Lewis acidbase theory Lewis acidbase theory Very different

Lewis acid-base theory

Lewis acid-base theory Very different from Brønsted-Lowrey acid-base.

Lewis acid-base theory Very different from Brønsted-Lowrey acid-base. Not dependent on protons or p. H.

Lewis acid-base theory Definitions: An acid is a lone-pair acceptor.

Lewis acid-base theory Definitions: An acid is a lone-pair acceptor. H+ no electrons – by accepting 2 electrons it attains noble gas configuration.

Lewis acid-base theory An acid is a lone-pair acceptor. H+ no electrons – by accepting 2 electrons it attains noble gas configuration. BF 3 electron deficient compound - B needs 2 electrons for noble gas configuration.

B Group III 3 valence electrons

B Group III 3 valence electrons F Group VII 7 valence electrons

B Group III 3 valence electrons F Group VII 7 valence electrons


6 electrons for boron rather than 8 Lewis Acid

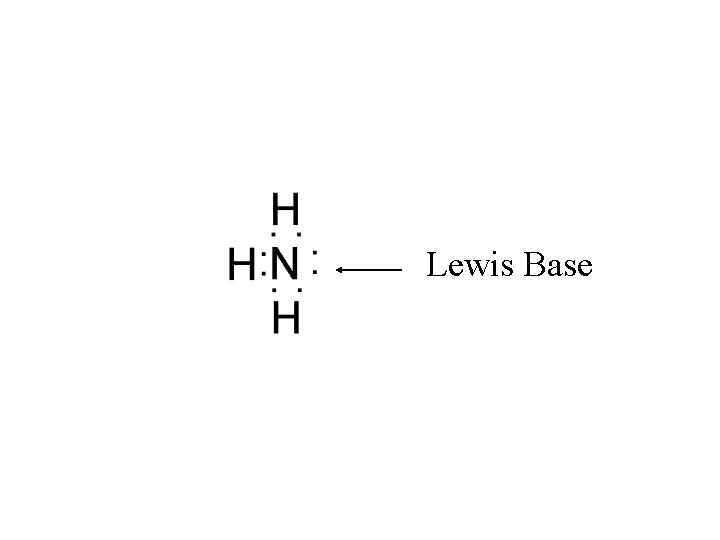
Lewis acid-base theory Definitions: Lewis base: lone-pair donor
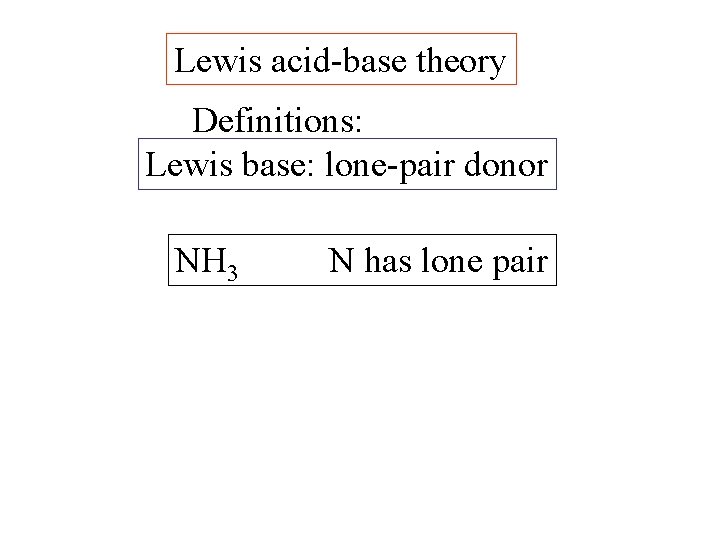
Lewis acid-base theory Definitions: Lewis base: lone-pair donor NH 3 N has lone pair
Group I Group V
Lewis Base
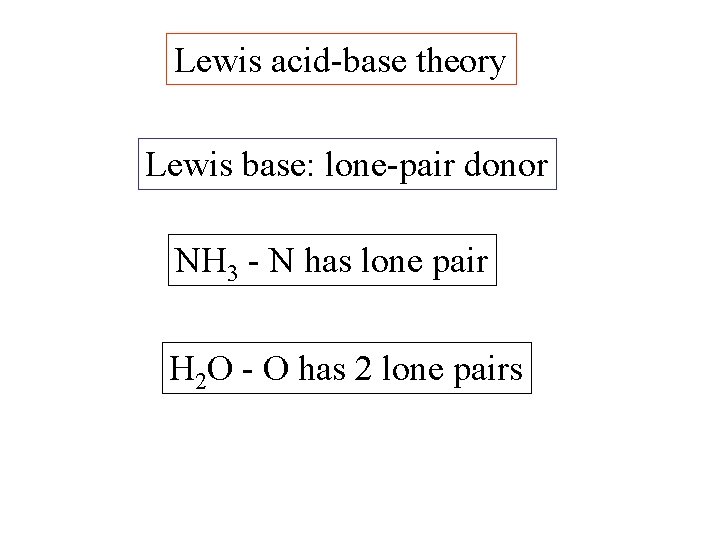
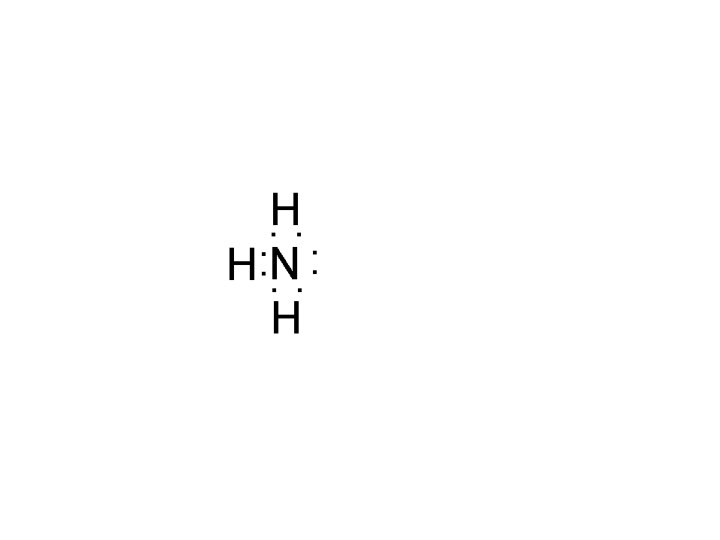
Lewis acid-base theory Lewis base: lone-pair donor NH 3 - N has lone pair H 2 O - O has 2 lone pairs

