Lets build a smarter pharmaceutical supply chain Smarter

Let’s build a smarter pharmaceutical supply chain. Smarter, Safer Drugs Info. Sphere Traceability Server IBM Software Group

Smarter planet opportunities are everywhere. . smarter safe smarter solutions pharmaceuticals for retail smarter data center smarter healthcare smarter supply chains smarter transportation smarter food smarter industrial manufacturing operations smarter traffic smarter water management smarter cities 2

Serialization and the New Era of Visibility The ability to obtain and verify the location, status and history of an item. Pharmaceuticals: Can I be sure my prescription drugs are not counterfeit? Parts: Which parts were assembled in my car and are they authentic? Food: Where does my food come from? Returnable Containers: Do my suppliers have enough containers to meet my production needs? The IBM Supply Chain solution enables customers in multiple industries to answer these questions today. 3

The world is changing and becoming more… Instrumented Interconnected Intelligent The explosion of new information, when integrated, analyzed, and acted upon using new types of intelligence, enables solutions that … help build Smarter Safer Drugs
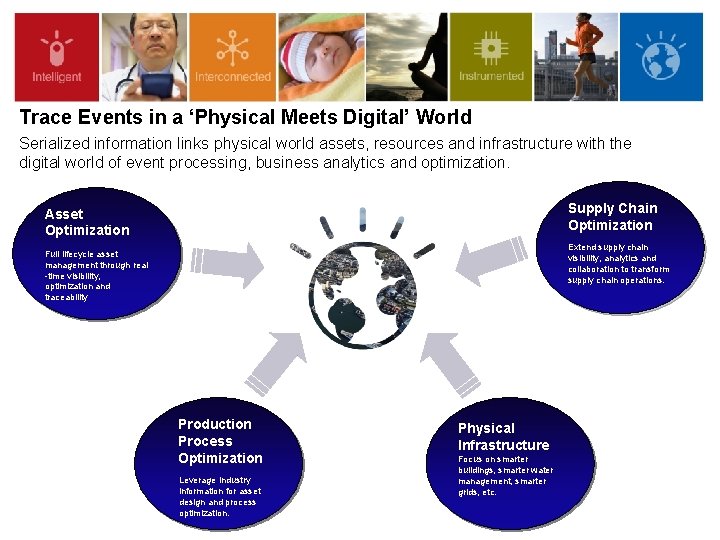
Trace Events in a ‘Physical Meets Digital’ World Serialized information links physical world assets, resources and infrastructure with the digital world of event processing, business analytics and optimization. Supply Chain Optimization Asset Optimization Extend supply chain visibility, analytics and collaboration to transform supply chain operations. Full lifecycle asset management through real -time visibility, optimization and traceability Production Process Optimization Leverage industry information for asset design and process optimization. Physical Infrastructure Focus on smarter buildings, smarter water management, smarter grids, etc.

Supply Chain Visibility Can Produce Better Outcomes Stock-Outs – Excess safety 4. 5% of revenue is lost due to supply chain inefficiencies. stock is 10% of inventory at Manufacturers and $700 -800 K of excess inventory at each hospital and retail pharmacy - IDC 8 -10% of Worldwide Drug Supply is Counterfeit – up to $75 billion a year in lost sales - WHO 6

Global Serialization Trends Concern for counterfeit drugs, diversion and patient safety is driving a global trend with legislative and serialization requirements IBM provides a common platform to allow flexibility to accommodate new standards EU Panel Votes to Crack Down on Illegal Medicines ASSOCIATED PRESS April 27, 2010 BRUSSELS - The battle against counterfeit medicines took a big step forward Tuesday when the European Parliament public health panel approved a bill to crack down on a fast growing industry worth (EURO)10. 5 billion ($14 billion) a year in Europe alone. If it becomes law, drug makers must produce medicines that can be tracked from the factory to the patient to make sure no one tampers with the content.

