LEUKEMIAS By Dr Aamer Aleem Consultant Hematologist Associate

LEUKEMIAS By Dr Aamer Aleem Consultant Hematologist Associate Prof. of Medicine College of Medicine & KKUH Riyadh, Saudi Arabia

Leukemias are a group of cancers of the blood or bone marrow and are characterized by an abnormal proliferation (production by multiplication) of blood cells, usually white blood cells (leukocytes). Leukemia is a broad term covering a spectrum of diseases. Any of various acute or chronic neoplastic diseases of the bone marrow in which unrestrained proliferation of white blood cells occurs and which is usually accompanied by anemia and thrombocytopenia

Classification of leukemias Two major types (4 subtypes) of leukemias Acute lymphoblastic leukemia (ALL) Acute myelogenous leukemia (AML) (also "myeloid" or "nonlymphocytic") Chronic leukemias Chronic lymphocytic leukemia (CLL) Chronic myeloid leukemia (CML) (Within these main categories, there are typically several subcategories)

Myeloid vs Lymphoid • Any disease that arises from the myeloid elements (white cell, red cell, platelets) is a myeloid disease …. . AML, CML • Any disease that arises from the lymphoid elements is a lymphoid disease …. . ALL, CLL

Acute vs. chronic leukemia • Acute leukemias: • Young, immature, blast cells in the bone marrow (and often blood) • More fulminant presentation • More aggressive course • Chronic leukemias: • Accumulation of mature, differentiated cells • Often subclinical or incidental presentation • In general, more indolent (slow) course • Frequently splenomegaly • Mature appearing cells in the B. marrow and blood

Acute vs. chronic leukemia • Leukemias are classified according to cell of origin: • Lymphoid cells ALL - lymphoblasts CLL – mature appearing lymphocytes • Myeloid cells AML – myeloblasts CML – mature appearing neutrophils • On a CBC, if you see: • Predominance of blasts in blood consider an acute leukemia • Leukocytosis with mature lymphocytosis consider CLL • Leukocytosis with mature neutrophilia consider CML

Acute leukemias Definition: Types: Malignancies of immature hematopeotic cells. (> 20% blast cells in the bone marrow) Acute Myeloid Leukaemia (AML) Acute Lymphoblastic leukemia Groups: Childhood (< 15) Adult (> 15) Elderly (> 60 years) (ALL) > 80% ALL > 80% AML


Etiology Acute leukemias • Drugs & chemicals • Alkylating agents (Chlorambucil, N mustard, Melphalan) Topoisomerase inhibitors (Etoposide) • Benzene • Ionizing radiation • Viruses HTLV-1 (Adult T-cell leukemia Lymphoma) • Genetic disorders Down’s syndrome • Myelodysplastic syndrome

Clinical presentation Symptoms • • • Usual 1 -3 Month History : MDS – 1 yr (Features of BM failure) Fatigue, malaise, dyspnea (anemia) Bleeding eg after dental procedure Easy bruisability Severe epistaxis Fever (infections) Bone Pain

Clinical Presentation Signs Ø Pallor Ø Hemorrhage from the gums, epistaxis, skin, fundus, GI tract, urinary tract Ø Hepato-splenomegaly Ø Enlarged lymph nodes Ø Gum (hypertrophy) or skin infiltration (M 5) Ø Fever (sepsis, pneumonia, peri-rectal abscess)

Differential Diagnosis 1. Aplastic anemia 2. Myelodysplastic syndromes 3. Multiple myeloma 4. Lymphomas 5. Severe megaloblastic anemia 6. Leukemoid reaction

Laboratory Tests 1. CBC a. Anemia b. Trombocytopenia c. WBC High Normal Low 2. Coagulation Studies (M 3 -DIC) 3. Biochemical Studies (U/E, LFT) Cont. .
4. Peripheral Blood smear – blasts in almost all cases 5. Bone Marrow Examination (>20% blasts) 6. Flow cyometry (Surface immunophenotype of blast cells) 4. Cytogenetics (chromosomal analysis) 5. CSF analysis (all ALL patients, some AML) 6. HLA typing (for younger high risk patients)