Lewis acid-base theory H 3 N : + BF 3 H 3 N : BF 3

Lewis acid-base theory H 3 N : + BF 3 Lewis base H 3 N : BF 3
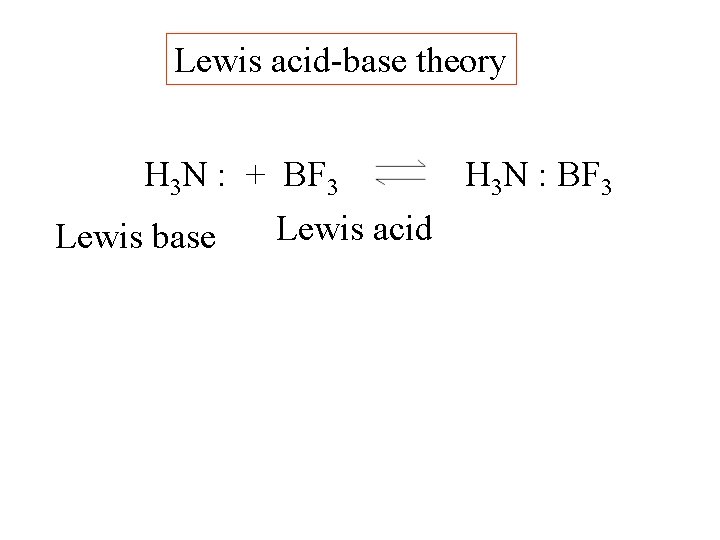
Lewis acid-base theory H 3 N : + BF 3 H 3 N : BF 3 Lewis acid Lewis base

Lewis acid-base theory H 3 N : + BF 3 H 3 N : BF 3 Lewis acid Lewis base Both electrons in this bond come from nitrogen.