Trusted Physical Data is Converted into Usable Information Reporting & Analytics Xylinke Blood Thinner ü End-to-end Traceability ü Inventory Visibility ü Temperature Tracking Business Benefits ü ü Increased supply chain visibility Reduced counterfeiting and diversion Increased Consumer Safety Increased efficiency for conducting recalls and returns TRUSTED Pharma Manufacturer INFORMATION Distribution/ Export Retailer / Hospital 8
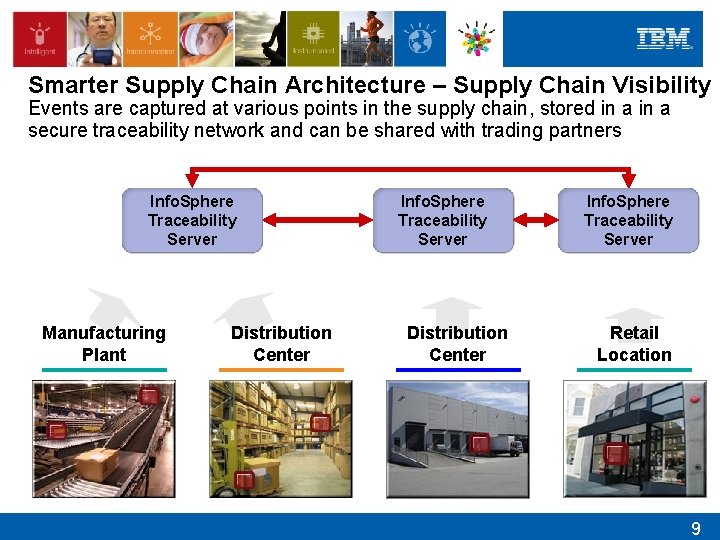
Smarter Supply Chain Architecture – Supply Chain Visibility Events are captured at various points in the supply chain, stored in a secure traceability network and can be shared with trading partners Info. Sphere Traceability Server Manufacturing Plant Distribution Center Info. Sphere Traceability Server Retail Location 9
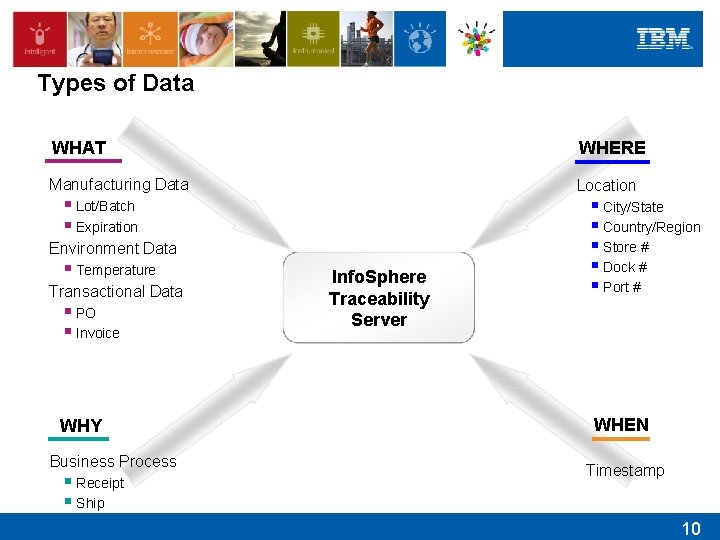
Types of Data WHAT WHERE Manufacturing Data Location § Lot/Batch § Expiration Environment Data § Temperature Transactional Data § PO § Invoice WHY Business Process § Receipt § Ship Info. Sphere Traceability Server § City/State § Country/Region § Store # § Dock # § Port # WHEN Timestamp 10
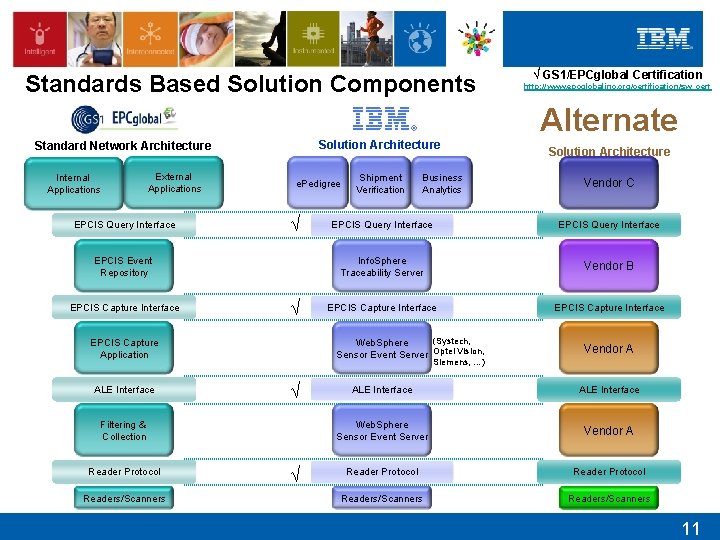
Standards Based Solution Components √ GS 1/EPCglobal Certification http: //www. epcglobalinc. org/certification/sw_cert Alternate Solution Architecture Standard Network Architecture Internal Applications External Applications EPCIS Query Interface e. Pedigree √ EPCIS Event Repository EPCIS Capture Interface √ Readers/Scanners Vendor C EPCIS Query Interface Info. Sphere Traceability Server Vendor B EPCIS Capture Interface Vendor