Lesson 8 42 A Ionic Bonds Whats here

Lesson 8. 4_2 A: Ionic Bonds • What’s here: – Review: how p+ and e- add up to an atom’s electrical charge. – Ions: cations and anions, & polyatomic ions – Ionic chemical bonds

Drill A. How many valence electrons does calcium have? B. Draw the electron dot diagram for calcium? C. What will calcium have to do to become a stable atom?

Examining Ions Red = Electron 1) 2) 3) 4) 5) 6) white = Proton Place 1 pair of chips (1 red and 1 white) in a group on your desk. Place 7 pairs of chips (7 red and 7 white) in a group on your desk. Move 1 red chip from the smaller group to the larger group. What is the overall charge for the larger group? What is the overall charge for the smaller group? What is the overall charge for both groups combined?

Examining Ions Red = Electron 1) 2) 3) 4) 5) 6) White = Proton Place 3 pairs of chips (3 red and 3 white) in a group on your desk. Place 5 pairs of chips (5 red and 5 white) in a group on your desk. Move a 3 red chips from the smaller group to the larger group. What is the overall charge for the larger group? What is the overall charge for the smaller group? What is the overall charge of both groups combined?

Examining Ions A. What happens to the charge of an atom when it loses electrons? B. What happens to the charge of an atom when it gains electrons? C. Why did one group end up with 8 electrons?

At the top of your notes… Date: INSERT DATE describe an ionic bond Objective: SWBAT

1) Ions A) An atom or group of atoms that have an electric charge B) When an atom loses an electron, it loses a negative charge and becomes a positive ion and is called an cation. C) When an atom gains an electron, it gains a negative charge and becomes a negative ion and is called an anion. D) Examples

Mnemonic Thinking: Anion and Cation • A Negative. ION • if anion means negative, then the other one, cation, must mean positive. A “mnemonic device” is a certain way to remember and recall information. These examples are anagram and conditional opposite.
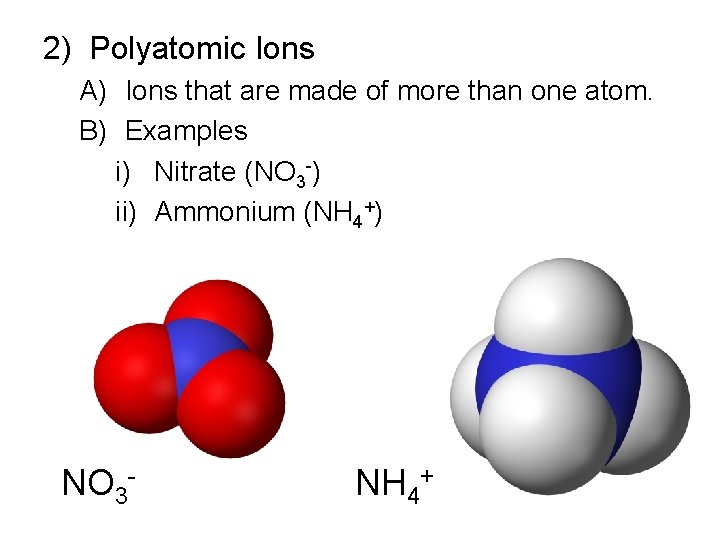
2) Polyatomic Ions A) Ions that are made of more than one atom. B) Examples i) Nitrate (NO 3 -) ii) Ammonium (NH 4+) NO 3 - NH 4+
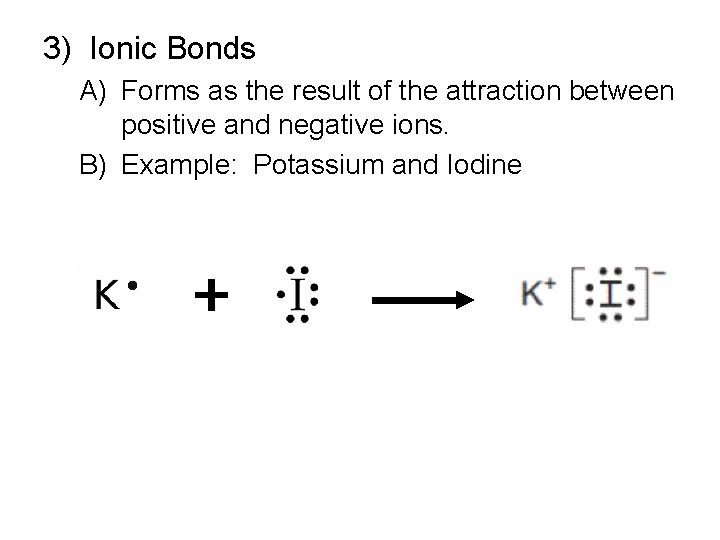
3) Ionic Bonds A) Forms as the result of the attraction between positive and negative ions. B) Example: Potassium and Iodine

3) Ionic Bonds C) Example: Magnesium and Chlorine The formula is Mg. Cl 2. Can you explain why it must be Mg. Cl 2 and not Mg. Cl?