Lewis acid-base theory H 3 N : + BF 3 H 3 N : BF 3 Lewis acid Lewis base Coordinate covalent bond
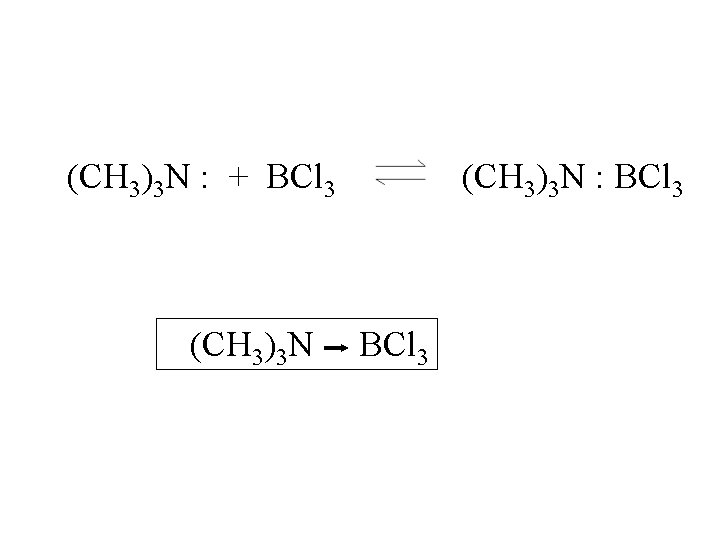
(CH 3)3 N : + BCl 3 (CH 3)3 N : BCl 3
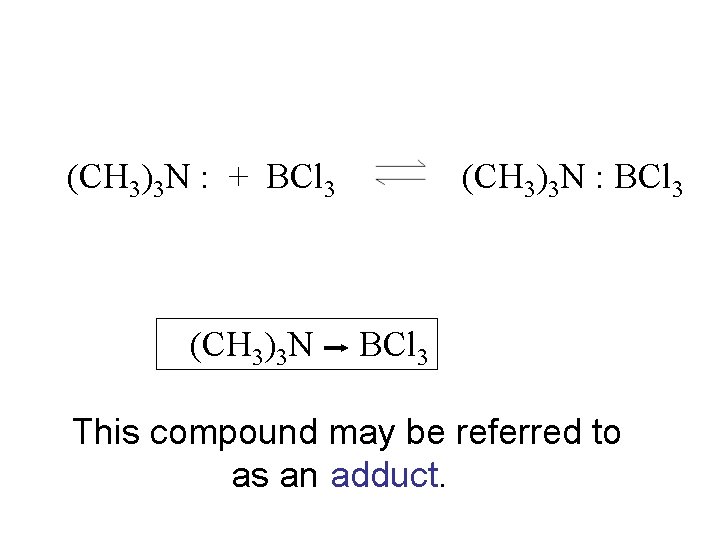
(CH 3)3 N : + BCl 3 (CH 3)3 N : BCl 3 This compound may be referred to as an adduct.
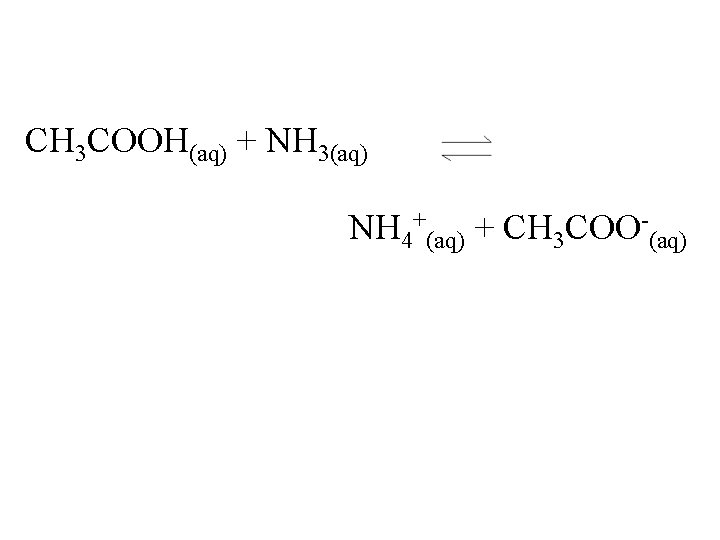
CH 3 COOH(aq) + NH 3(aq) NH 4+(aq) + CH 3 COO-(aq)
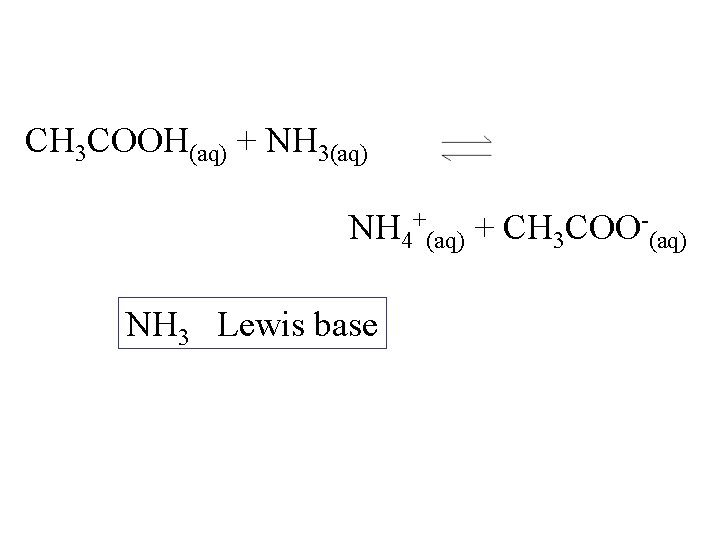
CH 3 COOH(aq) + NH 3(aq) NH 4+(aq) + CH 3 COO-(aq) NH 3 Lewis base
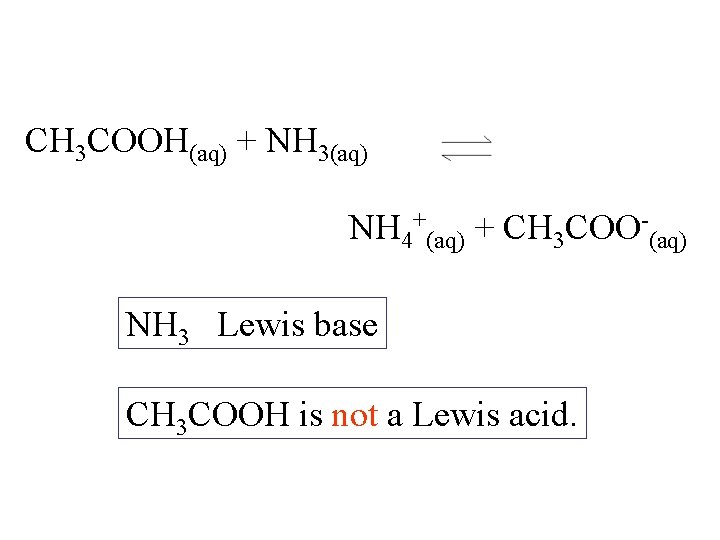
CH 3 COOH(aq) + NH 3(aq) NH 4+(aq) + CH 3 COO-(aq) NH 3 Lewis base CH 3 COOH is not a Lewis acid.
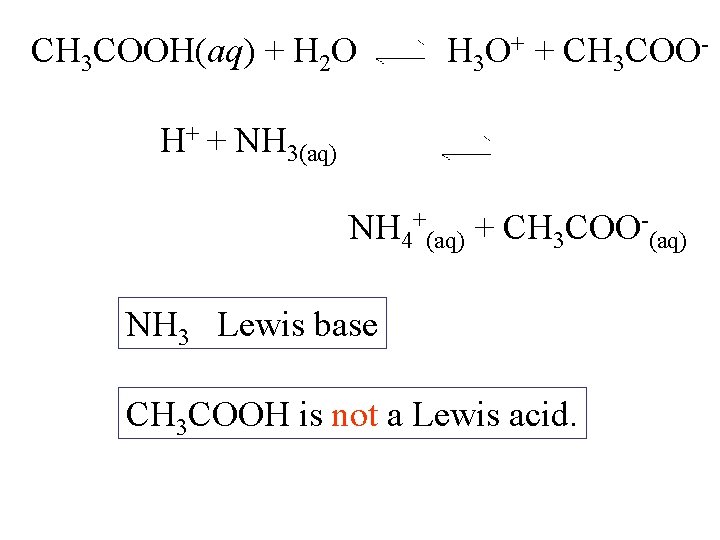
CH 3 COOH(aq) + H 2 O H 3 O+ + CH 3 COO- H+ + NH 3(aq) NH 4+(aq) + CH 3 COO-(aq) NH 3 Lewis base CH 3 COOH is not a Lewis acid.

CH 3 COOH(aq) + H 2 O H 3 O+ + CH 3 COO- H+ + NH 3(aq) NH 4+(aq) + CH 3 COO-(aq) NH 3 Lewis base CH 3 COOH is not a Lewis acid. H 3 O + H + + H 2 O

CH 3 COOH(aq) + NH 3(aq) NH 4+(aq) + CH 3 COO-(aq) NH 3 Lewis base CH 3 COOH is not a Lewis acid. H+ is the Lewis acid.

Al(OC 2 H 5)3

Al(OC 2 H 5)3 Tri-ethoxy aluminum

Al(OC 2 H 5)3 Al is Group III - electron deficient like BF 3.

Al(OC 2 H 5)3 Al is Group III - electron deficient like boron.

Al(OC 2 H 5)3 Al is Group III - electron deficient like boron. This molecule is a Lewis acid.