A Siemens, …) √ Filtering & Collection Reader Protocol Business Analytics (Systech, Web. Sphere Sensor Event Server Optel Vision, EPCIS Capture Application ALE Interface Shipment Verification Solution Architecture √ ALE Interface Web. Sphere Sensor Event Server Vendor A Reader Protocol Readers/Scanners 11
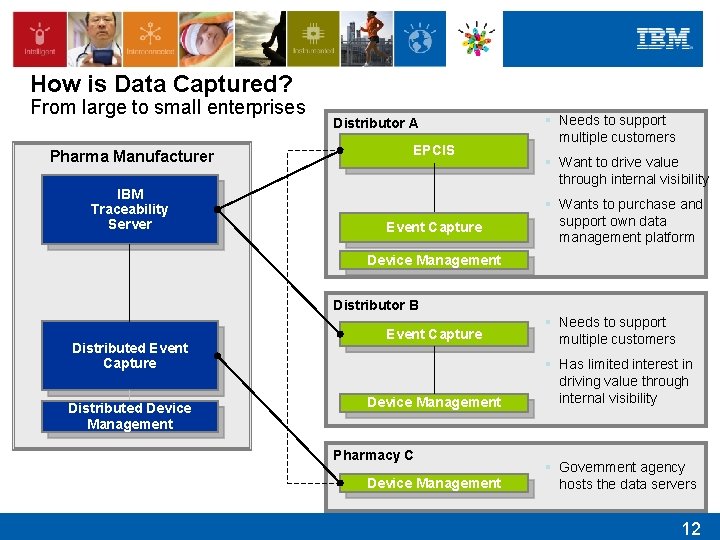
How is Data Captured? From large to small enterprises Pharma Manufacturer IBM Traceability Server Distributor A EPCIS Event Capture § Needs to support multiple customers § Want to drive value through internal visibility § Wants to purchase and support own data management platform Device Management Distributor B Distributed Event Capture Distributed Device Management Event Capture Device Management Pharmacy C Device Management § Needs to support multiple customers § Has limited interest in driving value through internal visibility § Government agency hosts the data servers 12
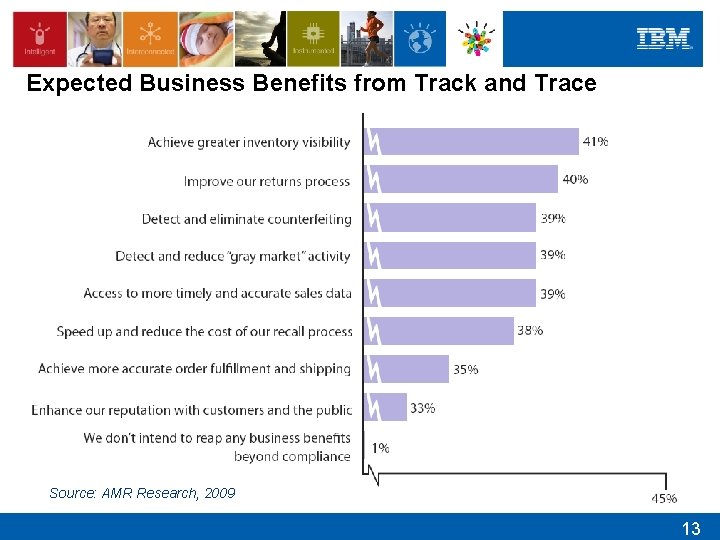
Expected Business Benefits from Track and Trace Source: AMR Research, 2009 13
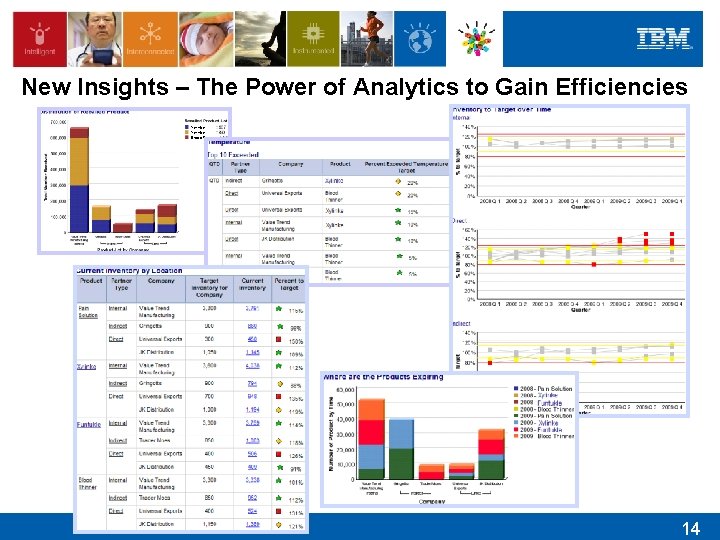
New Insights – The Power of Analytics to Gain Efficiencies Xylinke Blood Thinner 14
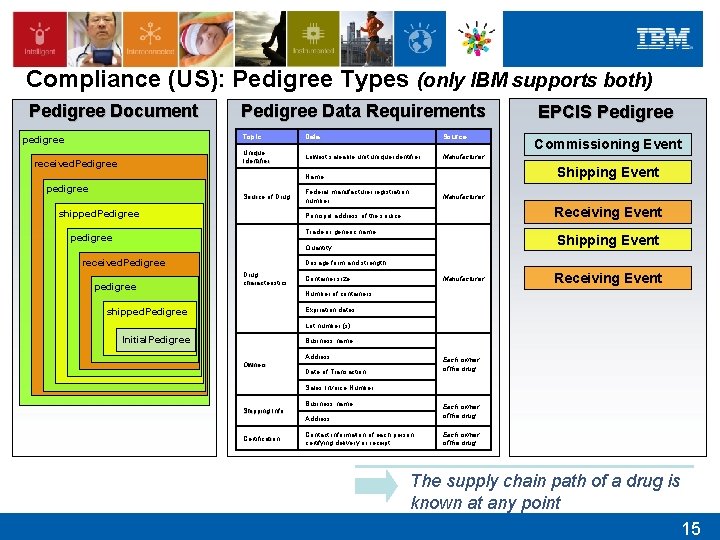
Compliance (US): Pedigree Types (only IBM supports both) Pedigree Document pedigree received. Pedigree Data Requirements Topic Data Source Unique Identifier Lowest saleable unit unique identifier Manufacturer Source of Drug shipped. Pedigree Federal manufacturer registration number Manufacturer Receiving Event Principal address of the source Trade or generic name pedigree Shipping