Diagnostic methods of importance • Bone marrow aspirate & Romanowsky stain (morphology) Enumeration of blasts, maturing cells, recognition of dysplasia • Cytochemistry Myeloperoxidase, Sudan Black B, esterases to determine involved lineages • Immunophenotyping Defines blast cell lineage commitment as myeloid, lymphoid or biphenotypic • Cytogenetics & molecular studies (FISH, PCR) Detects clonal chromosomal abnormalities, including those of prognostic importance
Blood Film-Normal
Blood Film-Normal
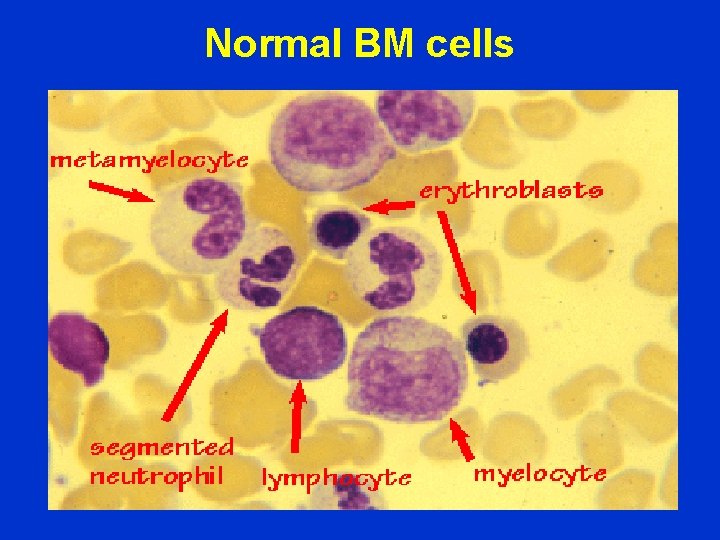
Normal BM cells
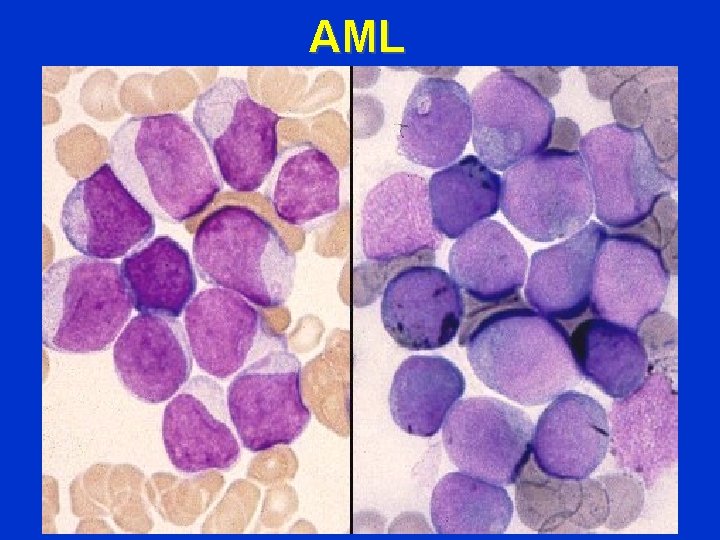
AML
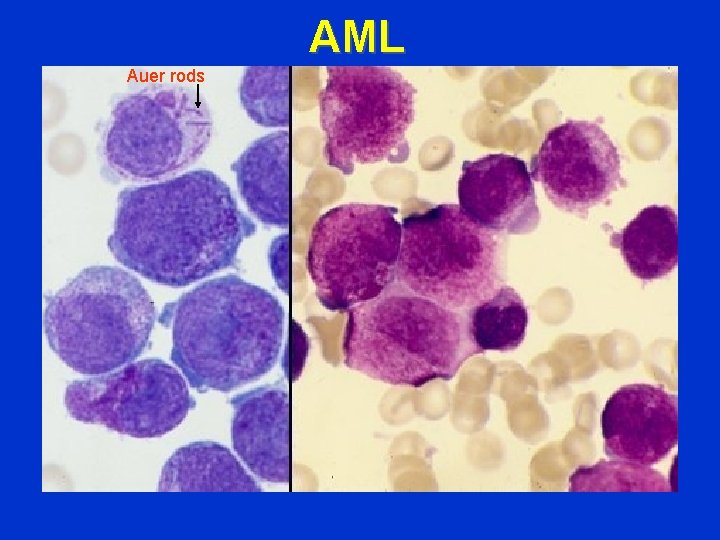
AML Auer rods
Image ID: 0147 -094 Copyrigh t 2001 Carden Jennings Publishing Co. , Ltd. All rights reserved. The material available at this site is for educational purposes only and is NOT intended for any diagnostic, clinically related, or other purpose. Carden Jennings Publishing Co. , Ltd. , assumes no responsibili ty for any use or misuse of this material and makes no warranty or representat ion of any kind with respect to the material available at this site.



