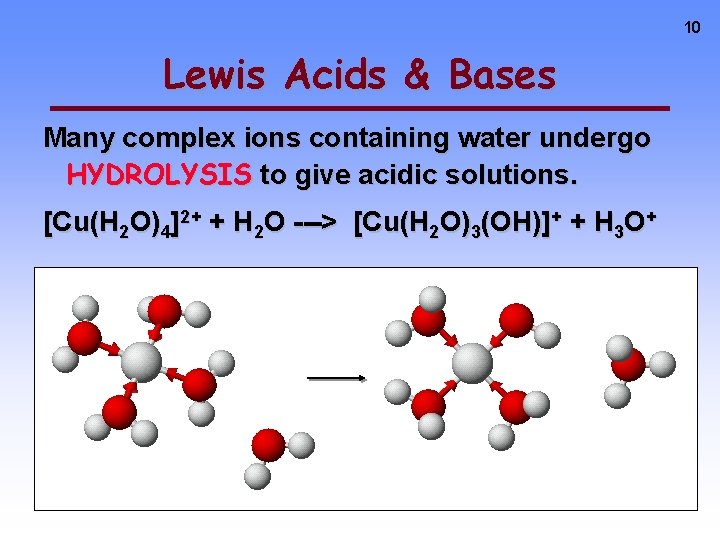
10 Lewis Acids & Bases Many complex ions containing water undergo HYDROLYSIS to give acidic solutions. [Cu(H 2 O)4]2+ + H 2 O ---> [Cu(H 2 O)3(OH)]+ + H 3 O+
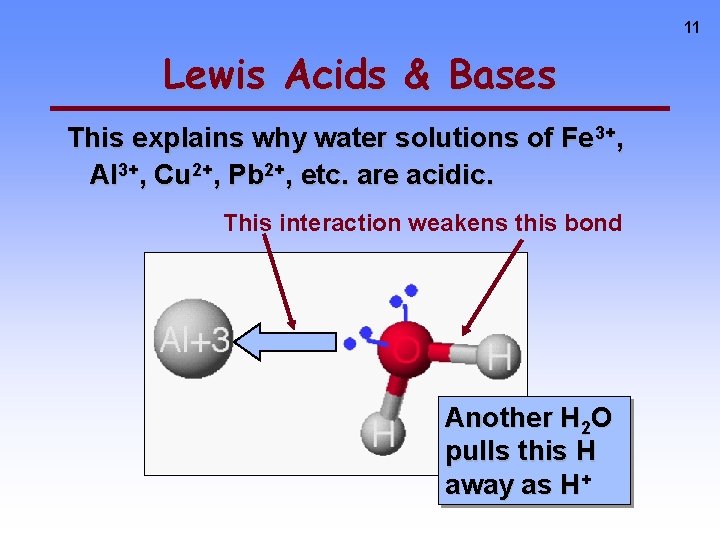
11 Lewis Acids & Bases This explains why water solutions of Fe 3+, Al 3+, Cu 2+, Pb 2+, etc. are acidic. This interaction weakens this bond Another H 2 O pulls this H away as H+

Amphoterism of Al(OH)3 12

Lewis Acids & Bases This explains AMPHOTERIC nature of some metal hydroxides. Al(OH)3(s) + 3 H+ --> Al 3+ + 3 H 2 O Here Al(OH)3 is a Brønsted base. Al(OH)3(s) + OH- --> Al(OH)4 Here Al(OH)3 is a Lewis acid. 13

Lewis Acids & Bases Formation of complex ions explains why you can dissolve a ppt. by forming a complex ion. Ag. Cl(s) e Ag+ + Cl. Ksp = 1. 8 x 10 -10 Ag+ + 2 NH 3 --> Ag(NH 3)2+ Kform = 1. 6 x 107 ------------------Ag. Cl(s) + 2 NH 3 e Ag(NH 3)2+ + Cl. Knet = _________ 14

Why? • Why are some compounds acids? • Why are some compounds bases? • Why do acids and bases vary in strength? • Can we predict variations in acidity or basicity? 15
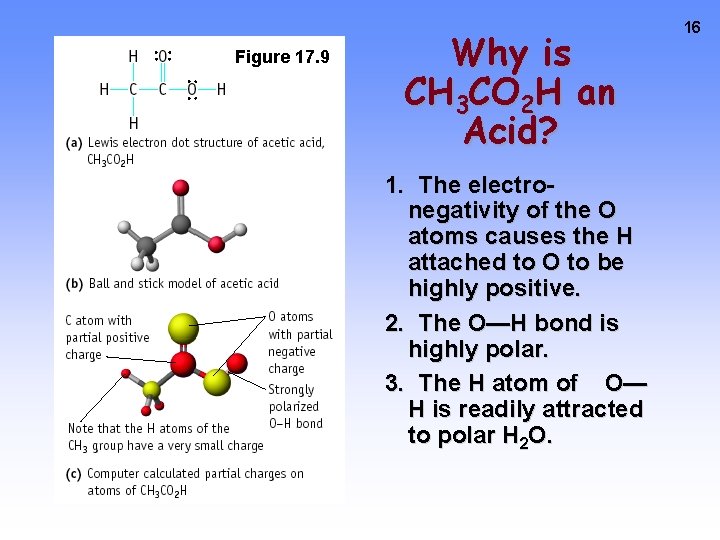
Figure 17. 9 Why is CH 3 CO 2 H an Acid? 1. The electronegativity of the O atoms causes the H attached to O to be highly positive. 2. The O—H bond is highly polar. 3. The H atom of O— H is readily attracted to polar H 2 O. 16

17 Acetic acid Ka = 1. 8 x 10 -5 Trichloroacetic acid Ka = 0. 3 Trichloroacetic acid is much stronger acid owing to the high electronegativity of Cl, which withdraws electrons from the rest of the molecule. This makes the O—H bond highly polar. The H of O—H is very positive.
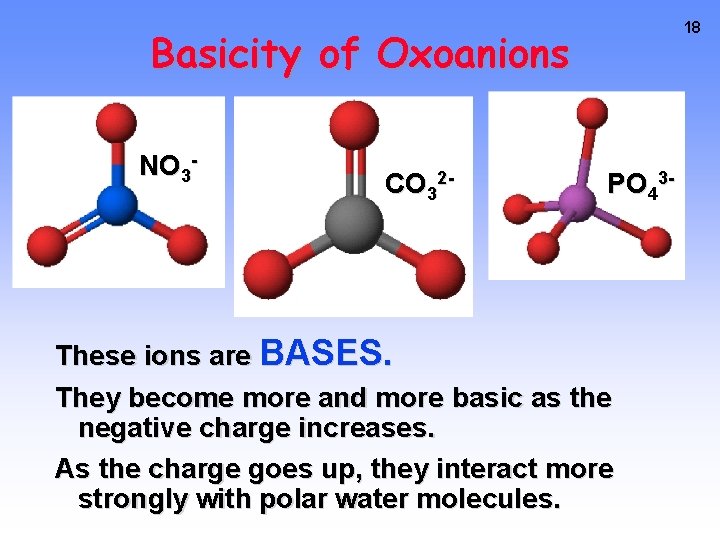
18 Basicity of Oxoanions NO 3 - CO 32 - PO 43 - These ions are BASES. They become more and more basic as the negative charge increases. As the charge goes up, they interact more strongly with polar water molecules.
- Slides: 18