Event Quantity received. Pedigree pedigree Commissioning Event Shipping Event Name pedigree EPCIS Pedigree Dosage form and strength Drug characteristics Container size Manufacturer Receiving Event Number of containers shipped. Pedigree Expiration dates Lot number (s) Initial. Pedigree Business name Owners Address Each owner of the drug Date of Transaction Sales Invoice Number Shipping Info Certification Business name Each owner of the drug Address Contact information of each person certifying delivery or receipt Each owner of the drug The supply chain path of a drug is known at any point 15

IBM’s Unique Value Proposition 1. IBM offers the most complete solution components: Device Management, Capture Middleware, EPCIS, and Pedigree App (both Document and EPCIS Pedigree) – the only “one stop shop” 2. IBM employs 400, 000 professionals, operates in 170 countries, and has local expertise and support in any part of the world 3. IBM has been working with many global regulatory agencies: FDA US (drug and devices regulation), EFPIA, Brazil, Turkey 4. IBM solution has been deployed for different use cases in many industries for cross-learning opportunity 5. IBM solution provides low operational risk: redundancy, HA, and fail-over contingency 6. IBM solution enables extension to business applications such as: consignment inventory, supply chain visibility, cold-chain management, chargeback resolution, targeted recall 7. IBM solution can easily be integrated with many other complementary suite of products: ILOG, Maximo, MDM, Cognos 16

