Lessons Learned from the Step Study Susan Buchbinder

Lessons Learned from the Step Study Susan Buchbinder, MD HIV Research Section San Francisco Department of Public Health International AIDS Society Meeting Mexico City August 4, 2008
MRKAd 5 trivalent vaccine • Vaccine: 1: 1: 1 admixture of 3 Ad 5 vectors – Encoded transgenes: codon-optimized, near-consensus clade B HIV-1 sequences • Placebo: vaccine dilution buffer without Ad 5 2

Trial Design • 3000 high-risk HIV uninfected men and women – Initial study: 1500 pts w/ Ad 5 NAb <200 (Dec 2004) – Modification: additional 1500 w/ Ad 5 NAb >200 (July 2005) – Randomization stratified by Ad 5 <18, 19 -200, 201 -1000, >1000 Primary hypotheses: Ad 5 <200 subset • Decrease in HIV acquisition and/or • Lower viral load setpoint (∼ 3 months post-diagnosis) Secondary hypotheses: Total population • Same as primary (Ad 5 <200 and Ad 5 >200 combined) 3
Step Study sites 4
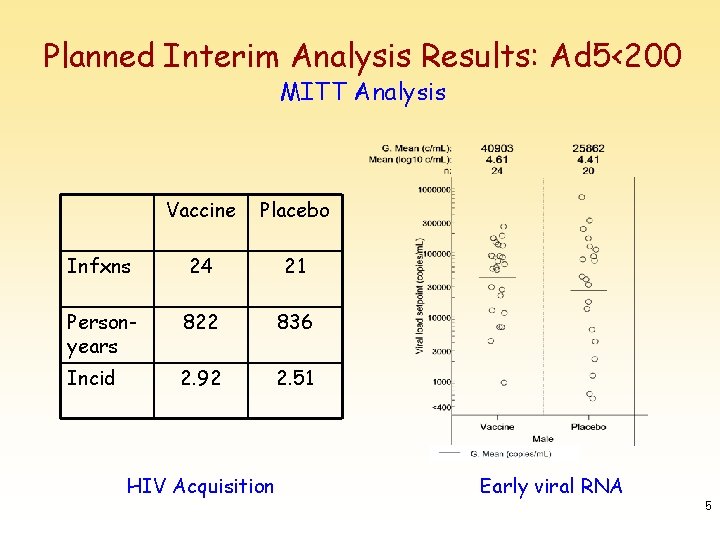
Planned Interim Analysis Results: Ad 5<200 MITT Analysis Vaccine Placebo Infxns 24 21 Personyears 822 836 Incid 2. 92 2. 51 HIV Acquisition Early viral RNA 5
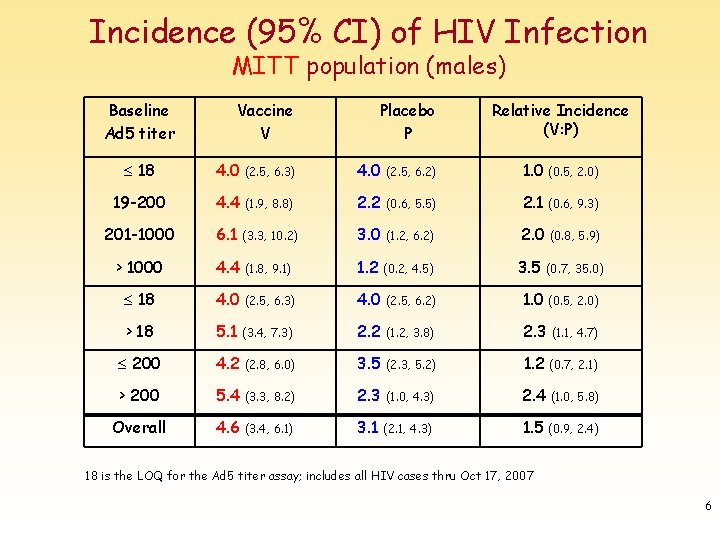
Incidence (95% CI) of HIV Infection MITT population (males) Baseline Ad 5 titer Vaccine V Placebo P Relative Incidence (V: P) 18 4. 0 (2. 5, 6. 3) 4. 0 (2. 5, 6. 2) 1. 0 (0. 5, 2. 0) 19 -200 4. 4 (1. 9, 8. 8) 2. 2 (0. 6, 5. 5) 2. 1 (0. 6, 9. 3) 201 -1000 6. 1 (3. 3, 10. 2) 3. 0 (1. 2, 6. 2) 2. 0 (0. 8, 5. 9) > 1000 4. 4 (1. 8, 9. 1) 1. 2 (0. 2, 4. 5) 3. 5 (0. 7, 35. 0) 18 4. 0 (2. 5, 6. 3) 4. 0 (2. 5, 6. 2) 1. 0 (0. 5, 2. 0) > 18 5. 1 (3. 4, 7. 3) 2. 2 (1. 2, 3. 8) 2. 3 (1. 1, 4. 7) 200 4. 2 (2. 8, 6. 0) 3. 5 (2. 3, 5. 2) 1. 2 (0. 7, 2. 1) > 200 5. 4 (3. 3, 8. 2) 2. 3 (1. 0, 4. 3) 2. 4 (1. 0, 5. 8) Overall 4. 6 (3. 4, 6. 1) 3. 1 (2. 1, 4. 3) 1. 5 (0. 9, 2. 4) 18 is the LOQ for the Ad 5 titer assay; includes all HIV cases thru Oct 17, 2007 6