Al(OC 2 H 5)3 Al is Group III - electron deficient like boron. This molecule is a Lewis acid. O has lone pairs, it is a Lewis base

Al(OC 2 H 5)3 Al is Group III - electron deficient like boron. This molecule is a Lewis acid. O has lone pairs, it is a Lewis base The molecule is both a Lewis acid and base.
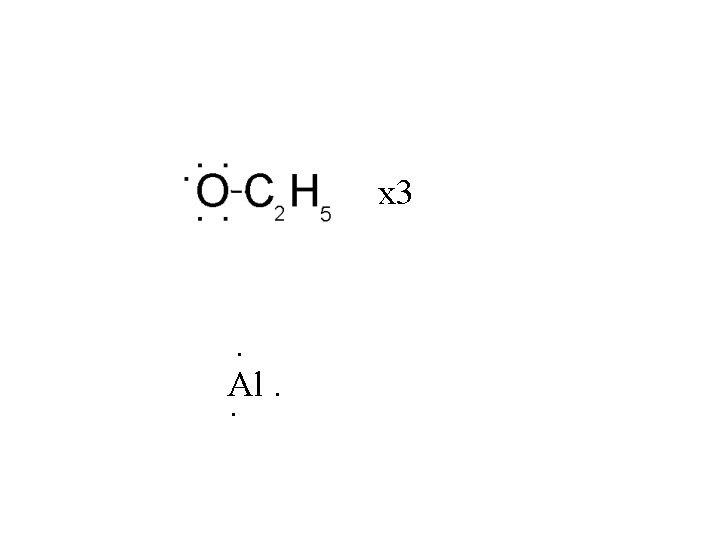
x 3 . Al. .

Al(OC 2 H 5)3

Al(OC 2 H 5)3 Covalent bonds

Al(OC 2 H 5)3 Electron deficient

Al(OC 2 H 5)3 Empirical formula
![Al(OC 2 H 5)3 Empirical formula [Al(OC 2 H 5)3]2 Molecular formula Al(OC 2 H 5)3 Empirical formula [Al(OC 2 H 5)3]2 Molecular formula](http://slidetodoc.com/presentation_image/2ab492749fa17083ab909bb4ac9f4d15/image-43.jpg)
Al(OC 2 H 5)3 Empirical formula [Al(OC 2 H 5)3]2 Molecular formula
![[Al(OC 2 H 5)3]2 dimer [Al(OC 2 H 5)3]2 dimer](http://slidetodoc.com/presentation_image/2ab492749fa17083ab909bb4ac9f4d15/image-44.jpg)
[Al(OC 2 H 5)3]2 dimer
![[Al(OC 2 H 5)3]2 Covalent bonds dimer [Al(OC 2 H 5)3]2 Covalent bonds dimer](http://slidetodoc.com/presentation_image/2ab492749fa17083ab909bb4ac9f4d15/image-45.jpg)
[Al(OC 2 H 5)3]2 Covalent bonds dimer
![[Al(OC 2 H 5)3]2 Coordinate covalent Covalent bonds dimer [Al(OC 2 H 5)3]2 Coordinate covalent Covalent bonds dimer](http://slidetodoc.com/presentation_image/2ab492749fa17083ab909bb4ac9f4d15/image-46.jpg)
[Al(OC 2 H 5)3]2 Coordinate covalent Covalent bonds dimer
![[Al(OC 2 H 5)3]2 dimer [Al(OC 2 H 5)3]2 dimer](http://slidetodoc.com/presentation_image/2ab492749fa17083ab909bb4ac9f4d15/image-47.jpg)
[Al(OC 2 H 5)3]2 dimer

Al. Cl 3

Al. Cl 3 Empirical formula
![Al. Cl 3 Empirical formula [Al. Cl 3]2 Molecular formula Al. Cl 3 Empirical formula [Al. Cl 3]2 Molecular formula](http://slidetodoc.com/presentation_image/2ab492749fa17083ab909bb4ac9f4d15/image-50.jpg)
Al. Cl 3 Empirical formula [Al. Cl 3]2 Molecular formula
![[Al. Cl 3]2 [Al. Cl 3]2](http://slidetodoc.com/presentation_image/2ab492749fa17083ab909bb4ac9f4d15/image-51.jpg)
[Al. Cl 3]2


s block
s block p block
s block p block Main Group Elements

Oxides of s and p block elements are acid and base anhydrides, with definite trends in respect to the elements’ location in the periodic table.
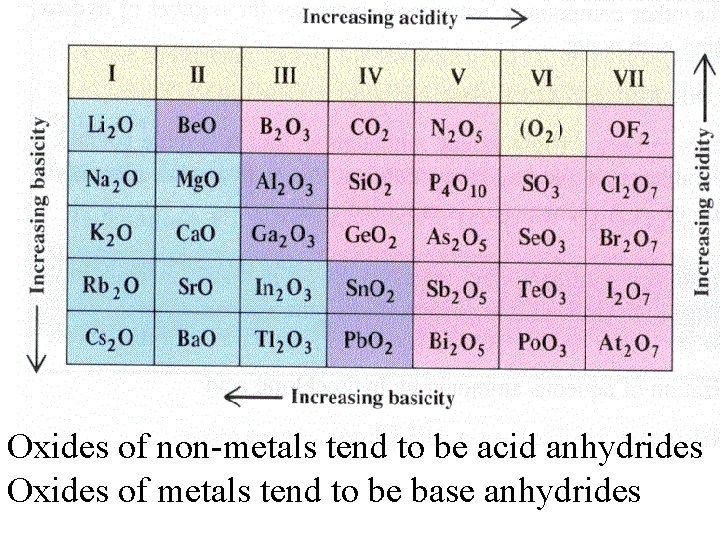
Oxides of non-metals tend to be acid anhydrides Oxides of metals tend to be base anhydrides
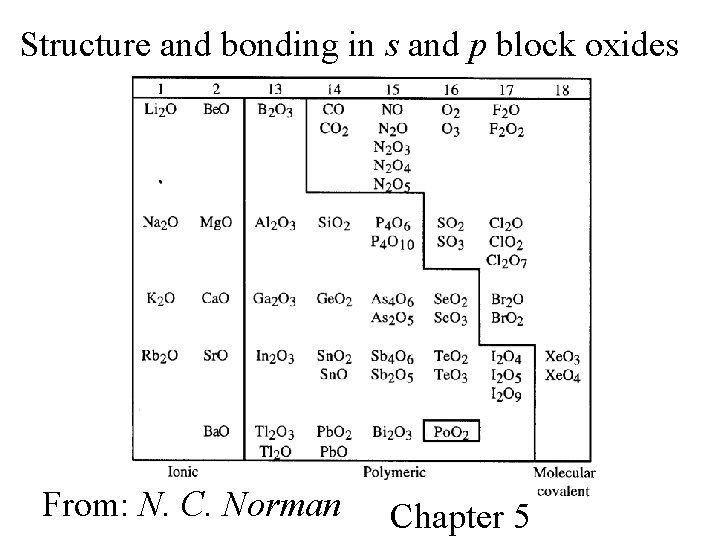
Structure and bonding in s and p block oxides From: N. C. Norman Chapter 5

Norman: Chapter 6 should be read for acid and base information.