Workshop Invitation § Ready to learn more about how IBM can help your business? § IBM experts will arrange to meet with your team to evaluate your current readiness and make suggestions for next phase deployment needs. § Our teams have worked with several manufacturers and distributors globally, and can help you define a process roadmap to obtain serialization, compliance and help offset on-going costs by evaluating overall business process efficiencies.

Thank you for your Attention Smarter, Safer Drugs Info. Sphere Traceability Server IBM Software Group

BACKUP SLIDES
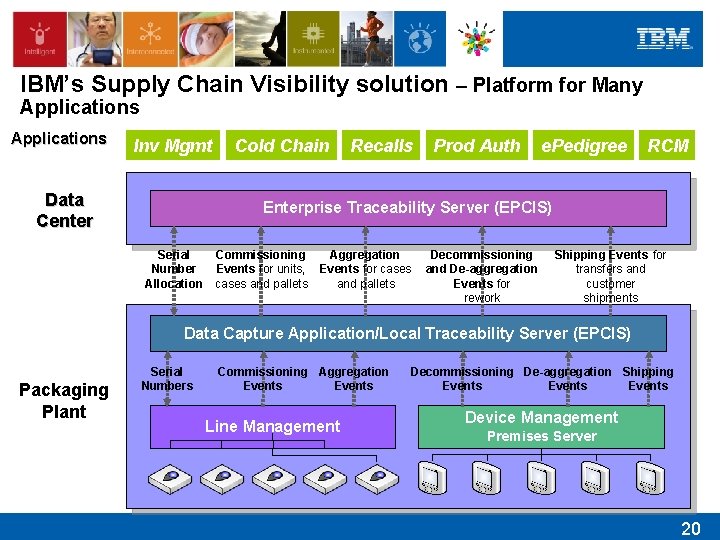
IBM’s Supply Chain Visibility solution – Platform for Many Applications Inv Mgmt Data Center Cold Chain Recalls Prod Auth e. Pedigree RCM Enterprise Traceability Server (EPCIS) Serial Commissioning Number Events for units, Allocation cases and pallets Aggregation Events for cases and pallets Decommissioning and De-aggregation Events for rework Shipping Events for transfers and customer shipments Data Capture Application/Local Traceability Server (EPCIS) Packaging Plant Serial Numbers Commissioning Aggregation Events Line Management Decommissioning De-aggregation Shipping Events Device Management Premises Server 20
Governments & Industry are concerned for patient safety 21

Regulations are Increasing World-wide California FDA Pedigree and serialization regulations are a growing trend among domestic and international regulatory bodies Brazil EU Turkey China Korea