Variables included in univariate/multivariate analyses • • Vaccine vs. placebo Baseline Ad 5 Circumcision (self-report) Age Race Region Baseline risk factors (previous 6 months) – # male sex partners – Unprotected receptive anal sex – Unprotected insertive anal sex – Substance use – Self-reported sexually transmitted infection 7

Variables included in univariate/multivariate analyses • • Vaccine vs. placebo Baseline Ad 5* Circumcision (self-report)* Age Race Region Baseline risk factors (previous 6 months) – # male sex partners – Unprotected receptive anal sex – Unprotected insertive anal sex – Substance use – Self-reported sexually transmitted infection *significant interaction with vaccine vs. placebo 8
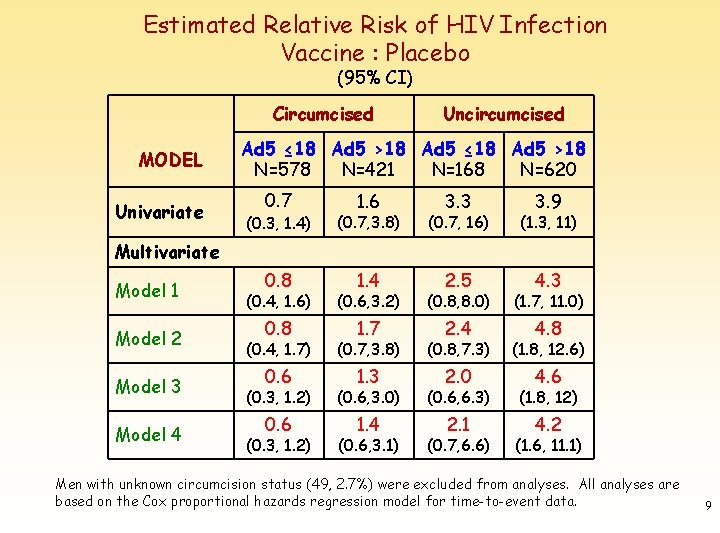
Estimated Relative Risk of HIV Infection Vaccine : Placebo (95% CI) Circumcised MODEL Univariate Uncircumcised Ad 5 ≤ 18 Ad 5 >18 N=578 N=421 N=168 N=620 0. 7 1. 6 3. 3 3. 9 (0. 3, 1. 4) (0. 7, 3. 8) (0. 7, 16) (1. 3, 11) 0. 8 1. 4 2. 5 4. 3 Multivariate Model 1 Model 2 Model 3 Model 4 (0. 4, 1. 6) (0. 6, 3. 2) (0. 8, 8. 0) (1. 7, 11. 0) 0. 8 1. 7 2. 4 4. 8 (0. 4, 1. 7) (0. 7, 3. 8) (0. 8, 7. 3) (1. 8, 12. 6) 0. 6 1. 3 2. 0 4. 6 (0. 3, 1. 2) (0. 6, 3. 0) (0. 6, 6. 3) (1. 8, 12) 0. 6 1. 4 2. 1 4. 2 (0. 3, 1. 2) (0. 6, 3. 1) (0. 7, 6. 6) (1. 6, 11. 1) Men with unknown circumcision status (49, 2. 7%) were excluded from analyses. All analyses are based on the Cox proportional hazards regression model for time-to-event data. 9
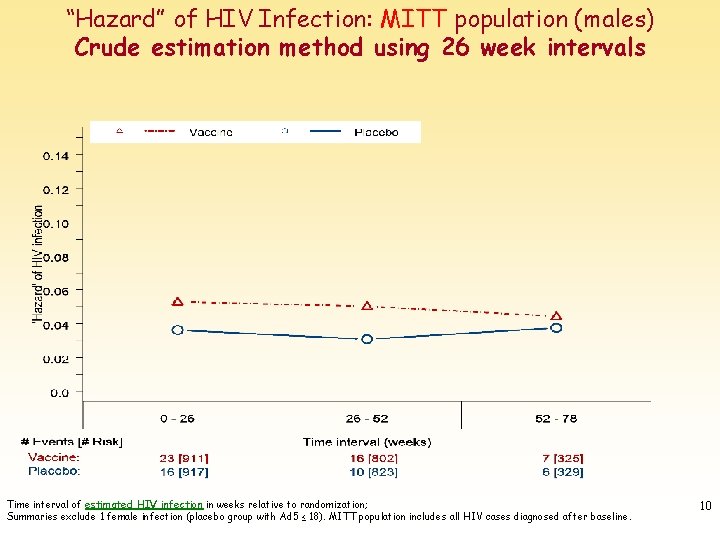
“Hazard” of HIV Infection: MITT population (males) Crude estimation method using 26 week intervals Time interval of estimated HIV infection in weeks relative to randomization; Summaries exclude 1 female infection (placebo group with Ad 5 ≤ 18). MITT population includes all HIV cases diagnosed after baseline. 10