Structure and bonding in s and p block oxides Group numbers are changed in Norman. From: N. C. Norman
Groups 1 - 18 13 14 15 16 17 18 3 4 5 6 7 8 9 10 11 12
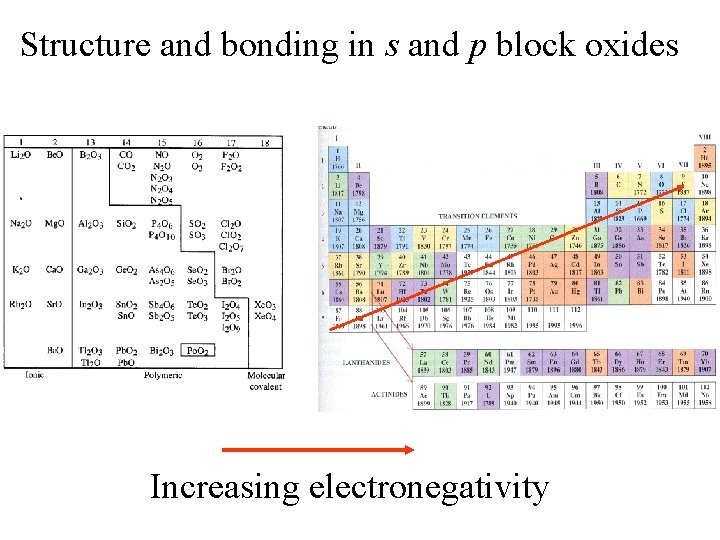
Structure and bonding in s and p block oxides Increasing electronegativity

Binary ionic compounds form from elements having very different electronegativities.

Binary ionic compounds form from elements having very different electronegativities.
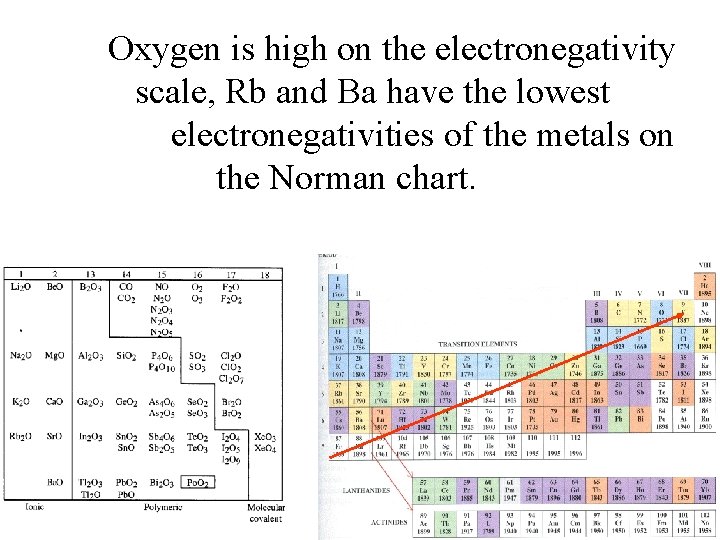
Oxygen is high on the electronegativity scale, Rb and Ba have the lowest electronegativities of the metals on the Norman chart.

K 2 O : ionic material

K 2 O : ionic material K 2 O(s) + H 2 O(l) 2 K+(aq) + O 2 -(aq)
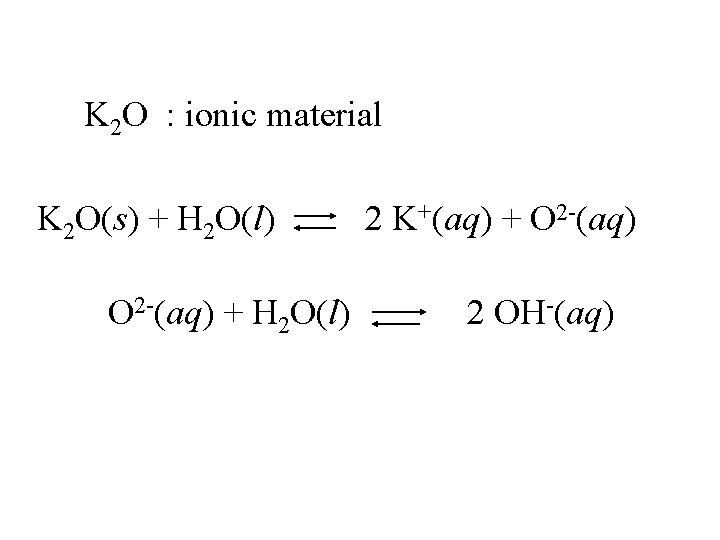
K 2 O : ionic material K 2 O(s) + H 2 O(l) O 2 -(aq) + H 2 O(l) 2 K+(aq) + O 2 -(aq) 2 OH-(aq)
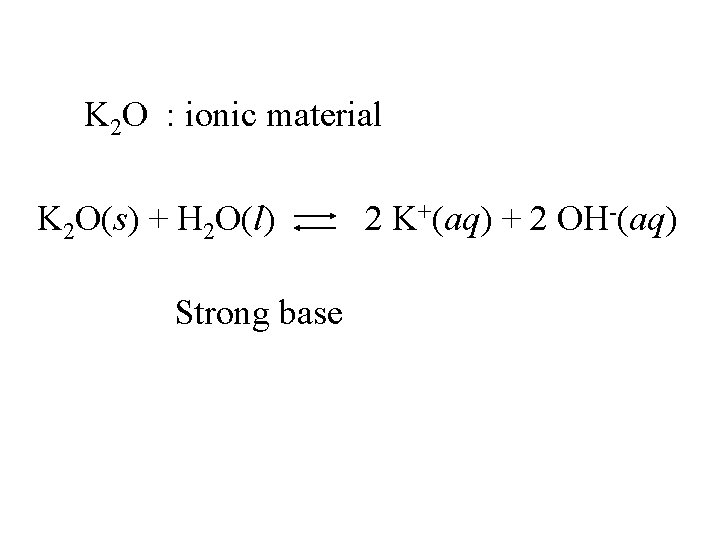
K 2 O : ionic material K 2 O(s) + H 2 O(l) Strong base 2 K+(aq) + 2 OH-(aq)

K 2 O : ionic material K 2 O(s) + H 2 O(l) Strong base Base anhydride 2 K+(aq) + 2 OH-(aq)


Small differences in electronegativities lead to covalent bonds.