Risk Management: Product Authentication & Diversion Detection Challenges • High risk of product counterfeit and diversion • High theft • Protecting brand identity • Increased inventory stock WHO estimates counterfeit drug sales between $35 and 40 billion per year • Between 8 and 10% of the worldwide drug supply is estimated to be counterfeit • By 2010, more than $75 billion of sales will be lost per year Benefits • Minimize lost revenue due to counterfeits • Minimize lost revenue due to diversions • Increase patient and consumer safety • Brand protection and risk mitigation • “Leveraging Track and Trace in the Pharmaceutical Industry”, IBM Whitepaper © 2009 Companies can identify counterfeit or stolen product entering their supply chain 23
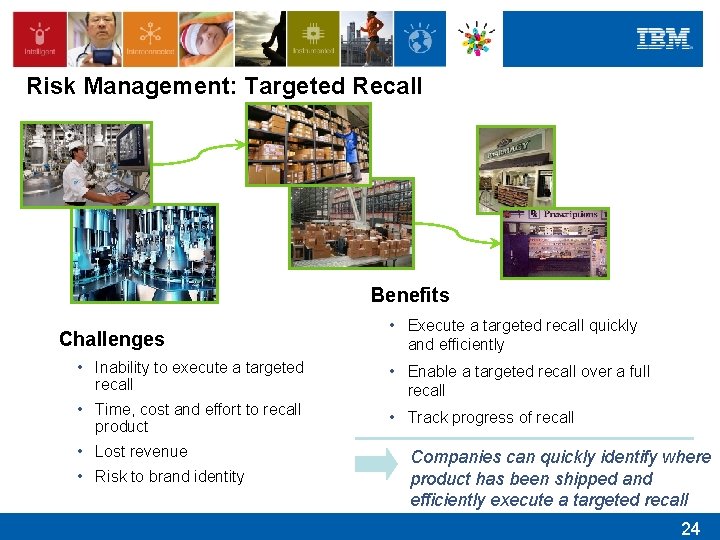
Risk Management: Targeted Recall Benefits Challenges • Execute a targeted recall quickly and efficiently • Inability to execute a targeted recall • Enable a targeted recall over a full recall • Time, cost and effort to recall product • Track progress of recall • Lost revenue • Risk to brand identity Companies can quickly identify where product has been shipped and efficiently execute a targeted recall 24
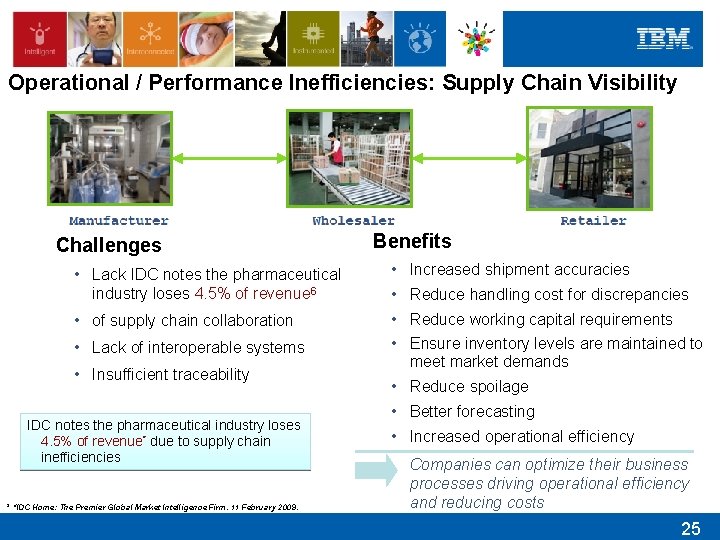
Operational / Performance Inefficiencies: Supply Chain Visibility Challenges Benefits • Lack IDC notes the pharmaceutical industry loses 4. 5% of revenue 6 • Increased shipment accuracies • of supply chain collaboration • Reduce working capital requirements • Lack of interoperable systems • Ensure inventory levels are maintained to meet market demands • Insufficient traceability IDC notes the pharmaceutical industry loses 4. 