Acute Myeloid Leukaemia (AML) Prognostic factors in AML • Age Above the age of 50 years the complete remission rate falls progressively • Cytogenetics Three risk groups defined – Good risk: patients with t(8; 21), t(15; 17) and inv/t(16) – Intermediate risk: Normal, +8, +21, +22, 7 q-, 9 q-, abnormal 11 q 23, all other – Poor risk: patients with -7, -5, 5 q-, abnormal 3 q and complex karyotypes 1. 2. Wheatley K, Burnett AK, Goldstone AH et al. Br J Haem 1999; 107: 69 -79 Grimwade D, Walker H, Oliver F et al. Blood 1998; 92: 2322 -33

Acute Myeloid Leukaemia (AML) Prognostic factors in AML • Treatment response – Patients with >20% blasts in the marrow after first course of treatment have short remissions (if achieved) and poor overall survival • Secondary AML – Patients with AML following chemotherapy or myelodysplasia respond poorly • Trilineage myelodysplasia – Patients with trilineage myelodysplasia have a lower remission rate 1. 2. Wheatley K, Burnett AK, Goldstone AH et al. Br J Haem 1999; 107: 69 -79 Grimwade D, Walker H, Oliver F et al. Blood 1998; 92: 2322 -33

Acute Myeloid Leukaemia (AML) Treatment and prognosis of AML • Intensive chemotherapy – Patients < 55 years old: 80% remissions – Patients > 55 years old: progressive reduction in remission rate • Bone marrow (stem cell) transplantation – Autologous and allogeneic transplants reduce the relapse rate • Importance of cytogenetics for prognosis in children and adults < 55 years old • Good risk cytogenetic group – 91% remissions, 65% five year survival 1. 2. Wheatley K, Burnett AK, Goldstone AH et al. Br J Haem 1999; 107: 69 -79 Grimwade D, Walker H, Oliver F et al. Blood 1998; 92: 2322 -33

Acute Lymphoblastic Leukaemia (ALL) Prognostic factors in ALL Poor Prognostic Factors • Age < 2 yrs and > 10 yrs • Male sex • High WBC count ( > 50 х109/L) • Presence of CNS disease • Cytogenetics Good risk Hyperdiploid (>50 ch) Poor risk Hypodiploid, t(9: 22), t(4: 11) • Bone Marrow: Blasts present on day 14 • Day 28: No complete response
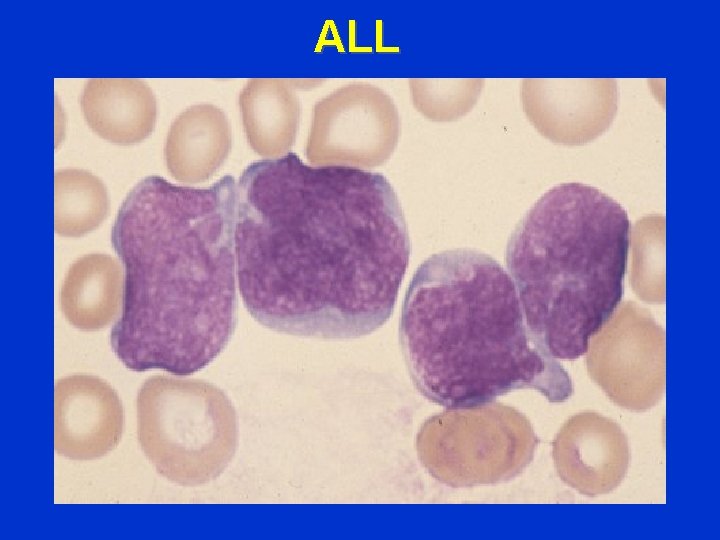
ALL
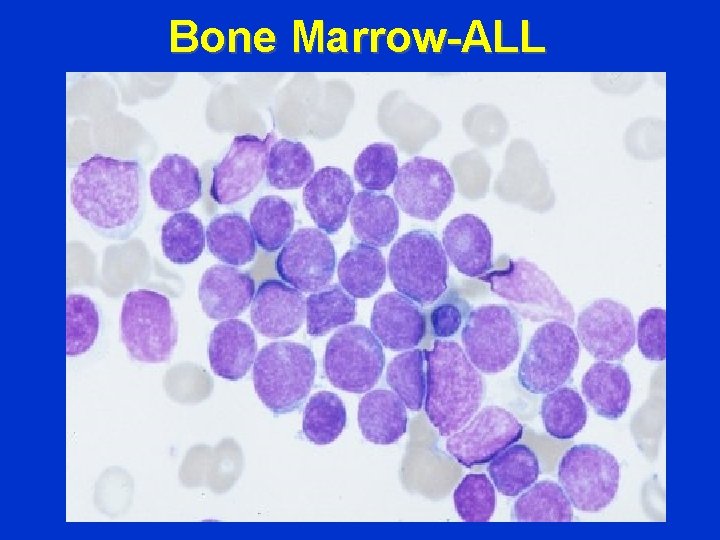
Bone Marrow-ALL