Your Turn • Draw the electron dot diagram for calcium. • Draw the electron dot diagram for oxygen. • Draw the ionic bond between calcium and oxygen.

Your Turn • Draw the electron dot diagram for lithium. • Draw the electron dot diagram for sulfur. • How could lithium and sulfur combine in an ionic bond?

Please take out Tuesday’s Homework (Ionic Bonding Worksheet) for a quick check. Drill: Day 2 Cabonate is a polyatomic ion with the formula CO 3 -2. Carbonic acid is an ionic compound made of carbonate and hydrogen ions Draw the electron dot diagram for a molecule of carbonic acid.

Ocean Acidification from Atmospheric CO 2 • Acids have lots of H+ ions. This is how CO 2 in the atmosphere causes ocean acidification

Calcium Carbonate • Draw the electron dot diagram for calcium carbonate. • THINK: Why would more carbonic acid make it harder for sea creatures to make calcium carbonate shells?

Baking Soda • Baking Soda (which is also the main ingredient of baking powder) is sodium bicarbonate. • Draw the electron dot diagram for sodium bicarbonate. It includes a carbonate ion, a hydrogen ion, and a sodium ion.
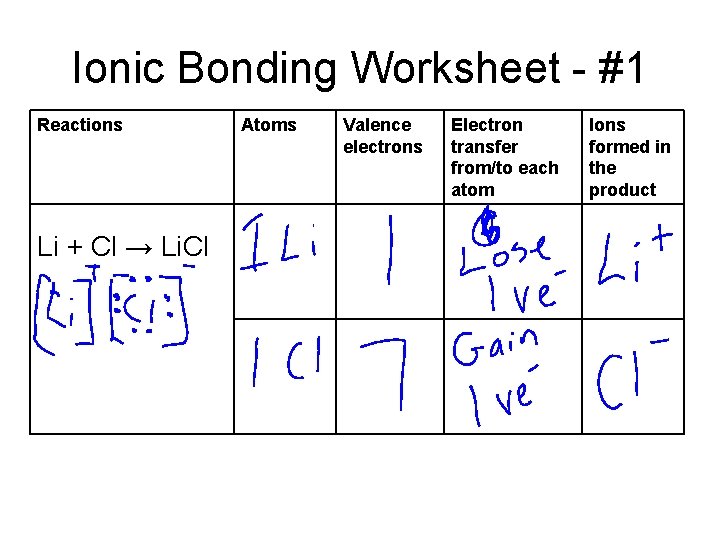
Ionic Bonding Worksheet - #1 Reactions Li + Cl → Li. Cl Atoms Valence electrons Electron transfer from/to each atom Ions formed in the product
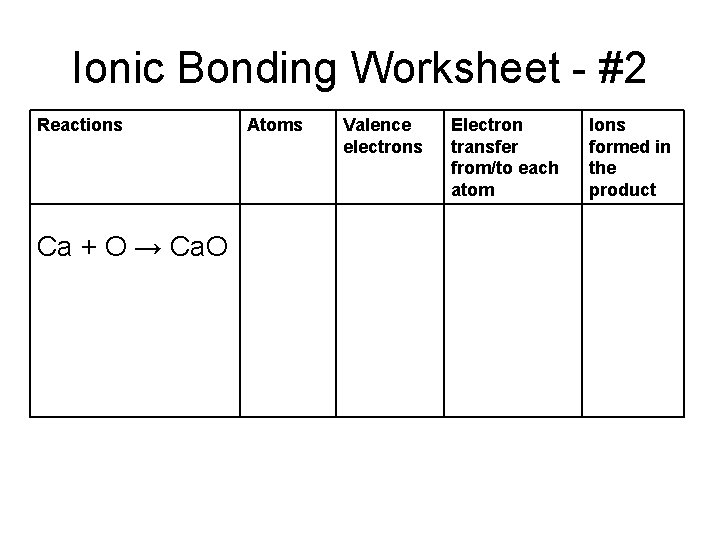