5% of revenue* due to supply chain inefficiencies * “IDC Home: The Premier Global Market Intelligence Firm. 11 February 2009. • Reduce handling cost for discrepancies • Reduce spoilage • Better forecasting • Increased operational efficiency Companies can optimize their business processes driving operational efficiency and reducing costs 25

Operational / Performance Inefficiencies: Cold Chain Management Challenges • High spoilage • Lack of compliance validation Benefits • Improved product efficacy and safety • Reduced spoilage • Proof of SOP – regulatory • Track temperature and shelf-life data • IMS Health reports worldwide sales of biologic medications, which require temperaturecontrolled storage and shipping conditions, increased by 12. 5% in 2007 $75 billion • Nearly double the 6. 4% increase in sales of traditional pharmaceuticals Companies can increase product safety and prevent spoilage due to inefficient processes 26
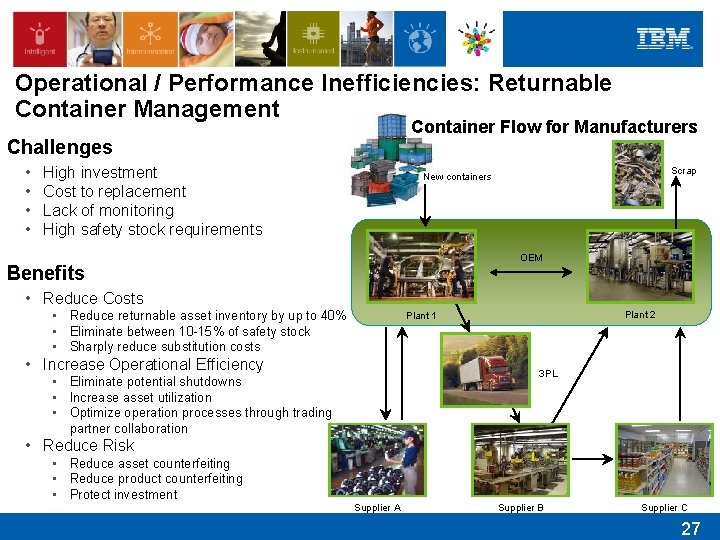
Operational / Performance Inefficiencies: Returnable Container Management Container Flow for Manufacturers Challenges • • High investment Cost to replacement Lack of monitoring High safety stock requirements Scrap New containers OEM Benefits • Reduce Costs • Reduce returnable asset inventory by up to 40% • Eliminate between 10 -15% of safety stock • Sharply reduce substitution costs Plant 2 Plant 1 • Increase Operational Efficiency 3 PL • Eliminate potential shutdowns • Increase asset utilization • Optimize operation processes through trading partner collaboration • Reduce Risk • Reduce asset counterfeiting • Reduce product counterfeiting • Protect investment Supplier A Supplier B Supplier C 27