Treatment of acute leukemias 1. Specific therapy (chemotherapy) 2. Supportive treatment Stages of Therapy a. Induction b. Consolidation c. Maintenance
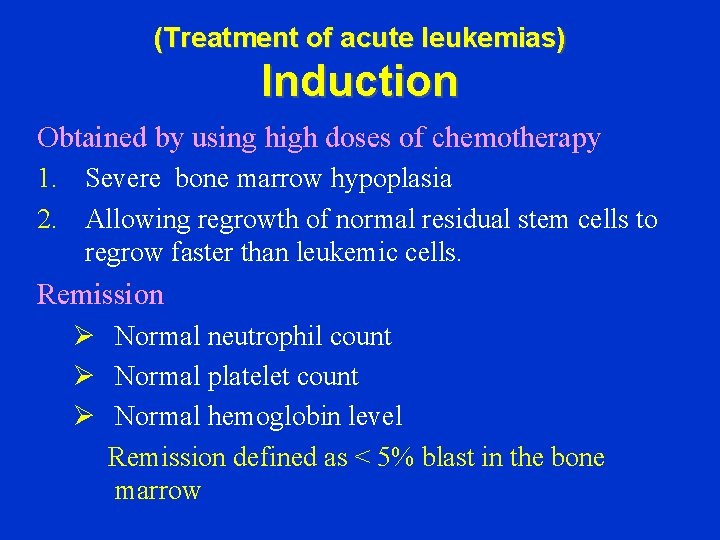
(Treatment of acute leukemias) Induction Obtained by using high doses of chemotherapy 1. Severe bone marrow hypoplasia 2. Allowing regrowth of normal residual stem cells to regrow faster than leukemic cells. Remission Ø Normal neutrophil count Ø Normal platelet count Ø Normal hemoglobin level Remission defined as < 5% blast in the bone marrow

(Treatment of acute leukemias) Consolidation • Different or same drugs to those used during induction • Higher doses of chemotherapy • Advantage: Delays relapse and improved survival

(Treatment of acute leukemias) Maintenance • Smaller doses for longer period • Produce low neutrophil counts & platelet counts • Objective is to eradicate progressively any remaining leukemic cells.

(Treatment of acute leukemias) Supportive Care 1. 2. 3. 4. Vascular access (Central line) Prevention of vomiting Blood products (Anemia, ↓Plat) Prevention & treatment of infections (antibiotics) 5 Management of metabolic complications

ALL vs AML ALL AML § Induction § Consolidation § Maintenance § No maintenance § CNS prophylaxis all patients § CNS – Selected group only

CHRONIC LEUKEMIAS Definition: Neoplastic proliferations of mature haemopoeitic cells. Types: Chronic lymphocytic leukemia (CLL) Chronic myeloid leukemia (CML)

CHRONIC LYMPHOCYTIC LEUKAEMIA (CLL) Ø Ø Neoplastic proliferations of mature lymphocytes. Distinguished from ALL by a. b. c. d. Morphology of cells. Degree of maturation of cells. Immunologically immature blasts in ALL. CLL affects mainly elderly.

SYMPTOMS of CLL Ø May be entirely absent in 40% Ø Weakness, easy fatigue, vague sense of being ill Ø Night sweats Ø Feeling of lumps Ø Infections esp pneumonia

PHYSICAL EXAMINAITON-CLL Ø Ø Pallor Lymphoadenopathy a. Cervical, supraclavicular nodes more commonly involved than axillary or inguino-femoral b. Non-tender, not painful, discrete, firm, easily movable on palpation Splenomegely, mild to moderate Hepatomegaly

CLINICAL STAGING-CLL

LABORATORY TESTS-CLL CBC Lymphocyte count > 5 x 109/L (5 -500 x 109/L ). Platelets may be decreased Hb may be low Blood film PB immunophenotyping Bone marrow biopsy (needed before starting treatment) Imaging

TREATMENT OF CLL Ø Ø Ø Observation Chemotherapy. Oral chlorambucil Fludarabine, cyclo Immunotherapy Anti-CD 20 (rituximab), Anti-CD 52 (Alemtuzumab) FC-R is the current standard Indications for starting chemotherapy a. b. c. d. Progressive Symptoms Progressive Anemia or Thrombocytopenia Bulky LN, large spleen Recurrent Infections

CHRONIC MYELOID LEUKEMIA Ø CML is a clonal stem cell disorder characterised by increased proliferation of myeloid elements at all stages of differentiation. Ø Incidence increases with age, M > F.