Ionic Bonding Worksheet - #2 Reactions Ca + O → Ca. O Atoms Valence electrons Electron transfer from/to each atom Ions formed in the product
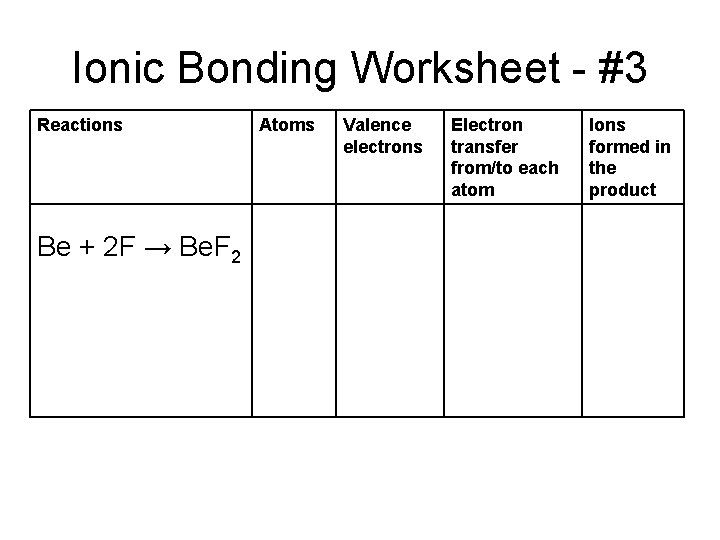
Ionic Bonding Worksheet - #3 Reactions Be + 2 F → Be. F 2 Atoms Valence electrons Electron transfer from/to each atom Ions formed in the product
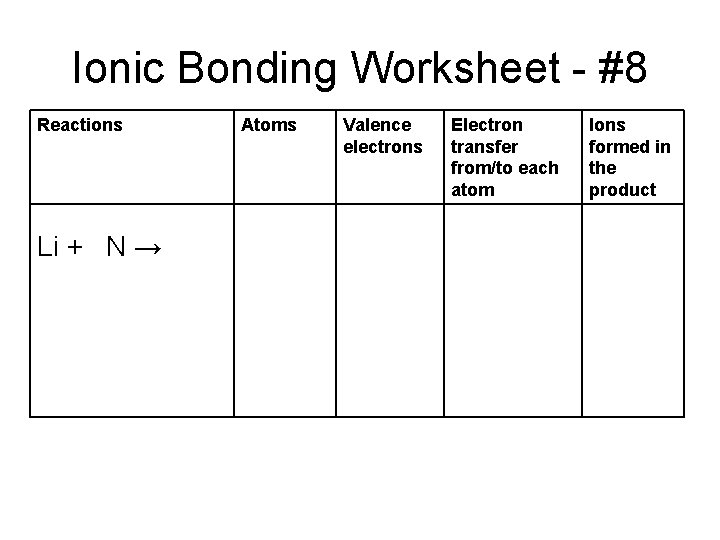
Ionic Bonding Worksheet - #8 Reactions Li + N → Atoms Valence electrons Electron transfer from/to each atom Ions formed in the product
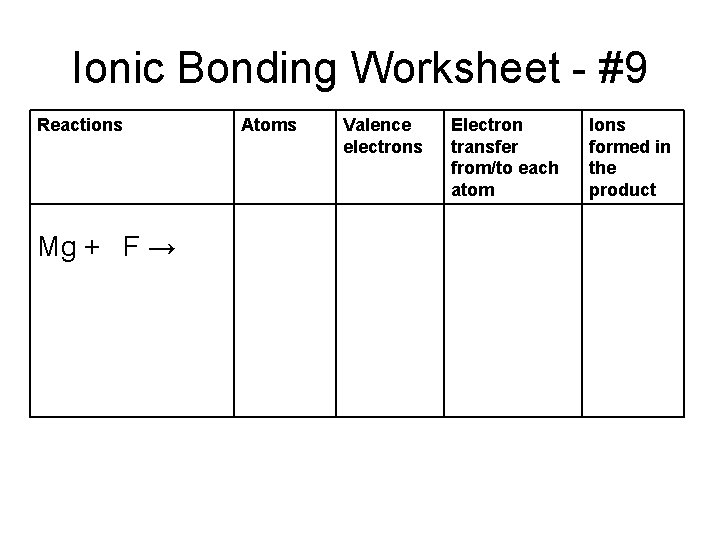
Ionic Bonding Worksheet - #9 Reactions Mg + F → Atoms Valence electrons Electron transfer from/to each atom Ions formed in the product

IONIC BOND VIDEO • https: //www. youtube. com/watch? v=zpa. HP XVR 8 WU

Continue your notes from Wednesday… 4) Chemical Formula A) The combination of symbols that shows the ratio of elements in a compound B) For an ionic bond, the chemical formula represents how the ions come together to cancel out the charges. C) Example.

5) Compound Name A) The name of the positive ion comes first, followed by the name of the negative ion. B) If the negative ion is a single element, the end of its name changes to ide. C) If the negative ion is a polyatomic ion, the end of its name changes to ate or ite. D) We do not use prefixes for ionic compounds.

5) Compound Name (continued) E) Examples Na. Cl = Mg. O = Li 2 SO 3 = Al. F 3 = Na. NO 3 =

HOMEWORK Ionic Bond Worksheet
- Slides: 27