Small differences in electronegativities lead to covalent bonds.

SO 3 : covalent molecule

SO 3 : covalent molecule SO 3(g) + H 2 O(l) Strong acid H 2 SO 4(aq)
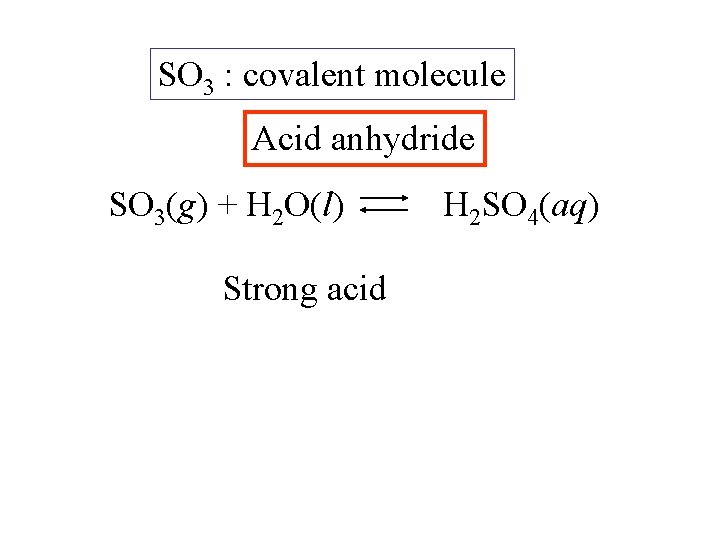
SO 3 : covalent molecule Acid anhydride SO 3(g) + H 2 O(l) Strong acid H 2 SO 4(aq)

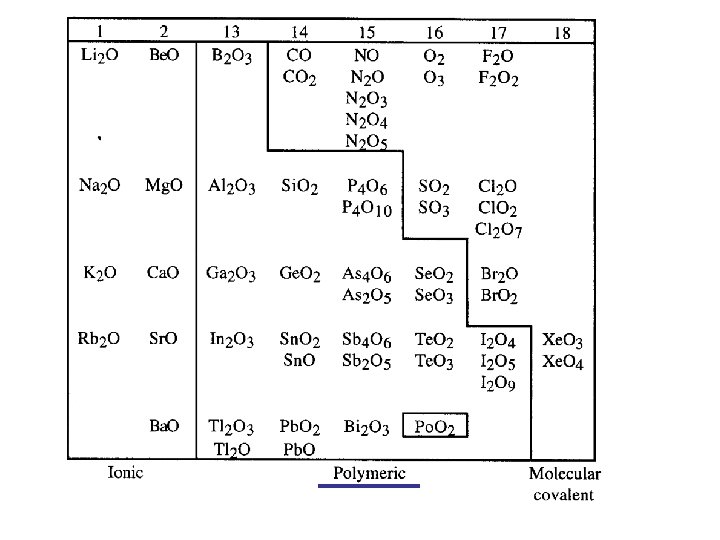

Some of the polymeric oxides will be amphoteric.

Some of the polymeric oxides will be amphoteric. They will act as acids or bases depending on how acidic or basic the environment is.

Some of the polymeric oxides will be amphoteric. Al 2 O 3(aq) + 6 H+(aq) 2 Al 3+(aq) + 3 H 2 O(l)

Some of the polymeric oxides will be amphoteric. Al 2 O 3(aq) + 6 H+(aq) 2 Al 3+(aq) + 3 H 2 O(l) Al 2 O 3 reduces H+ = basic

Some of the polymeric oxides will be amphoteric. Al 2 O 3(aq) + 6 H+(aq) 2 Al 3+(aq) + 3 H 2 O(l) Al 2 O 3 reduces H+ = basic Al 2 O 3(aq) + 2 OH-(aq) + 3 H 2 O(l) 2 [Al(OH)4]-(aq)

Some of the polymeric oxides will be amphoteric. Al 2 O 3(aq) + 2 OH-(aq) + 3 H 2 O(l) 2 [Al(OH)4]-(aq) Al 2 O 3 reduces OH- : acidic