CML is characterised by 3 distinct phases a) Chronic Phase: Proliferation of myeloid cells, which show a full range of maturation. b) Accelerated Phase decrease in myeloid differentiation occurs. c) Blast crisis (acute leukemia)

CLINICAL PRESENTAITON OF CML Symptoms Ø Ø Ø Ø Asymptomatic (50% of patients) Fatigue Weight loss Abdominal fullness and anorexia Abdominal pain, esp splenic area Increased sweating Easy bruising or bleeding

SIGNS OF CML Ø Splenomegaly (95%) (50% of patients have a palpable spleen ≥ 10 cm BCM, Usually firm and non-tender) Ø Hepatomegaly (50%)

DIAGNOSIS OF CML Chronic phase. Ø Peripheral blood – neutrophil leukocytes 20, 000 - >500, 000/ L basphilia LAP score blasts < 5% Nucleated RBCs Thrombocytosis Anaemia

CYTOGENETICS OF CML Ø Philadelphia (Ph) chromosome is an acquired cytogenetic abnormality in all leukaemia cells in CML Ø Reciprocal translocation of chromosomal material between chromosome 22 and chromosome 9. t(9; 22)







CML-Treatment Response Criteria • Hematological response Normalisation of blood count • Cytogenetic response Major cytogenetic response 1 -35% Ph +ve cells in metaphase Minor cytogenetic response 36 -65% Ph +ve cells in metaphase • Molecular response Absence of BCR/ABL gene

CML-Principles of Treatment • Control & prolong chronic phase (noncurative) - Tyrosine kinase inhibitors-Imatinib (Glivec) - Alpha-Interferon - Oral chemotherapy (Hydroxyurea, ARA-C) • Eradicate malignant Clone (curative) - Allogeneic BM/stem cell transplantation - Alpha Interferon? - Imatinib? 2 nd line TKIs

TREATMENT OF CML • • • Tyrosine kinase inhibitor (TKI) Imatinib (Glivec) is the first line treatment In resistent cases 2 nd line TKIs (Nilotinib, Dasatinib, Bosutinib) very useful Allogenic bone marrow trasnsplantation can be curative in pts resisrant to TKIs but has significant complications & mortality Accelerated and blast phase Glivec, 2 nd line TKIs Treat like AML or ALL followed by BMT

CML VS LEUKEMOID REACTION 1. 2. 3. 4. LA P Score Philadelphia Chromosome Basophilia Splenomegaly

Bone marrow or PBSC transplantation in leukemias Types of transplant 1. Autologous transplant 2. Allogeneic Transplant Purpose of transplant Autologous -To deliver a high dose of chemo to kill any residual cancer (lymphoma, multiple myeloma) Allogeneic -To eradicate residual leukemia cells -Graft vs leukemia effect

Bone marrow or PBSC transplantation in leukemias Technique of transplantation ØMHC + HLA matching ØChemotherapy ØTotal body irradiation ØGVHD prophylaxis Complications of transplantation • • Prolonged BM suppression (graft failure) Serious infections Mucositis Graft versus host disease (GVHD)
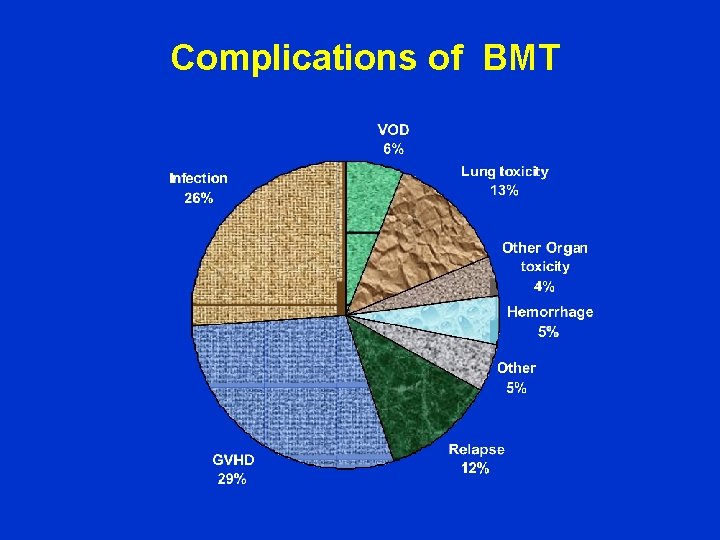
Complications of BMT
- Slides: 61