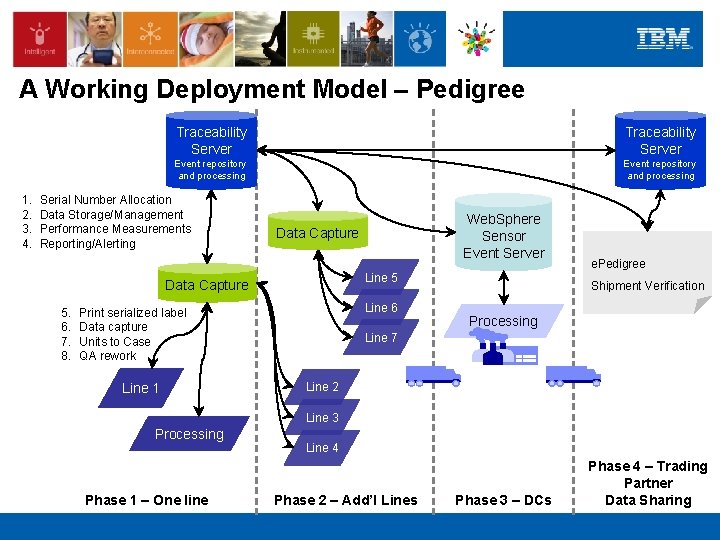
A Working Deployment Model – Pedigree 1. 2. 3. 4. Traceability Server Event repository and processing Serial Number Allocation Data Storage/Management Performance Measurements Reporting/Alerting Data Capture Line 5 Data Capture 5. 6. 7. 8. Line 6 Print serialized label Data capture Units to Case QA rework Line 1 Web. Sphere Sensor Event Server e. Pedigree Shipment Verification Processing Line 7 Line 2 Line 3 Processing Phase 1 – One line Line 4 Phase 2 – Add’l Lines Phase 3 – DCs Phase 4 – Trading Partner Data Sharing
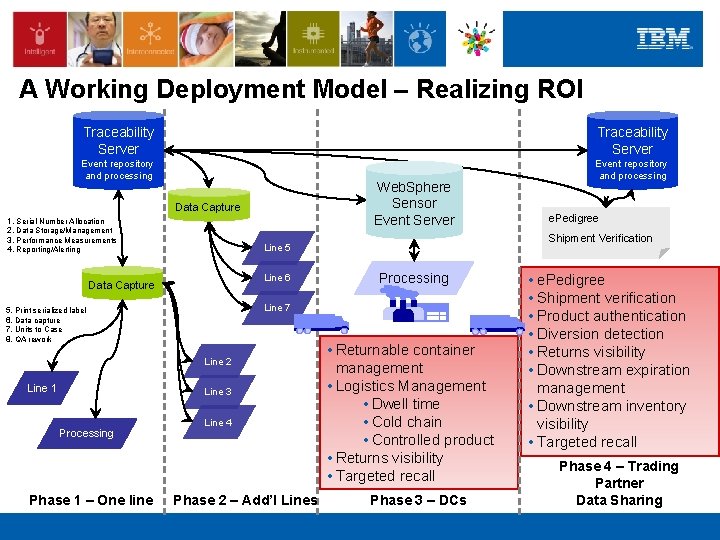
A Working Deployment Model – Realizing ROI Traceability Server Event repository and processing Web. Sphere Sensor Event Server Data Capture 1. Serial Number Allocation 2. Data Storage/Management 3. Performance Measurements 4. Reporting/Alerting Line 6 Line 2 Line 3 Processing Phase 1 – One line Processing Line 7 5. Print serialized label 6. Data capture 7. Units to Case 8. QA rework Line 1 Shipment Verification Line 5 Data Capture Line 4 Phase 2 – Add’l Lines e. Pedigree • Returnable container management • Logistics Management • Dwell time • Cold chain • Controlled product • Returns visibility • Targeted recall Phase 3 – DCs • e. Pedigree • Shipment verification • Product authentication • Diversion detection • Returns visibility • Downstream expiration management • Downstream inventory visibility • Targeted recall Phase 4 – Trading Partner Data Sharing
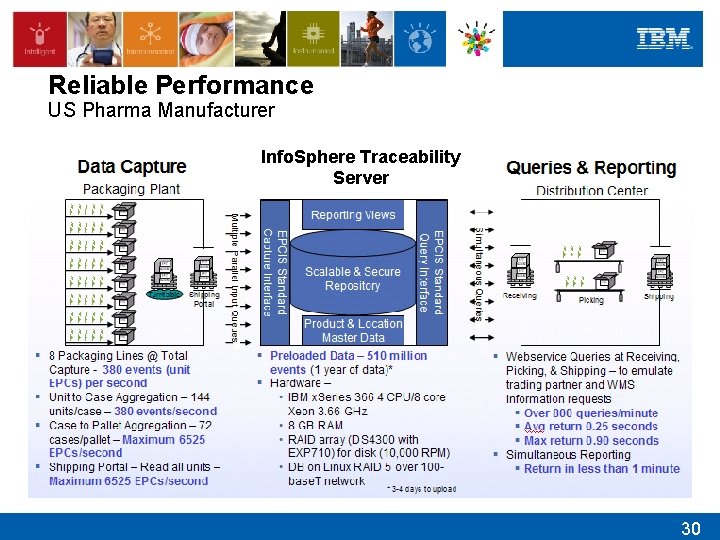
Reliable Performance US Pharma Manufacturer Info. Sphere Traceability Server 30
- Slides: 30