Structures of polymeric oxides

quartz Si. O 4 Empirical formula

quartz Si. O 4 Empirical formula
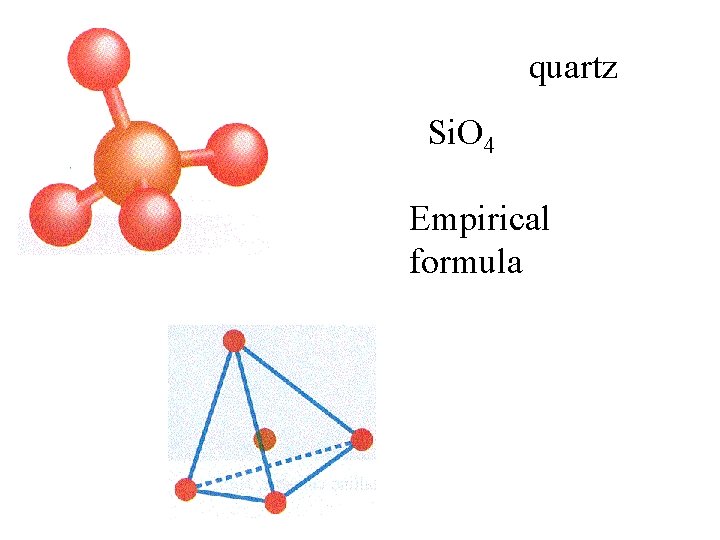
quartz Si. O 4 Empirical formula
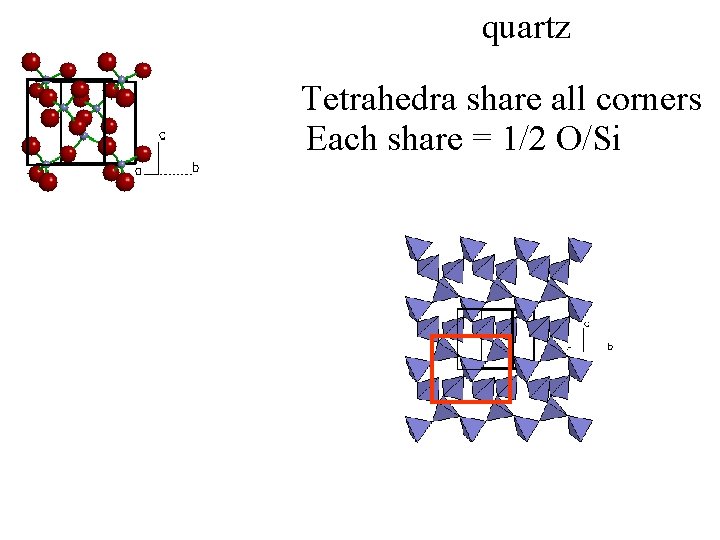
quartz Tetrahedra share all corners Each share = 1/2 O/Si
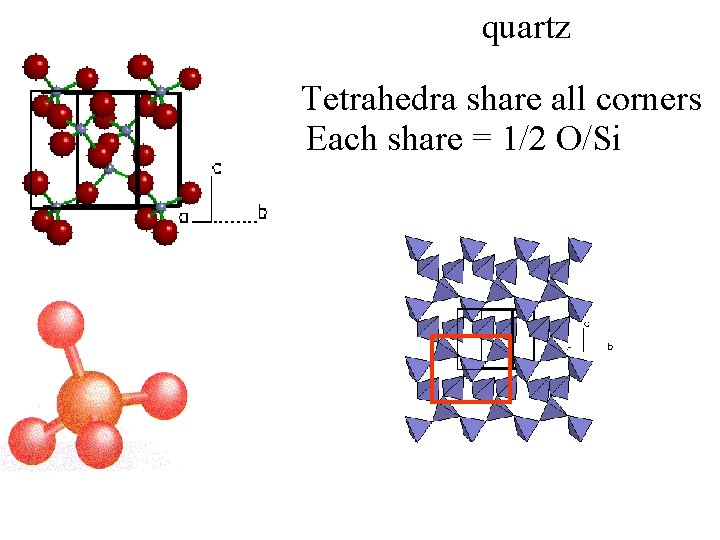
quartz Tetrahedra share all corners Each share = 1/2 O/Si
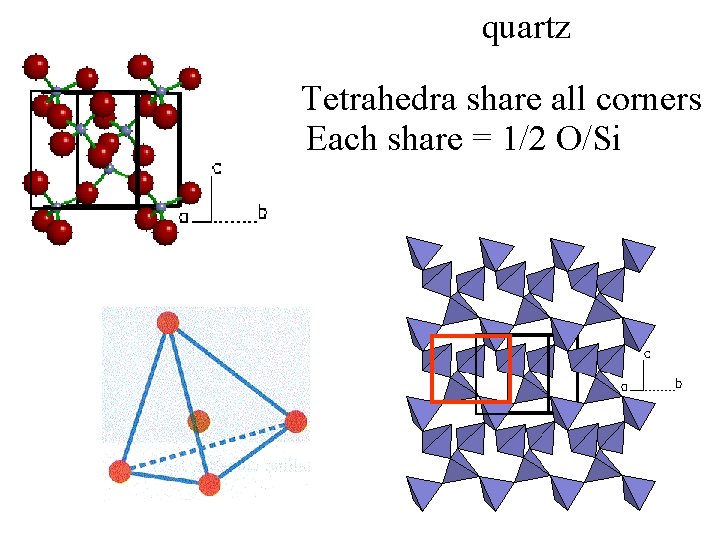
quartz Tetrahedra share all corners Each share = 1/2 O/Si