Next Steps • Ongoing follow-up of study volunteers – All pts unblinded – >95% retention since results announced – HVTN 504 to • Evaluate longer-term effects • Provide risk reduction counseling, linkage to prevention services • Collect additional data and specimens • Awaiting data on – HSV-2 status, HLA typing, sexual network clustering – Multivariate analysis with longitudinal risk – Laboratory investigation of reasons for failure, control of viremia • Opening up data, specimens to external investigators – External Scientific Review Committee prioritizes laboratory studies, specimens 11

Important Scientific Lessons from Step • Demonstrated utility of test-of-concept trials – Quicker awareness of beneficial and adverse effects • Recalibrated the NHP challenge model – SHIV 89. 6 P not useful – Consideration of genetic factors (MAMU-A 01) • New questions not easily addressed in NHP studies – Importance of vector immunity, incl. tissue-specific responses – Transmission across mucosal barriers • Data and specimens to directly address failure to protect – Need to develop validated, functional assays as correlates – Exploration of breakthrough infections • Intriguing leads in potential protection in subgroups 12

Important “Community” Lessons from Step • Transparency is critical • Prepare (as best you can) for the unexpected – Communication w/ investigators, participants, public • Each prevention trial impacts on all others • Community is important at each stage of a trial • Study volunteers are critical to our search for an HIV vaccine 13

Conclusions • The Step Study was a pivotal trial for the HIV vaccine field • Definitive results on vaccine efficacy reached 33 months after first participant enrolled • Important contributions that could only have been learned through clinical efficacy trials • Lessons continue to be mined from trial data and specimens. 14

Acknowledgments The Study Volunteers For their dedication and commitment in the search for an HIV vaccine

STEP Study Protocol Team • • • • Sarah Alexander Susan Buchbinder Gail Broder Lisa Bull Danny Casimiro Ann Duerr Cheryl Ewing Dan Fitzgerald Paula Frew Lori Gabryelski Peter Gilbert Tirzah Griffin Soyon Im Dale Lawrence • • • • Rosario Leon David Li Ellen Mac. Lachlan Julie Mc. Elrath Devan Mehrotra Robin Mogg Dewayne Mullis Gabriela O’Neill Mary Pleier Michael Robertson Steve Self Rosario Leon Amanda Vettori Steve Wakefield Amy Zhou 16
- Slides: 16