Binary compounds with halogens

chlorides
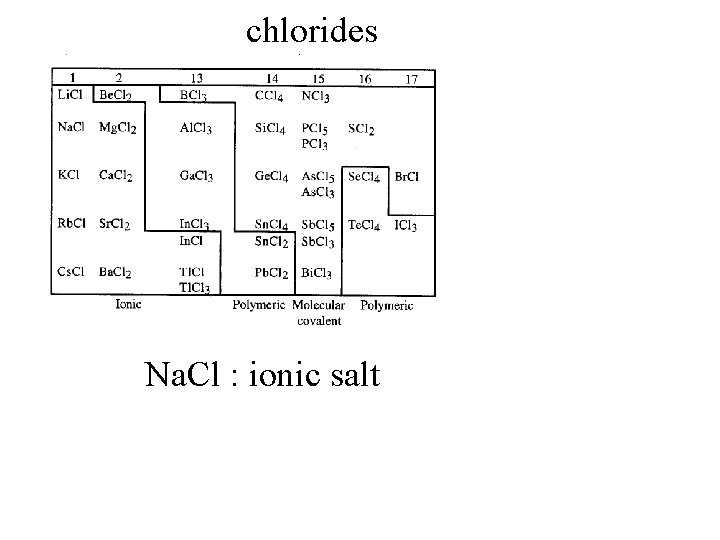
chlorides Na. Cl : ionic salt
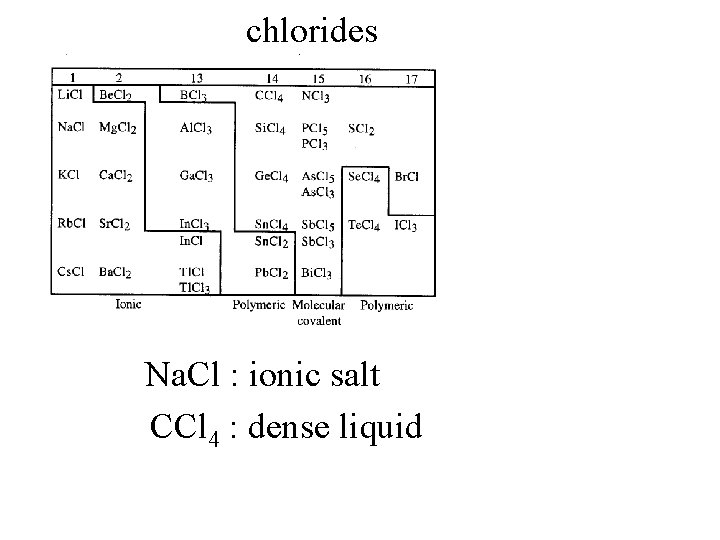
chlorides Na. Cl : ionic salt CCl 4 : dense liquid
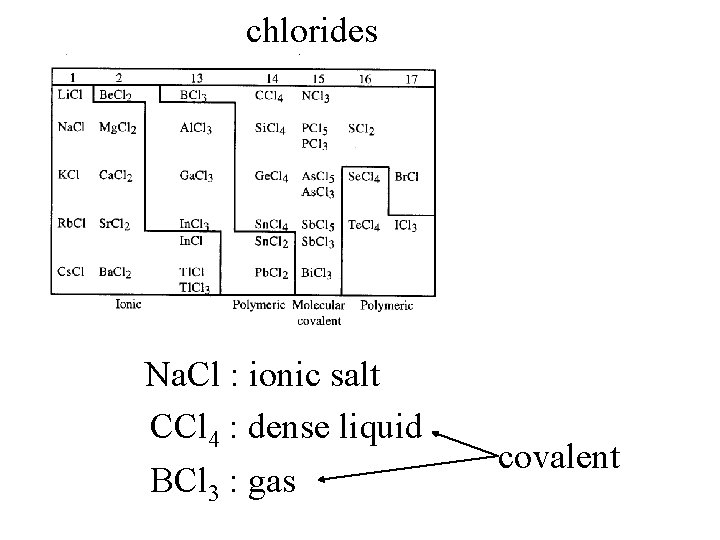
chlorides Na. Cl : ionic salt CCl 4 : dense liquid BCl 3 : gas covalent
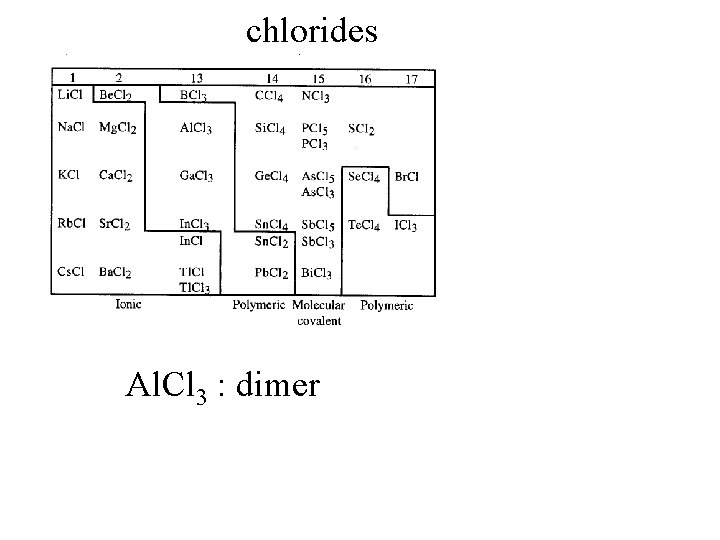
chlorides Al. Cl 3 : dimer

chlorides Al. Cl 3 : dimer Ga. Cl 3 : dimer
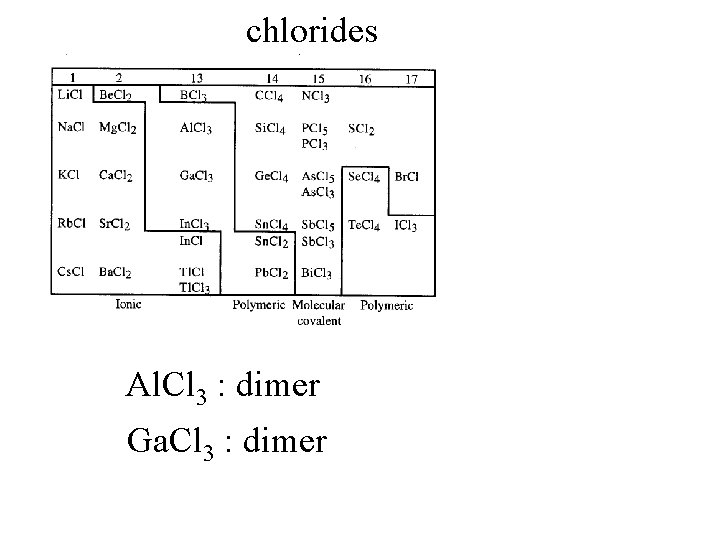
chlorides Al. Cl 3 : dimer Ga. Cl 3 : dimer
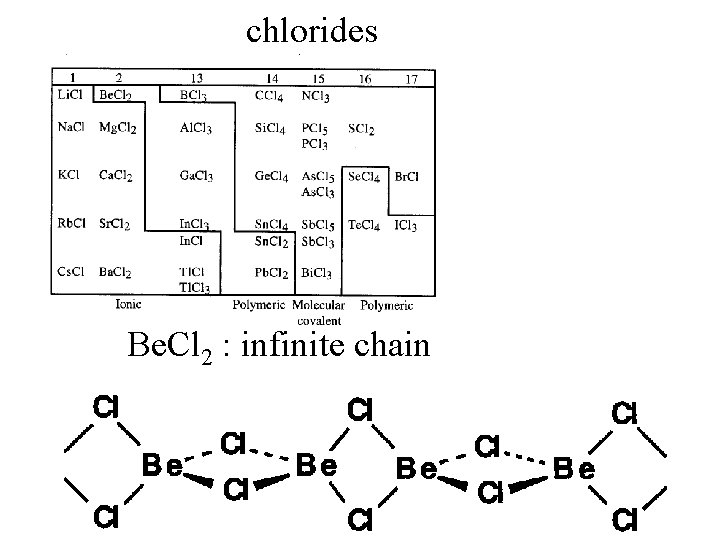
chlorides Be. Cl 2 : infinite chain
- Slides: 100